4.2.3 Sweetpotato Pests / Sweetpotato butterfly, Acraea acerata (Hewitson 1874)![]()
Synonyms: Actinote acerata (Hewitson 1874)
Acraea vinidia (Hewitson 1874)
Acraea tenella (Rogenhofer 1891)
Telchinia tenella (Rogenhofer 1891)
Acraea abbotti (Holland 1892)
Acraea brahmsi (Suffert 1904)
Acraea vinidia diavina (Suffert 1904)
Acraea pullula (Grünberg 1911)
Acraea vinidia ab. ruandae (Grünberg 1911)
Acraea acerata vinidia ab. burigensis (Strand 1913)
Acraea acerata tenella f. alluaudi (Le Cerf 1927)
Acraea acerata hoursti (Le Cerf 1927)
Taxonomic position: Insecta, Lepidoptera, Nymphalidae
Authors: J.S. Okonya, N. Mujica, P. Carhuapoma, & J. Kroschel
Common names
Small yellow-banded Acraea, the Falls Acraea (English)
Hosts
Larvae of Acraea acerata feed mainly on Ipomoea species, with Ipomoea batatus (L.) Lam. being most preferred. Other species affected include I. hochstetteri House, I. tenuirostris Choisy, I. cairica (L.) Sweet, I. obscura (L.) Ker-Gawl., I. lilacina Blume, I. kentrocarpa A. Rich., and I. wightii Choisy. There are records from other genera of the Convolvulaceae such as Lepistimone owariense Hall, and Merremia hederacea (Burm. f.) Hallier f.
Detection and identification
Only larvae of A. acerata cause injuries to the plant foliage. The larvae primarily feed on leaves but can also feed on the soft parts of vines in absence of leaves (Photo 1A, B). Adult sweetpotato butterflies, egg batches, and clusZters of young larvae in a web are usually the first and most obvious signs of the presence of A. acerata. Green third to fifth instar larvae can be seen feeding on sweetpotato leaves and can completely defoliate the plant at high densities. The presence of dark green frass on the leaves or vines indicates the presence of A. acerata larvae. Highly infested crop fields are without leaves; only vines and leaf petioles are left (Photo 1C).

Morphology
Egg
Eggs (0.5 x 0.7 mm) are oval, pale yellow, and laid in bunches on the underside of the leaves (Photo 2A).
Larva
First and second instar larvae are green, turning to pale green in the third to fifth instar. The fifth instar larva measures 24 x 1 mm and has interrupted longitudinal streaks (Photo 2B). Spines of the first two and the last three segments are dark brown, the other is pale yellow. The head is dark brown with a black inverted V line in the center, with a few brownish dorsal and dorsolateral marks on each segment. Lateral line on the body is pale yellow.
Pupa
Pupa is pale with dark brown markings on the back and varies in size (12–15 cm long, 0.7–1.1 mm wide) (Photo 2C).
Adult
Adult butterflies have orange wings with brown margins; wingspan is 30–40 mm (Photo 2D, E).
Photo 2. The developmental stages of the sweetpotato butterfly, Acraea acerata: (A) eggs, (B) larva, (C) pupae, (D) adult male and female, and (E) adult male with open wings. Photos: Courtesy of CIP.
Biology
Adult females lay their eggs mostly on the underside of sweetpotato leaves; few eggs are laid on the upper side of leaves or on stems. Newly hatched larvae stay in a group and feed together, usually on a single leaf in protective webbing. Third instar larvae become solitary and feed on the whole leaf. Older larvae can be seen at the base of the plant or in dry leaves escaping from the day’s heat. Pupation is on plant parts. Male butterflies emerge 1 or 2 days before the females; females emerge with fully grown eggs. After mating, eggs are laid in batches of 70–500, at intervals of 1–4 days.
Temperature-dependent development
Development of all A. acerata live stages is possible at 17.5°–30°C. The upper threshold of development is 32°C for eggs and 30°C for larvae and pupae. Adult males live longer than females. Optimal temperatures for both males and females range 17.5°–25°C. Egg mortality is quite low (<10%) at all temperatures where development was possible. Larvae mortality is highest (42%) at 17.5°C and lowest (4%) at 20°C. All pupae died at extremely low (17.5°C) and extremely high (32°C) temperatures. Oviposition is highest at 30°C, with 131 eggs laid, and drops with decreasing temperature. The lowest oviposition of 4.5 eggs per female was registered at 17.5°C.
Life-table parameters—intrinsic rate of increase (rm), finite rate of increase (λ), doubling time (Dt), mean generation time (T), gross reproduction rate (GRR), and net reproduction rate (Ro)—were estimated through deterministic phenology modelling (see Annex 7.3.6). The population of A. acerata increased exponentially: it was highest at 30°C (rm=0.08) and decreased drastically above 30°C. The highest values for Ro (10) and GRR (40) were recorded at temperatures of 20°–30°C. This range of temperatures is therefore the most suited for high reproduction rates of A. acerata; Ro>1 indicates an increase in females over the previous generation. Generation time was longest at 17.5°C (60 days) and shortest at about 30°C (25 days). Temperatures below 20°C and above 30°C had the longest doubling time (100–150 days) for A. acerata population, whereas temperatures of 20°–30°C had the shortest Dt (about 12 days). The rate of population growth for A. acerata was highest (λ=1.08) at about 27°C.
Means of movement and dispersal
Although adults are weak flyers, they can migrate over long distances. Eggs or first–second instar larvae can be spread by humans via transporting infested planting material (vines).
Economic impact
A. acerata is a serious pest of sweetpotato in sub-Saharan Africa (SSA). Defoliation caused by larval feeding reduces photosynthesis activity. Complete defoliation can result in failure of the plant to establish if outbreaks occur early in the season or reduced root yields when outbreaks occur late in the season. There is little or no published information on the yield losses of A. acerata.
Geographical distribution
A. acerata occurs throughout tropical Africa (Fig. 1).
| Africa | Angola, Benin, Botswana (north), Burundi, Cameroon, DR Congo, Ethiopia (South), Ghana, Guinea, Ivory Coast, Kenya, Liberia, Madagascar, Mozambique, Namibia (north), Nigeria, Rwanda, Sierra Leone, Sudan, Tanzania, Togo, Uganda, Zambia, Zimbabwe (northwest) |
Phytosanitary risks
A. acerata is primarily a wet tropical species that has been found at altitudes of up to 2,261 masl in Uganda. It is a major pest of sweetpotato in Ethiopia and Rwanda but a minor pest in Uganda and Kenya.
Risks mapping under current and future climates
Global Risks
Changes in establishment and future distribution
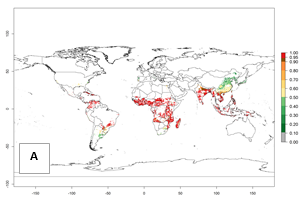
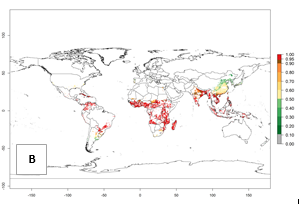
The establishment risk index (ERI) of 0.95–1 reflects well the current global distribution of A. acerata under the current climate of the year 2000 (Fig. 2A); those regions include SSA countries (compare with Fig. 1). Therefore, regions with an ERI>0.95 are considered to be at high risk of A. acerata establishment. Those regions include Central America (El Salvador, Honduras, Panama); South America (Colombia, Venezuela, Southern Brazil, Paraguay); the Caribbean (Cuba, Haiti, Dominican Republic, Puerto Rico); Oceania; Southern Asia (Southern India and Bangladesh); and most countries of Southeast Asia. In zones where the ERI drops below the maximum number of 1, the likelihood of long-term establishment is reduced, as in Southern Africa.
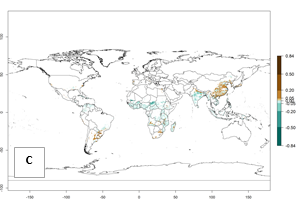
Global predictions for 2050 indicate that A. acerata will remain a high risk pest (ERI>0.9) for most tropical countries (Fig. 2B, C). A slight increase in the spread of A. acerata but with a low establishment potential (ERI<0.6) is predicted for subtropical regions of Asia (southern China); South America (northern Argentina, Uruguay, southern Brazil); and Southern Africa (Angola, South Africa, Zambia, Malawi).
 |
 |
 |
|
Figure 2. Changes in establishment and potential distribution of the sweetpotato butterfly, Acraea acerata, in sweetpotato production regions worldwide according to model predictions, using the ERI for the years 2000 (A) and 2050 (B), and changes of the ERI between 2000 and 2050 (C). An ERI>0.95 is associated with potential permanent establishment.
Changes in abundance
The number of generations predicted in SSA for the sweetpotato-growing areas with existing A. acerata infestation ranges mainly 6–10 (Fig. 3A), although in some parts of West Africa up to 12 generations might develop. There are
few reports on the number of generations that develop at specific geographies for validation of the simulations. Field studies, for instance in Welayita, Southern Ethiopia, reported on average 6 generations per year between 1994 and 1997. The simulated generation index (GI) values of 4–8 in this country are in the range of those reported in the literature. In the other tropical zones (Southern and Southeast Asia, the Caribbean, and northern South America), 8–12 generations per year are predicted. The GI change estimates a potential increase of 1–2 generations per year for most subtropical and tropical regions (Fig. 3B, C).
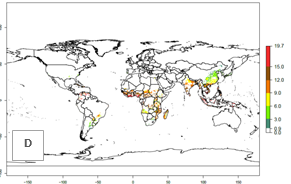
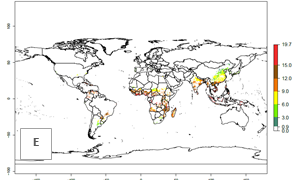
Global maps of the activity index (AI) in year 2000 estimate a high activity of A. acerata by a factor of 12–19 in most tropical regions, and by a factor of 6–12 for most subtropical regions of South America, East and Central Africa, and Southeast Asia (Fig. 3D). Predictions of changes for the 2050 temperature scenario show a decrease in the potential growth of A. acerata in tropical regions of Central and South America, Africa (western region), and Asia (Fig. 3E, F). In most of subtropical regions the AI increases by values up to 5.
| GI | AI | |
| 2000 | 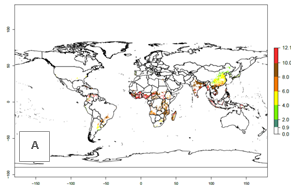 |
 |
| 2050 | 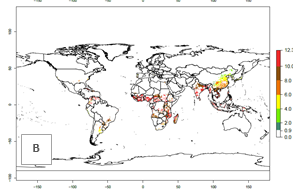 |
 |
| Index change (2000 – 2050) | 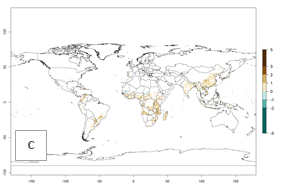 |
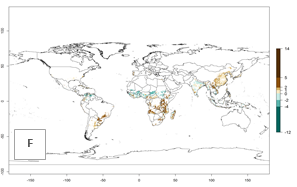 |
Figure 3. Changes in abundance (GI, damage potential) and activity (AI, potential population growth) of the sweetpotato butterfly, Acraea acerata, in sweetpotato production regions worldwide according to model predictions, using the GI (A, B) and the AI (D, E) for the years 2000 and 2050, and the absolute index change (C, F).
Regional Risks for Africa
Changes in establishment and future distribution
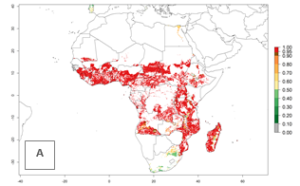
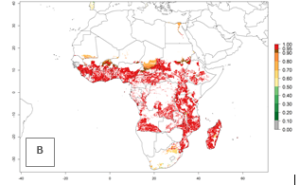
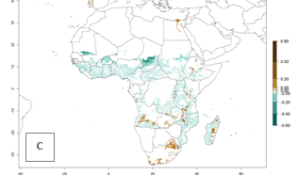
A. acerata is already present in most countries of East, West, and Central Africa, with an ERI>0.95, indicating a high risk in the sweetpotato production areas (Fig. 4A). However, A. acerata has not been reported to occur in some of the countries with an ERI>0.95 due to factors other than temperature that also influence establishment and distribution of A. acerata. Heavy rains and very dry conditions have, for instance, been observed to decrease the survival of young A. acerata larvae and pupae. This could explain why A. acerata is not permanently established in areas with favorable temperature conditions. Under the year 2050 temperature scenario, A. acerata will remain a high risk pest (ERI>0.95) for nearly all sweetpotato-growing countries in Africa (Fig. 4B, C). A slight increase in the spread of A. acerata is predicted for a few areas in Southern Africa (Angola, South Africa, Zambia, Malawi).
 |
 |
 |
|
Figure 4. Changes in establishment and potential distribution of the sweetpotato butterfly, Acraea acerata, in African sweetpotato production regions according to model predictions, using the ERI for the years 2000 (A) and 2050 (B), and changes of the ERI between 2000 and 2050 (C). An ERI>0.95 is associated with potential permanent establishment.
Changes in abundance
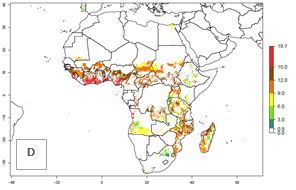
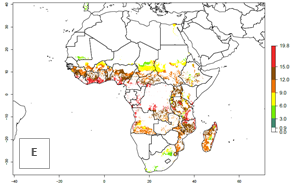
Under current climate of the year 2000, the number of generations is estimated at 6–12 in most of the sweetpotato production areas in Africa (Fig. 5A). The highest number of generations per year (10–12) is predicted for West Africa. Change of A. acerata GI between 2000 and 2050 shows that number of A. acerata generations per year will increase more rapidly in East and Southern Africa (1–2 more generations per year) than in West Africa, with a predicted increase of 1 generation per year (Fig. 5B, C). African countries with the largest sweetpotato production areas supporting up to 12 generations per year in 2050 scenario include Guinea, Sierra Leone, Ivory Coast, Togo, Ghana, Benin, Nigeria, Central African Republic, Sudan, Mozambique, Tanzania, and Madagascar.
The highest activity (AI>15) of A. acerata is estimated for West Africa for the year 2000, mainly for the coastal areas of Sierra Leone, Ivory Coast, Ghana, Togo, Benin, and Nigeria (Fig. 5D). For the year 2050, the potential A. acerata population increase will be relatively high (AI>12) in most of the sweetpotato-growing countries. The AI change between the years 2000 and 2050 indicates a higher population and spread potential for most of Central, Southern, and East Africa. An insignificant decrease in the spread potential is predicted for most of the countries in West Africa (Fig. 5E, F).
| GI | AI | |
| 2000 | 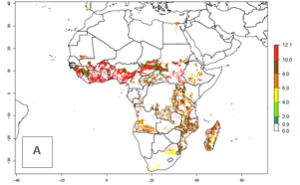 |
 |
| 2050 | 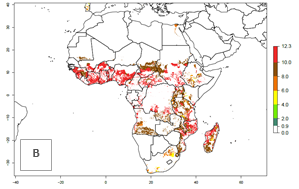 |
 |
| Index change (2000 – 2050) | 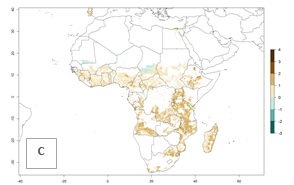 |
 |
Figure 5. Changes in abundance (GI, damage potential) and activity (AI, potential population growth) of the sweetpotato butterfly, Acraea acerata, in African sweetpotato production regions according to model predictions, using the GI (A, B) and the AI (D, E) for the years 2000 and 2050, and the absolute index change (C, F).
Country Risk Maps
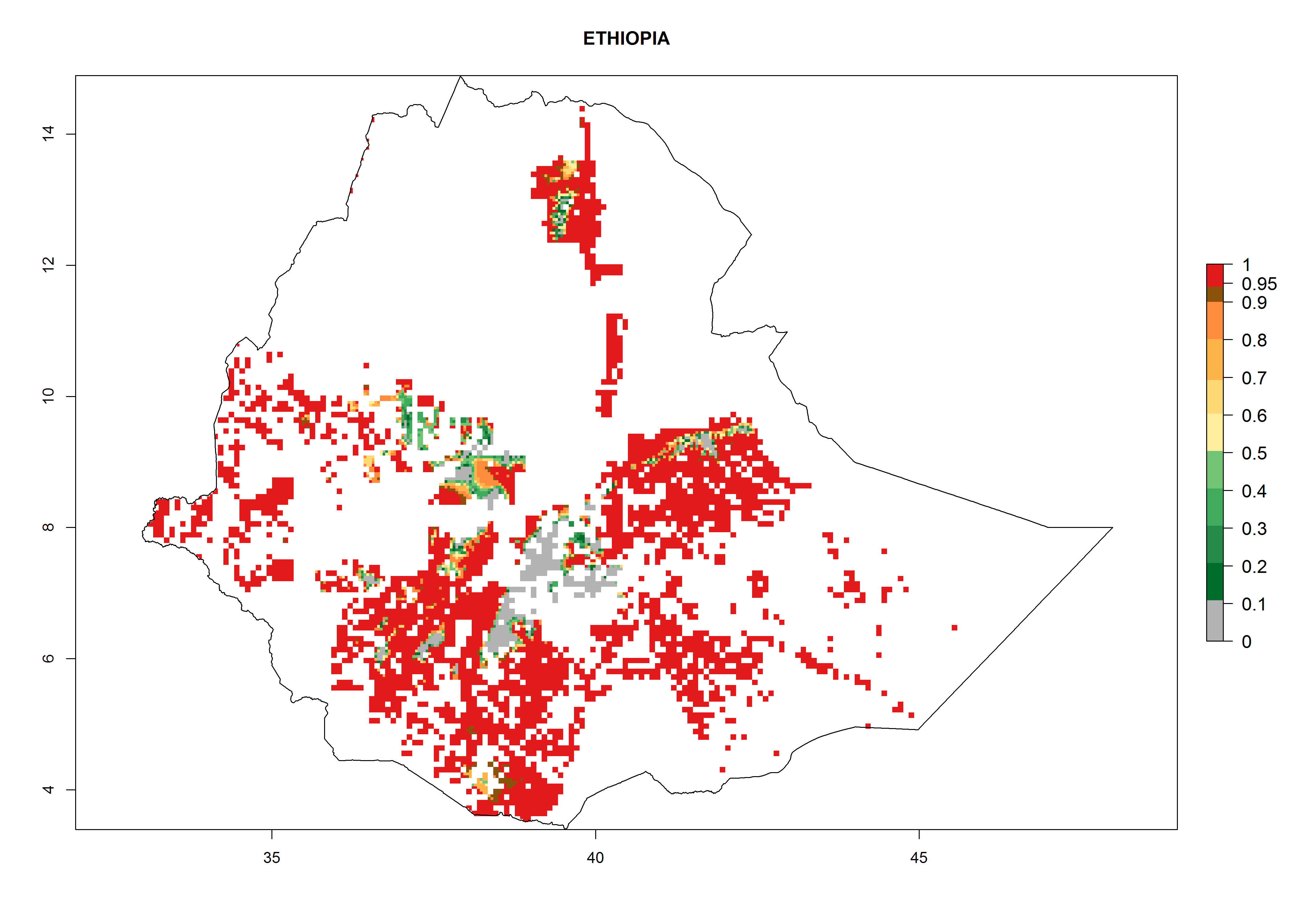
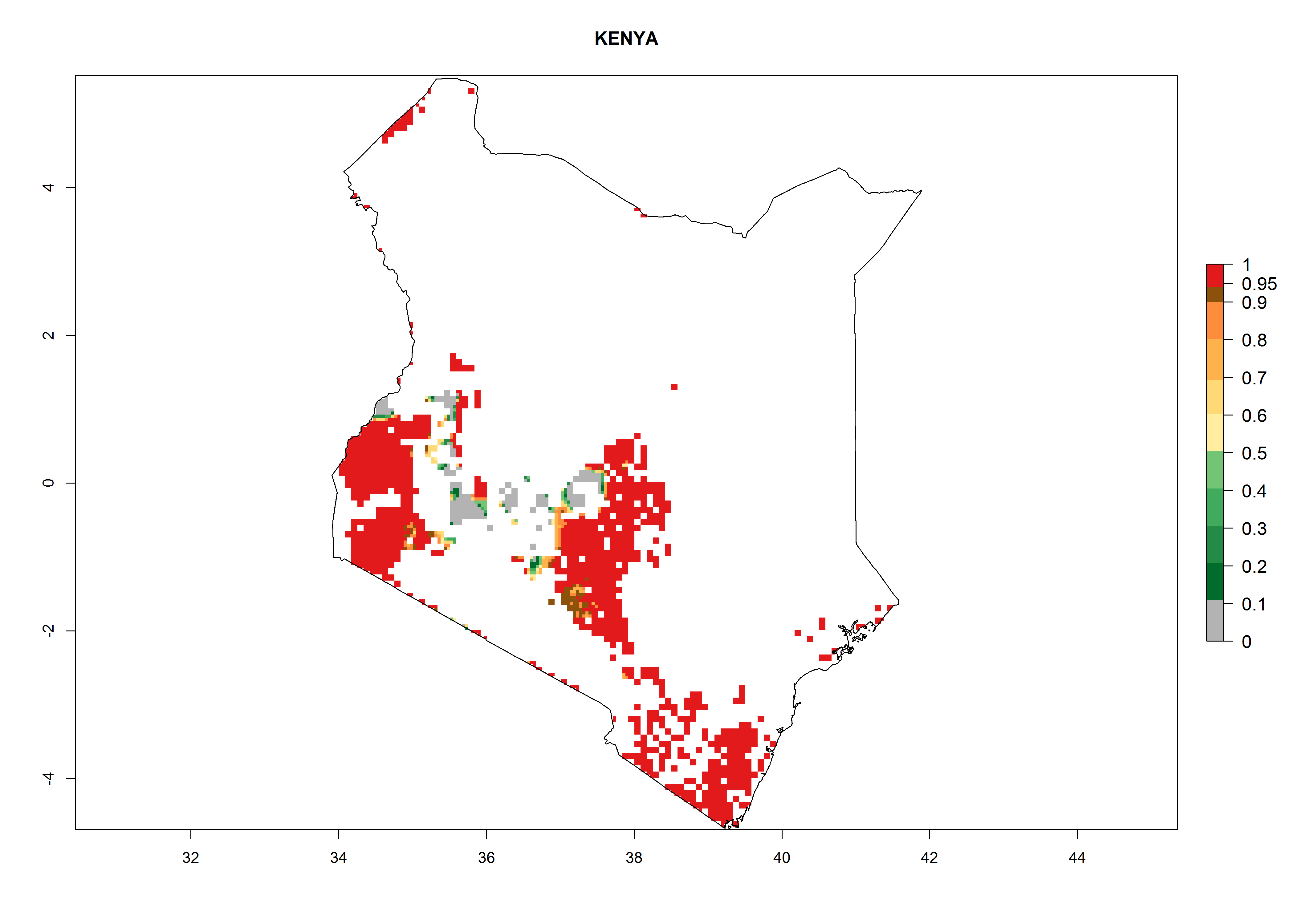
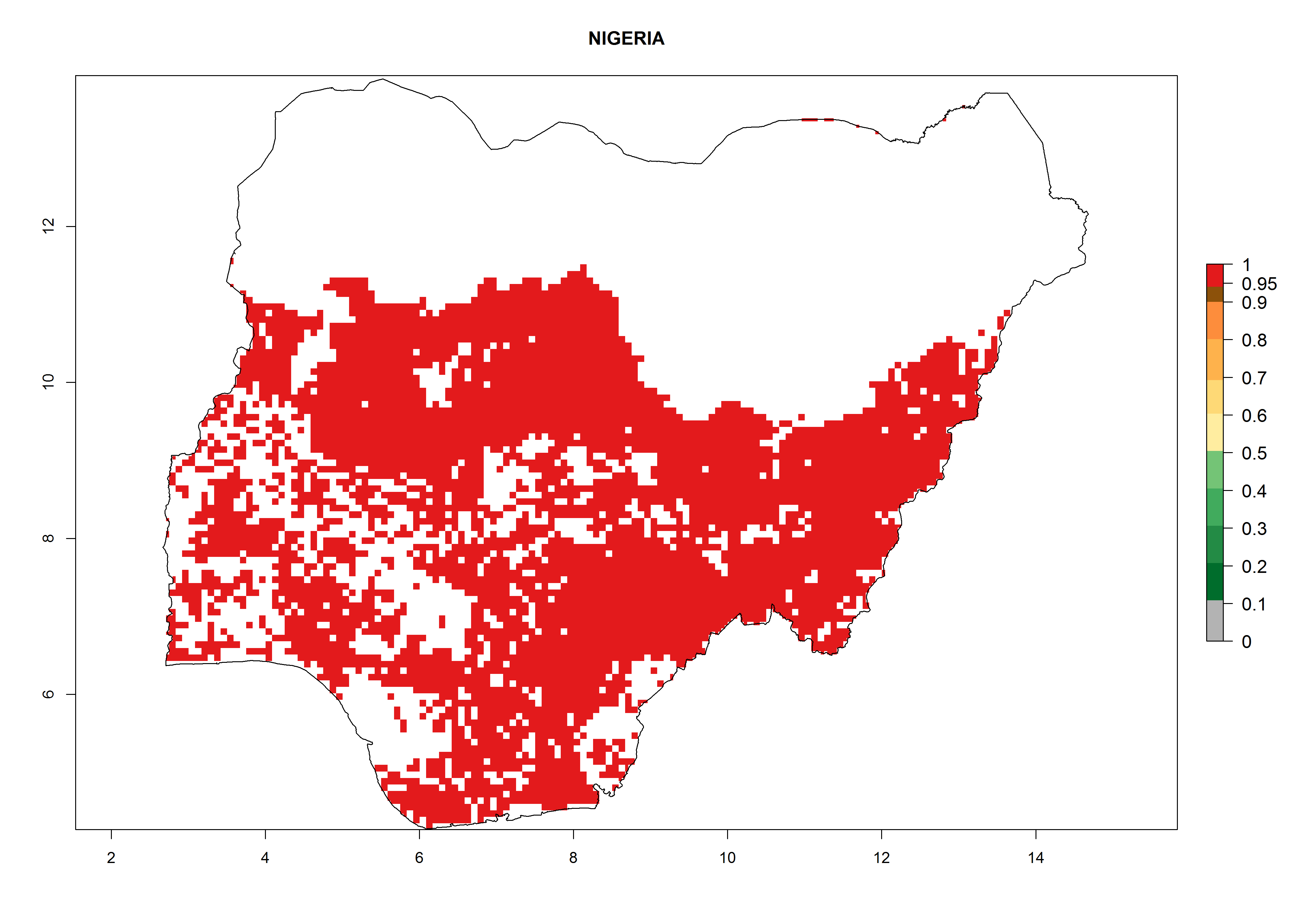
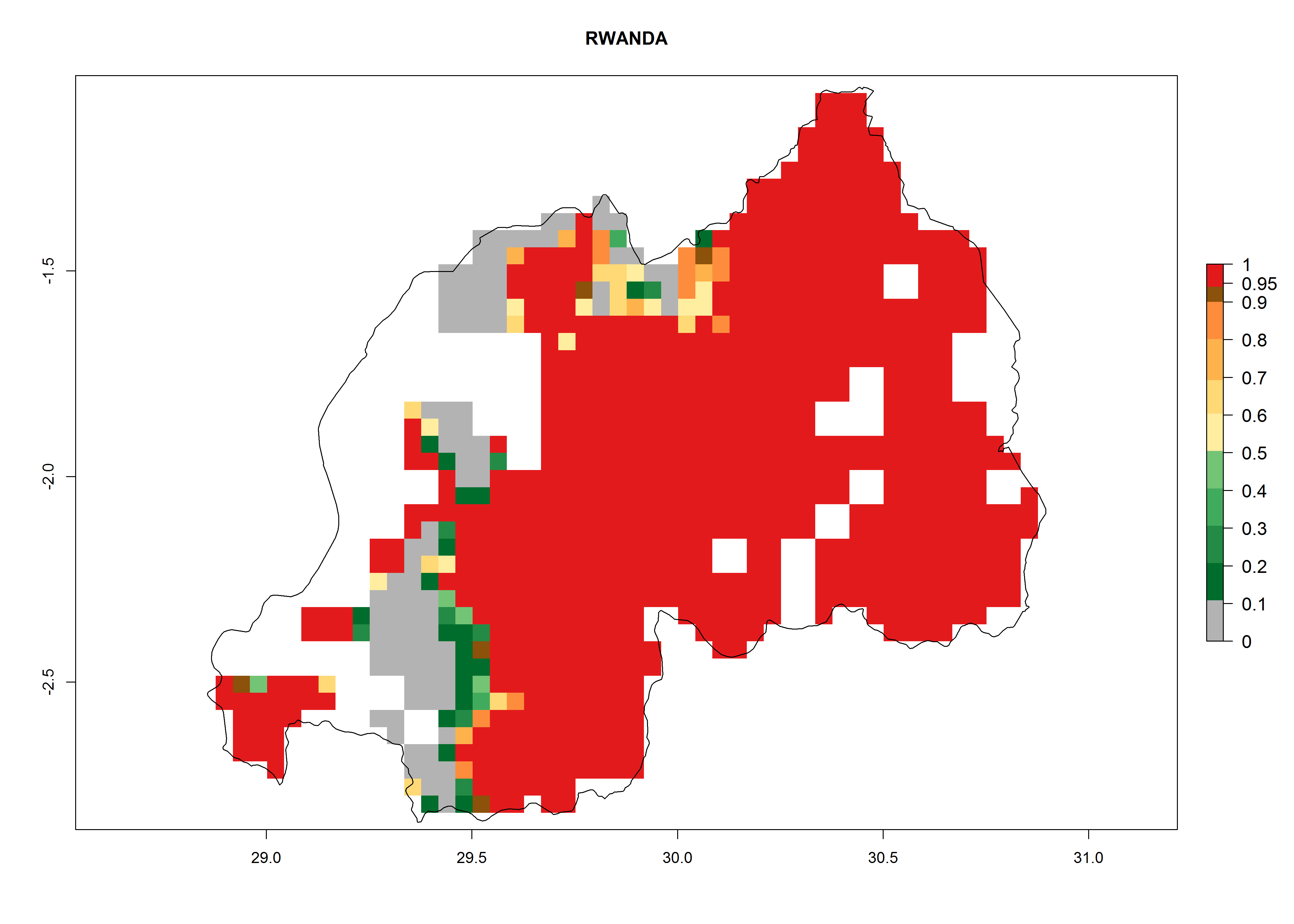
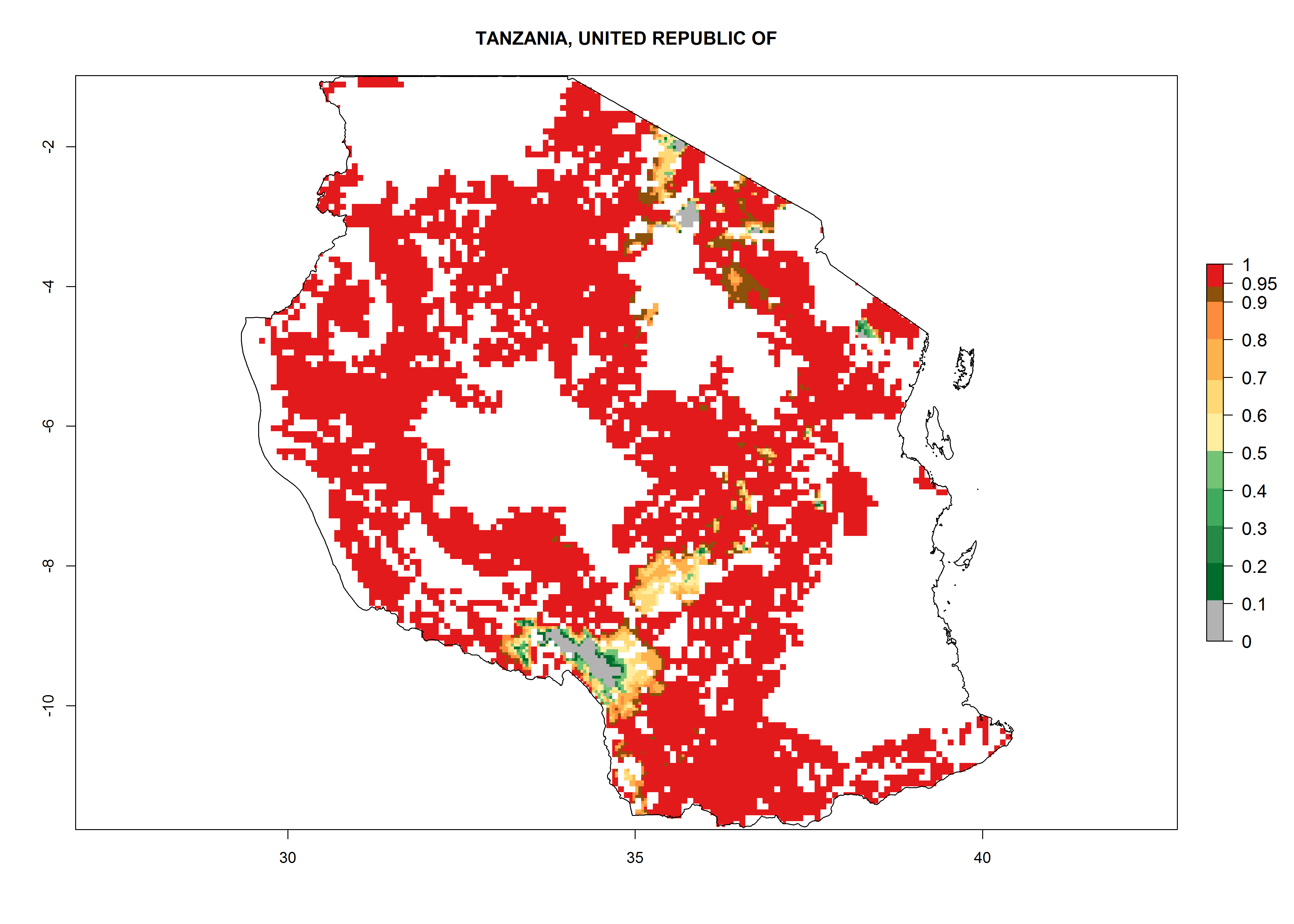
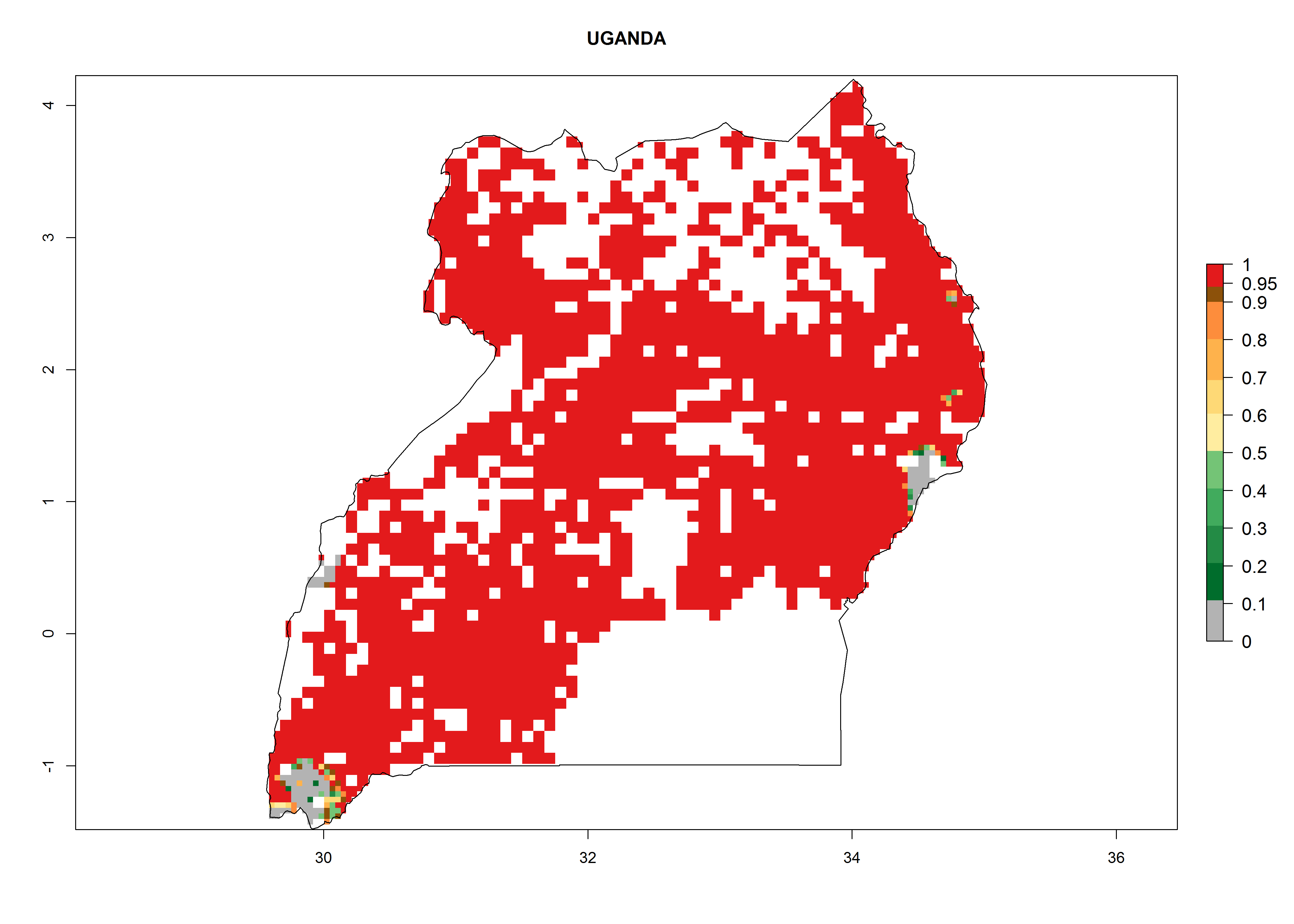
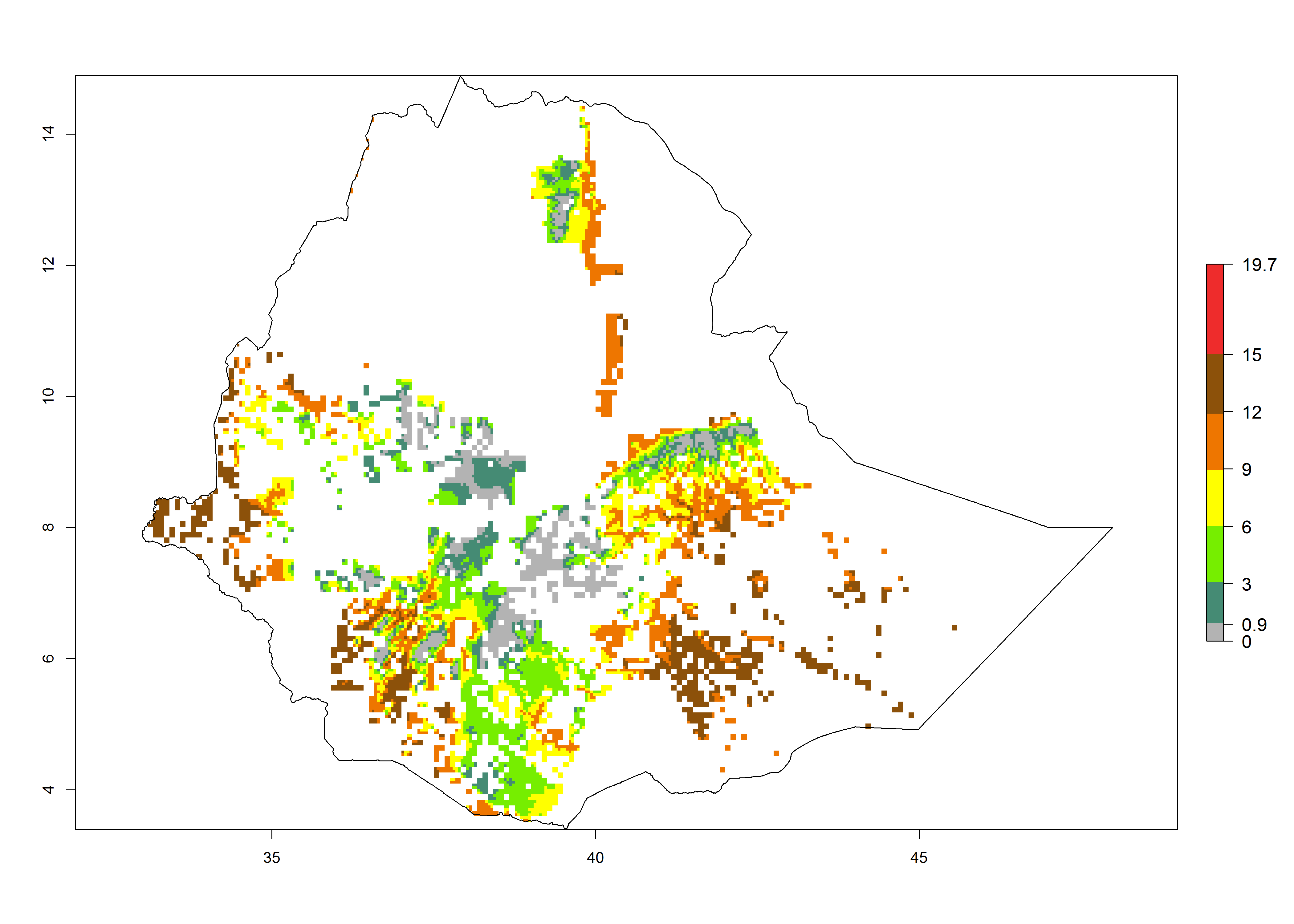
Current occurrences can be observed at ERI>0.95. The highest risks of establishment (ERI>0.95, red zones) are observed in the whole of Uganda, Nigeria (except in the north), Rwanda (except in a few areas in the west), and Tanzania (except the coastal area) in 2000 (Fig. 6). In terms of area at risk of invasion in 2050, Uganda, Rwanda, Tanzania, and Nigeria all have a very large area with an ERI>0.95 (Fig. 6). Overall, we note that A. acerata represents a severe risk in most of the major sweetpotato-producing countries in East and West Africa (Fig. 6). Up to 12 generations per year are predicted for most of the sweetpotato production areas, with Nigeria having the largest area that could experience the highest number of generations, followed by Uganda and Tanzania. Southern Ethiopia, coastal Kenya, southern Nigeria, West Nile, and Teso sub-regions of Uganda and Morogoro in Tanzania will also see a high number of generations per year and activity for this pest (Fig. 6).
| ERI | GI | AI |
| a) Ethiopia
|
 |
 |
| b) Kenya | 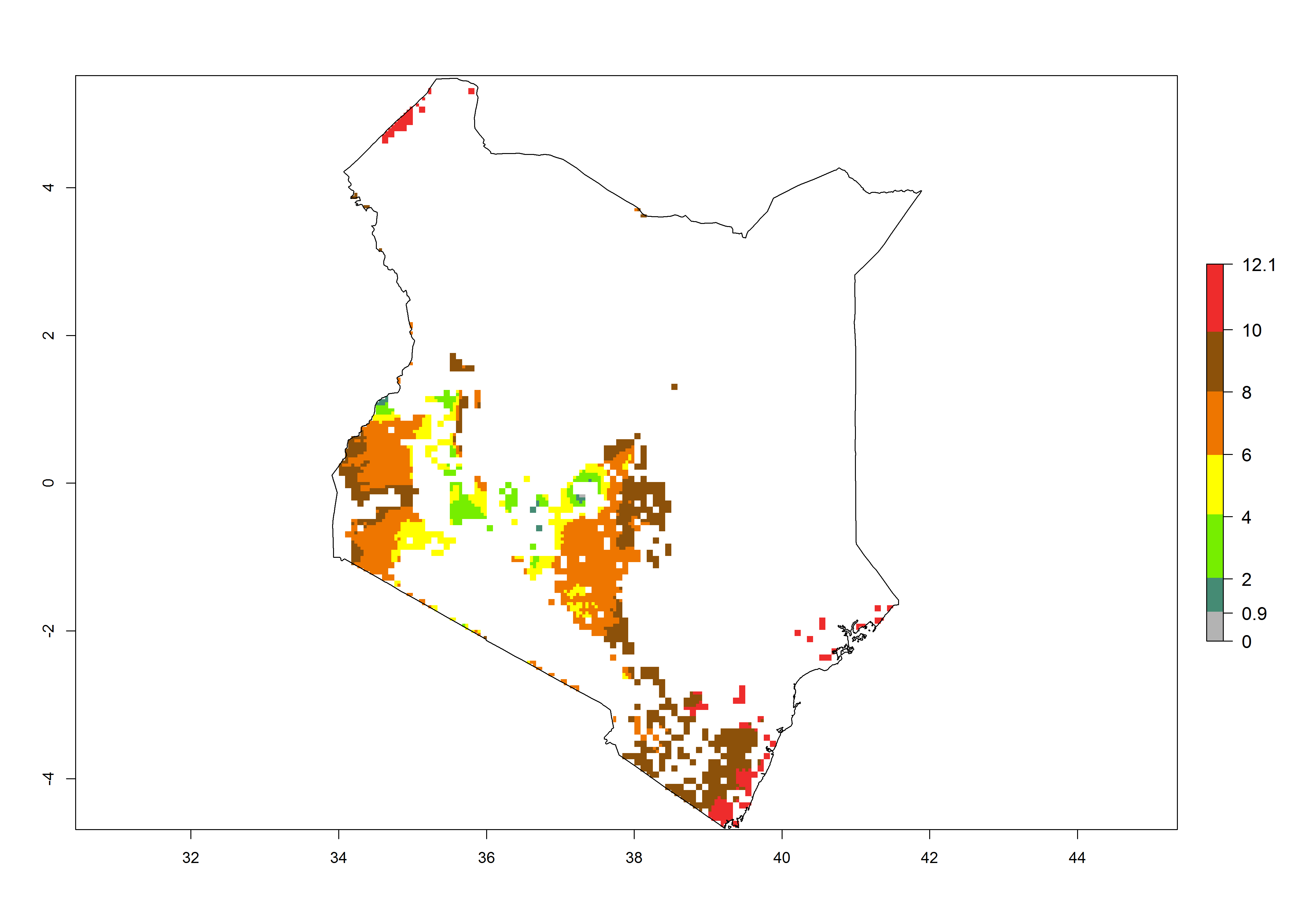 |
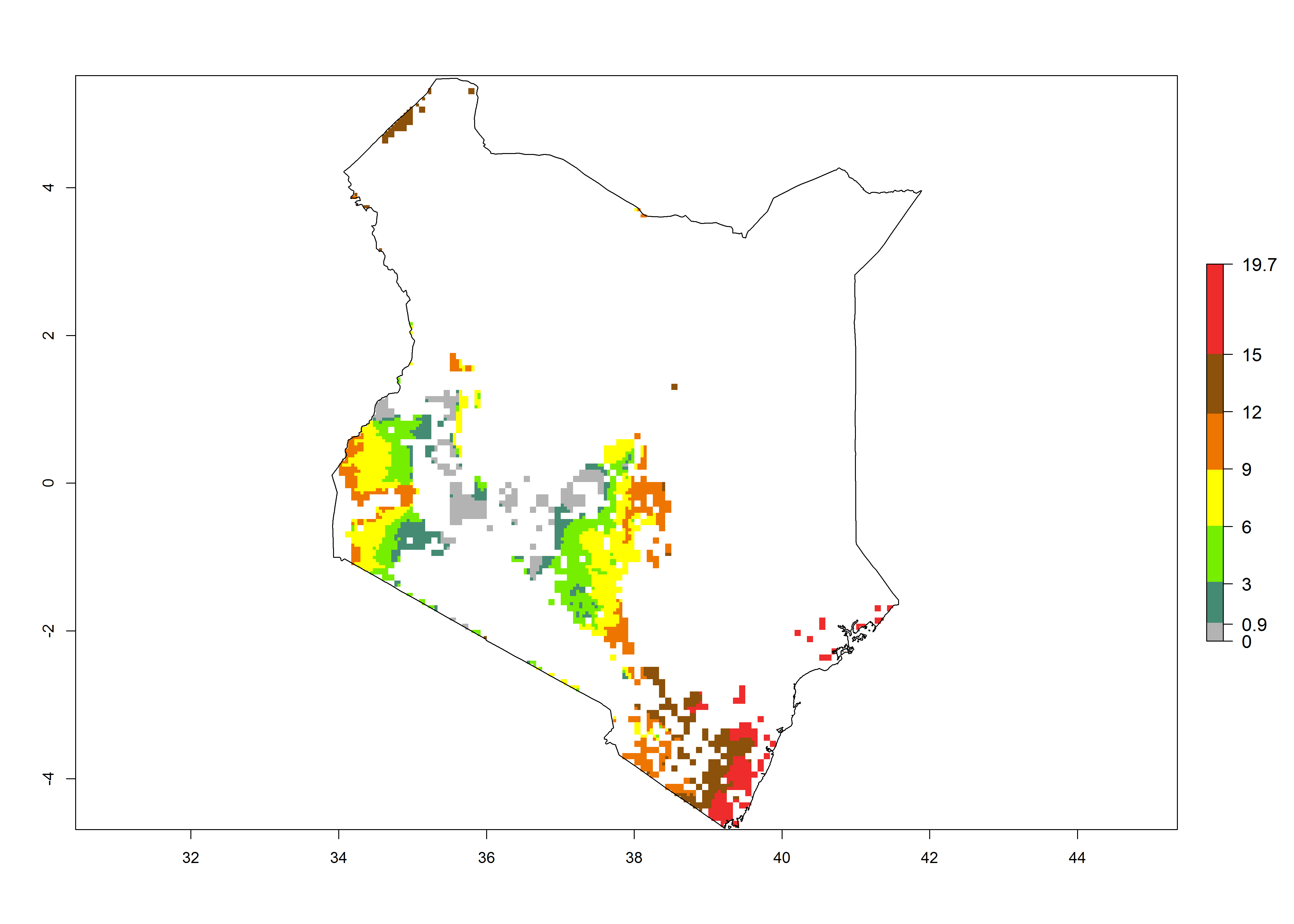 |
| c) Nigeria | 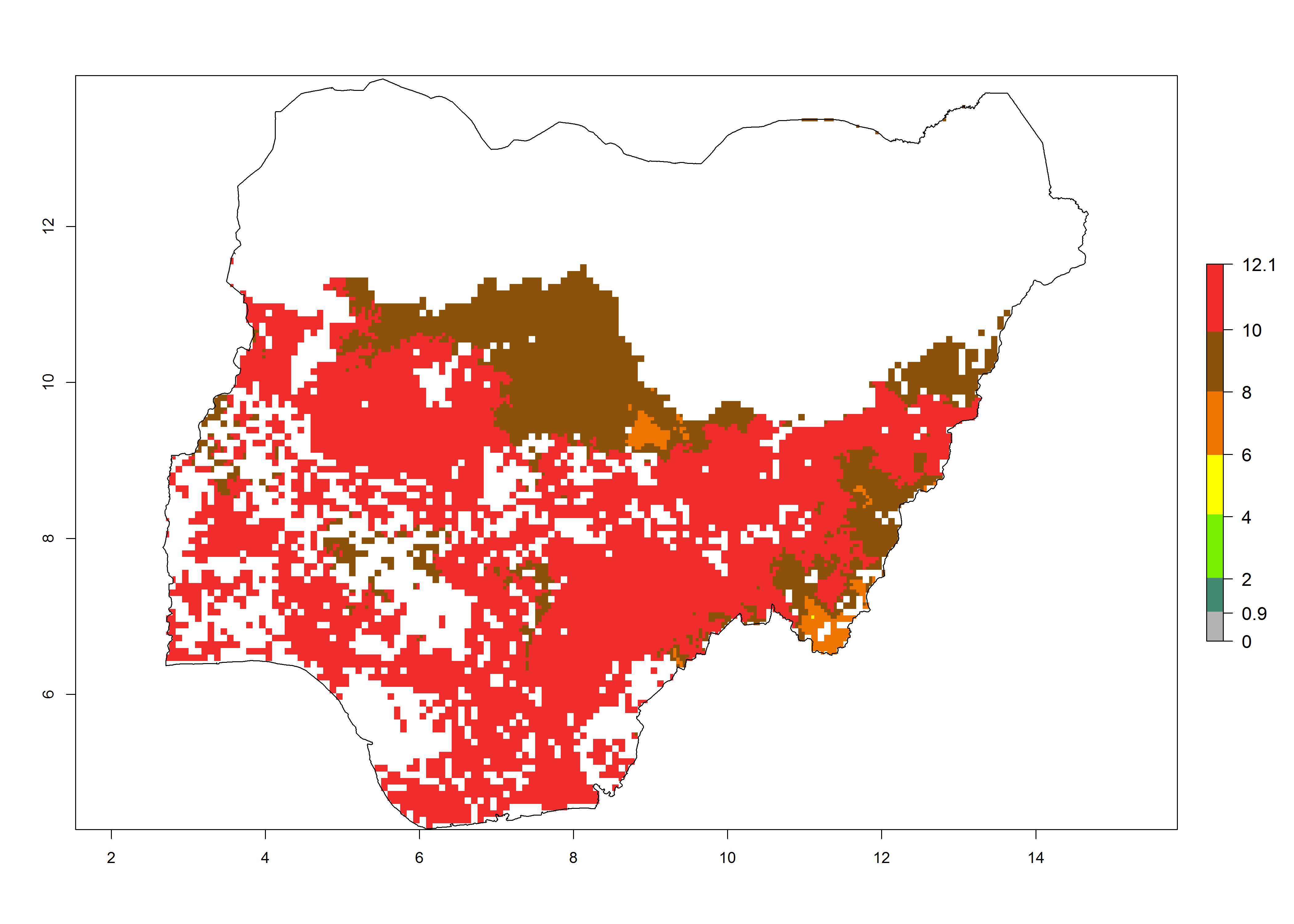 |
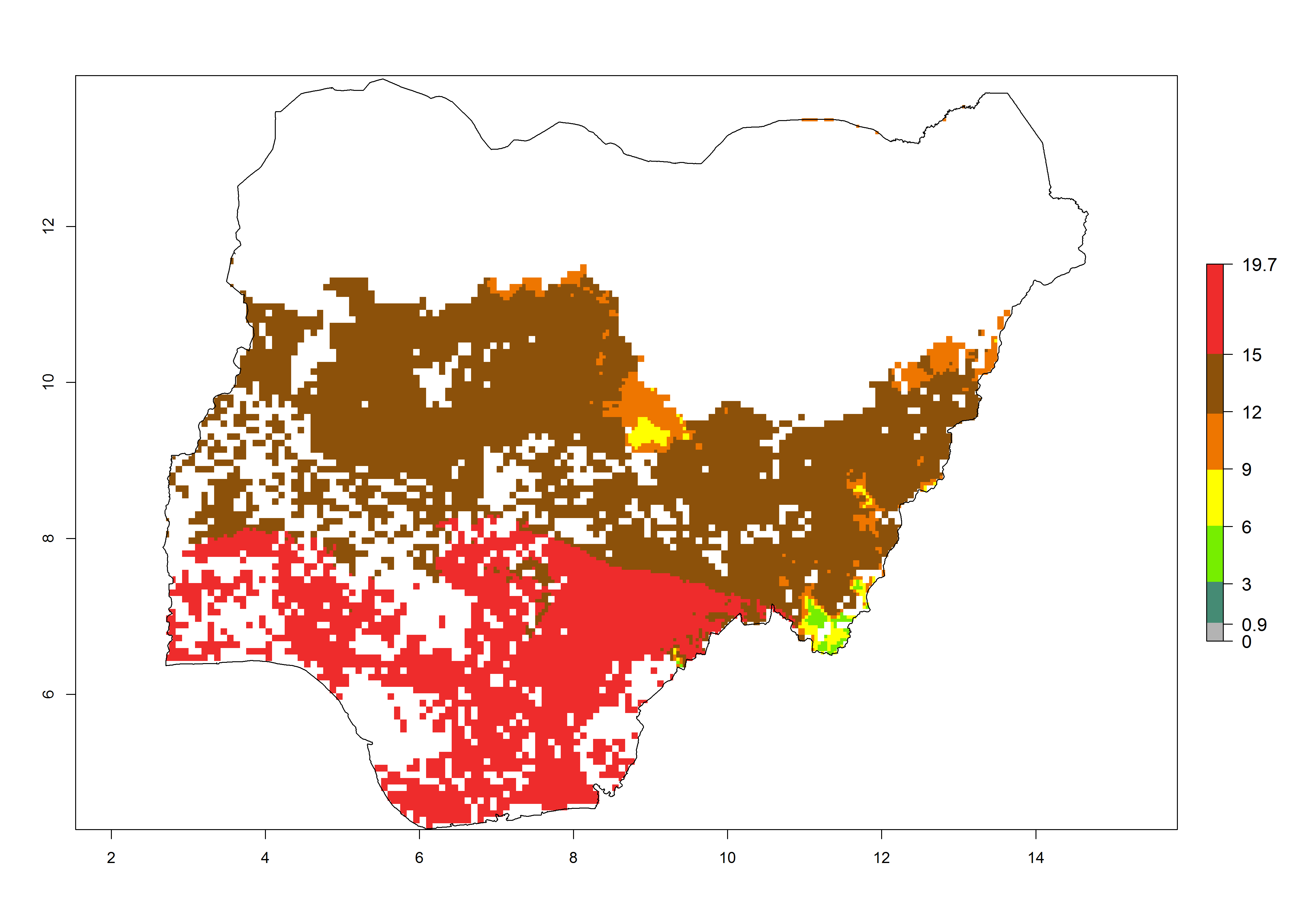 |
| d) Rwanda | 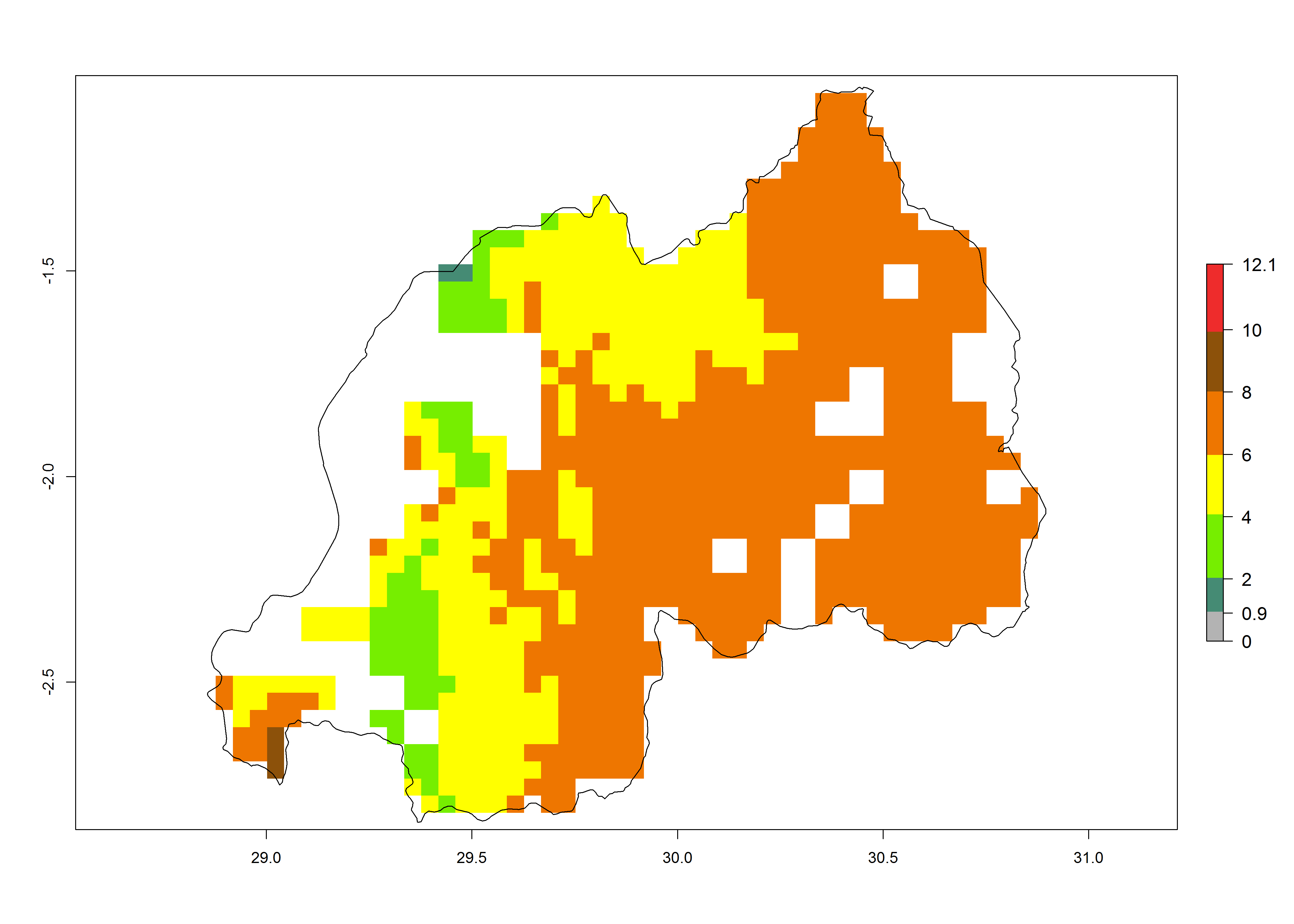 |
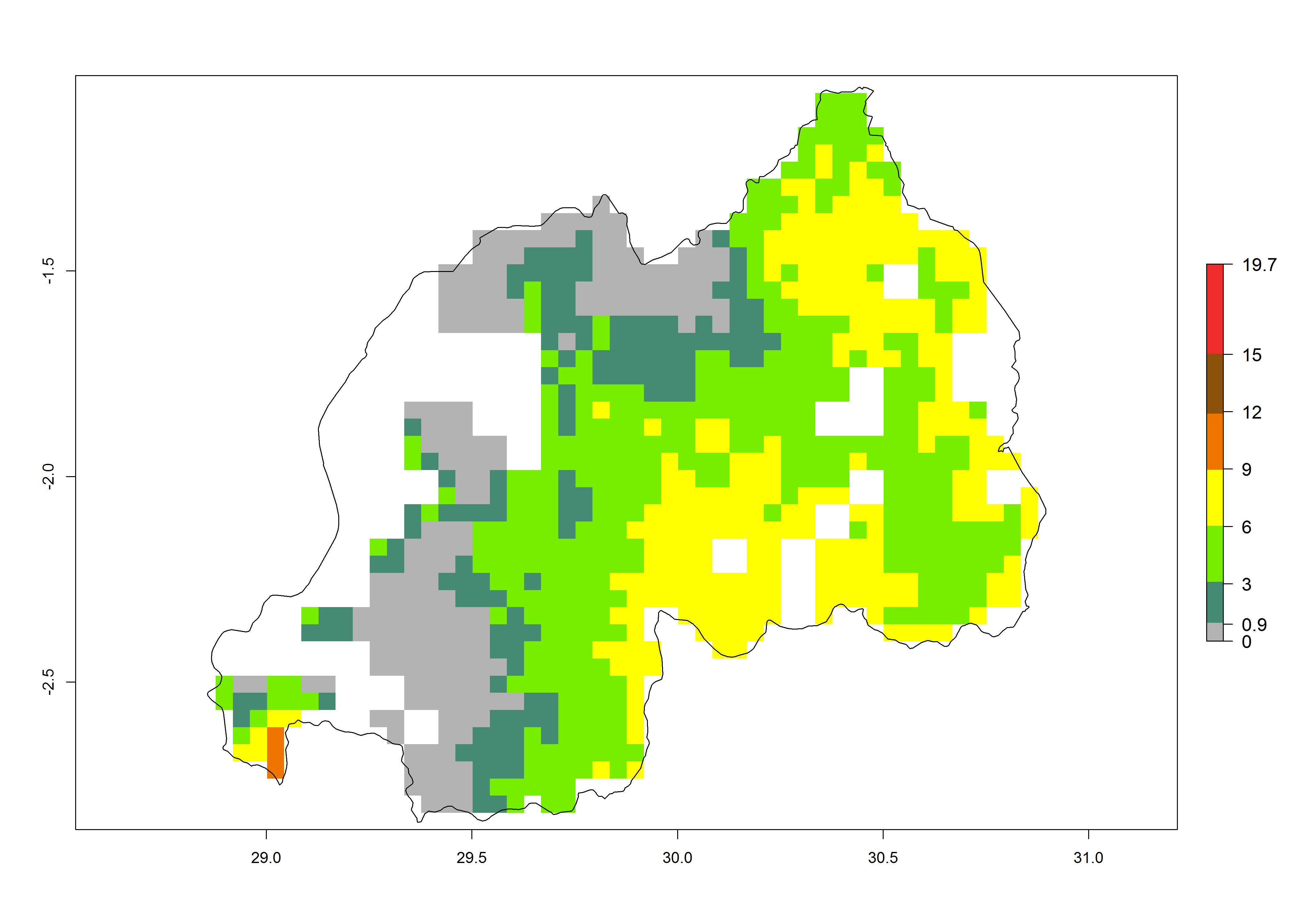 |
| e) Tanzania | 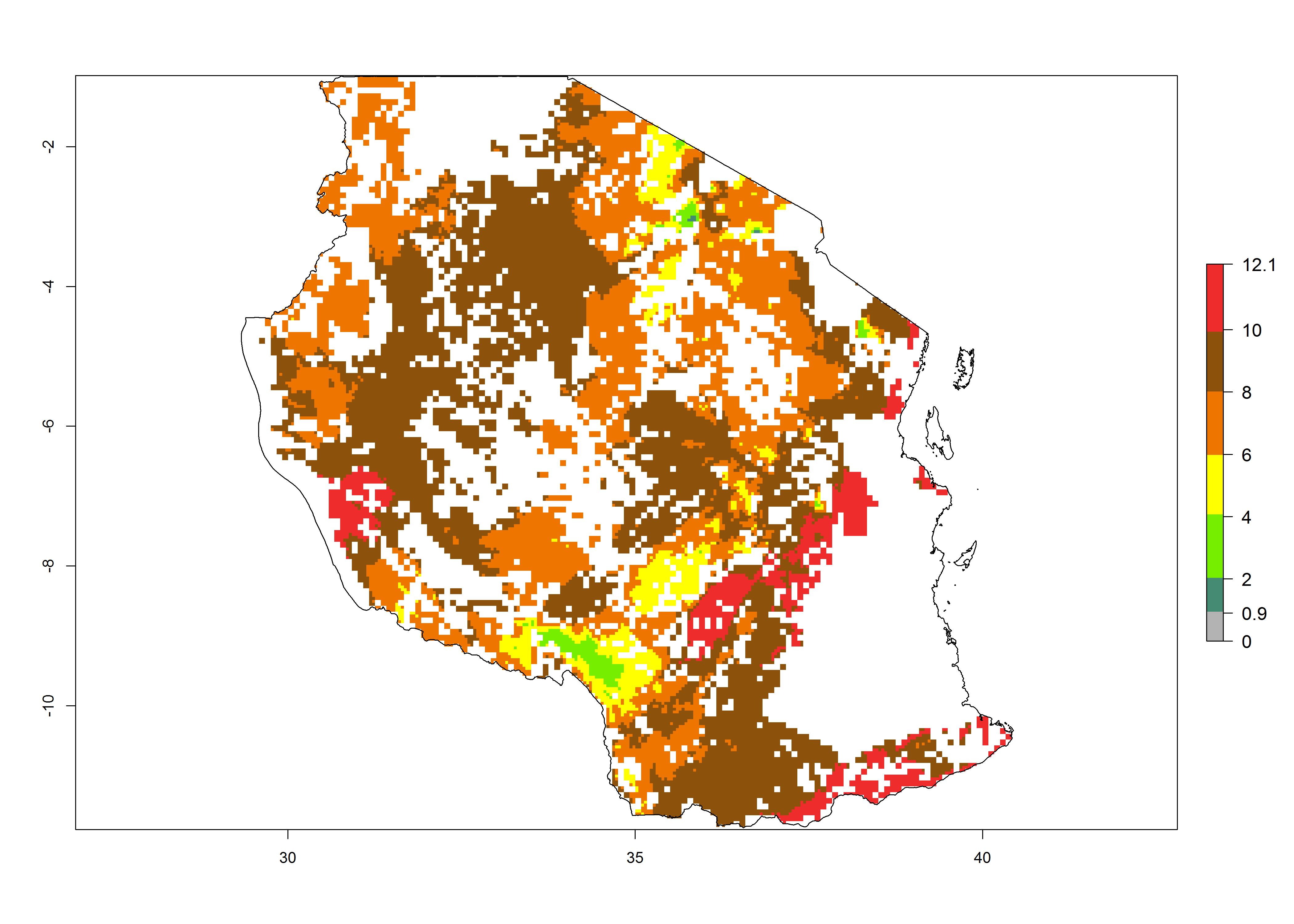 |
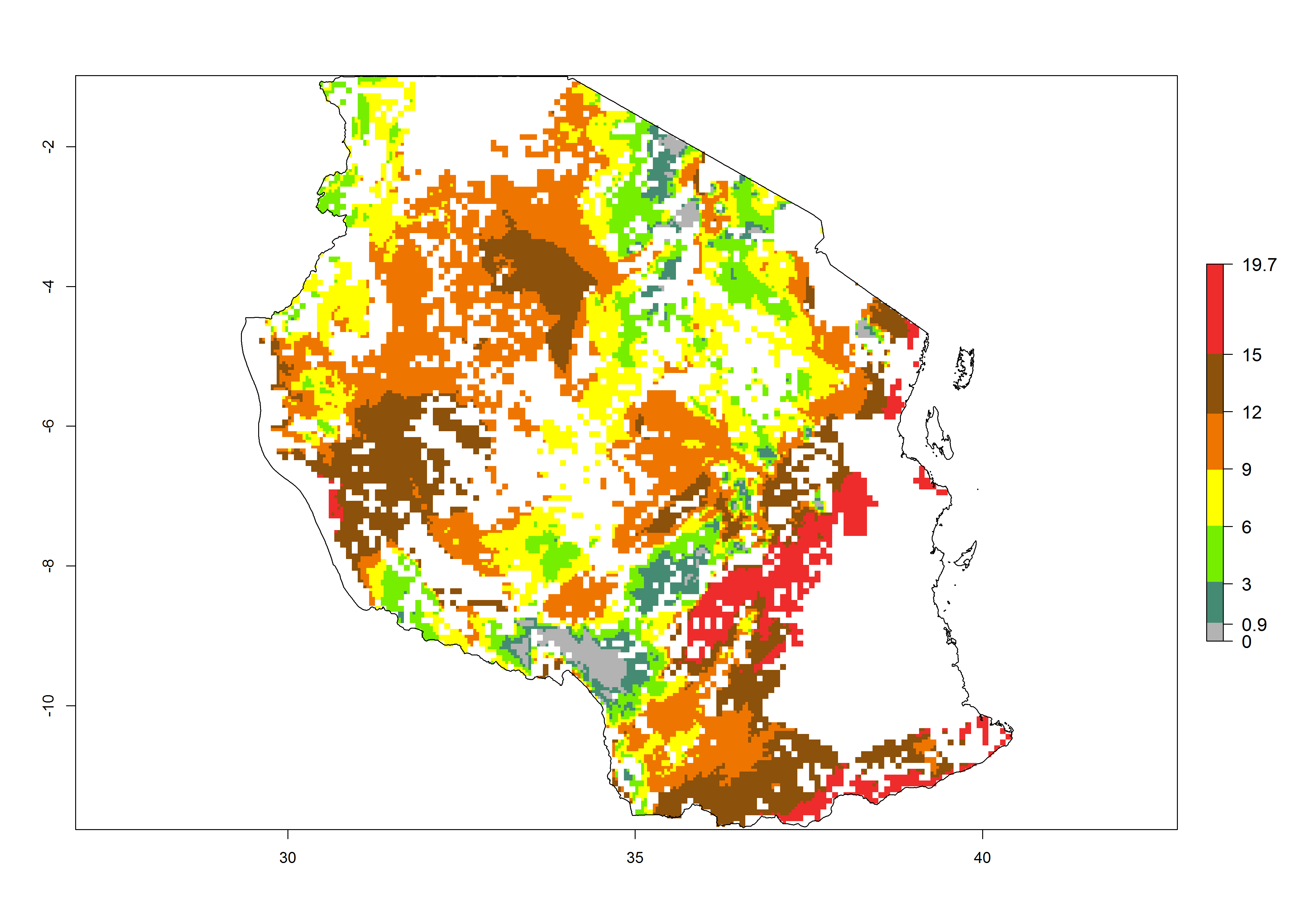 |
| f) Uganda | 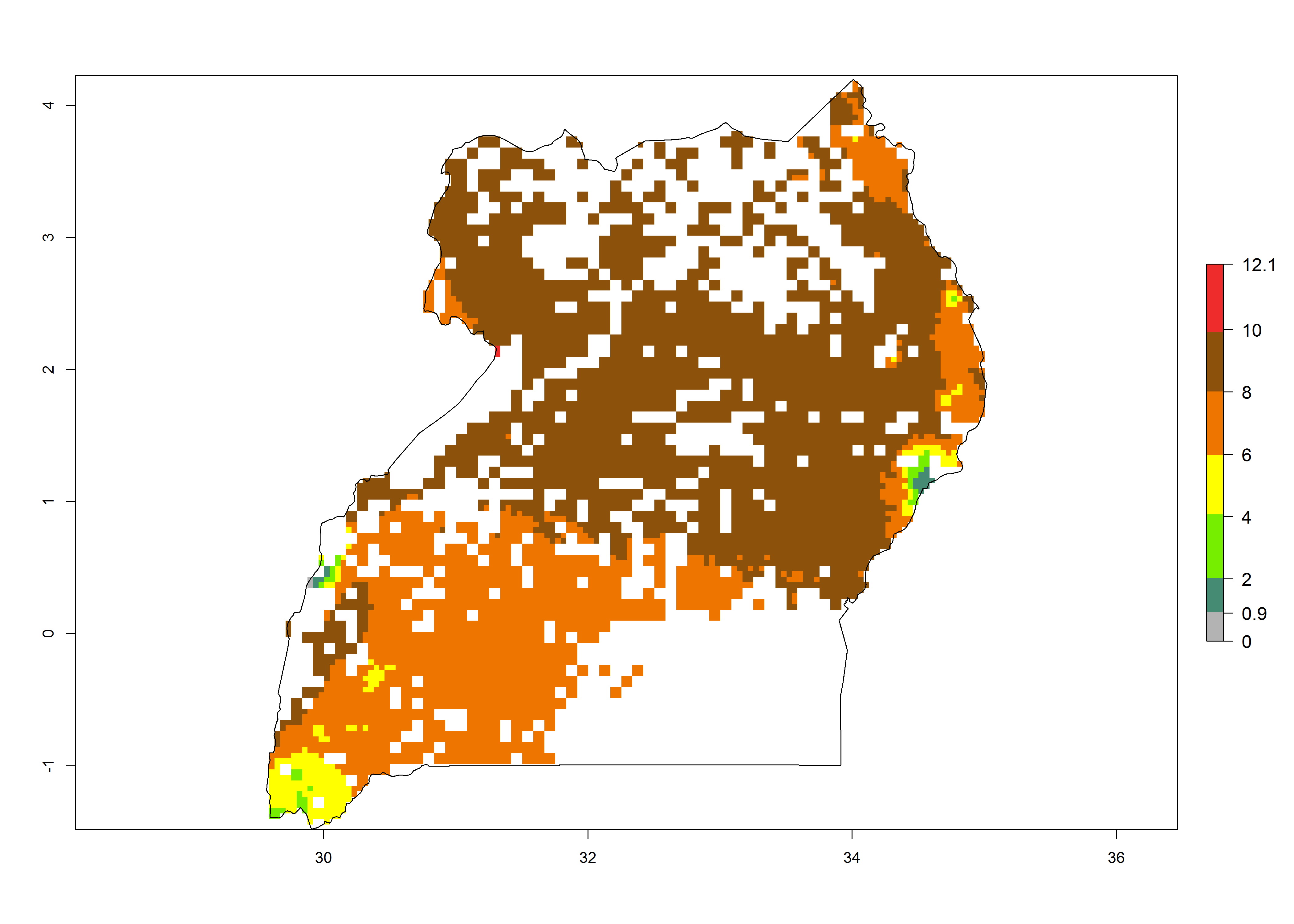 |
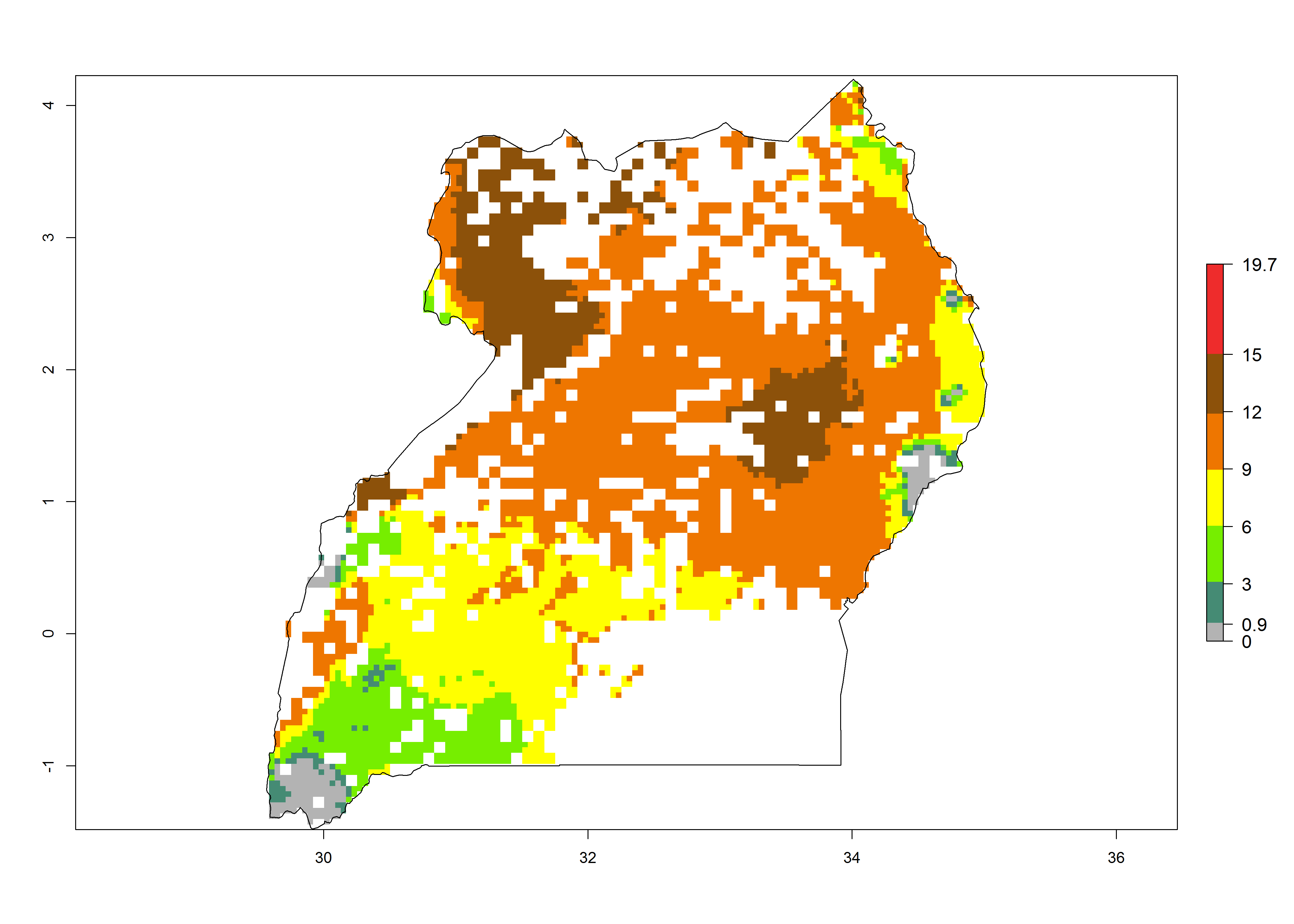 |
Figure 6. ERI, abundance (GI, potential damage), and activity (AI, potential population growth) of the sweetpotato butterfly, Acraea acerata, in sweetpotato production regions of selected African countries according to model predictions for the year 2000. An ERI>0.95 is associated with potential permanent establishment.
Phytosanitary measures
All pest developmental stages can be destroyed mechanically or by application of insecticide. To avoid the introduction of A. acerata into new areas via planting material, vines should be carefully controlled for egg batches and larvae or defoliated before transportation.
Adaptation to risk avoidance at farm level
Integrated pest management has been shown to be the most ecologically sound pest management strategy, including that for managing A. acerata.
Monitoring pest populations. To avoid routine insecticide applications, regular monitoring of pest populations through the occurrence of A. acerata larvae on the foliage would help to determine which control method to use. Although a single, artificially simulated defoliation of less than 70% has been shown not to significantly reduce sweetpotato root yields in experiments, farmers reported that even a one-time defoliation of <50% by A. acerata larvae reduced sweetpotato root yield in their fields. So, it could be possible that feeding by A. acerata larvae not only defoliates the plant, but also induces physiological reactions in the plant that impair storage root formation or growth.
Biological control. No parasitoids were recovered from eggs. Larvae have been observed to be parasitized by the hymenopterans Apanteles acraea Wlkn, Zenillia vara Cuarran, Charops sp. and Meteorus sp., and the dipteran Carcelia normula (Curran). Total parasitism could reach 25% in the dry season. Pupae have been observed to be parasitized by two species of Brachymeria (Chalicididae), while the ants Camponotus rufoglaucus Forel and Pheidole megacephala (F.) preyed on larvae. The pathogenic fungus Beauveria bassiana was observed on larvae in the field during the rainy season. Although parasitoids and predators occur at low densities, they help to keep the population of A. acerata under the control threshold.
Biopesticides. Bacillus thuringiensis has the potential to replace chemical insecticides in the control of A. acerata, although more research is needed to test its field efficacy and commercialization.
Cultural control. Use of planting material free from A. acerata eggs or young larvae can prevent infestations of new fields. Application of wood ash directly onto A. acerata larvae has shown to cause larval mortality. Intercropping sweetpotato with onion (Alium cepa L.) and/or the silverleaf desmodium, Desmodium uncinatum (Jacq.) DC, might reduce the number of eggs laid by the females on sweetpotato.
Mechanical control. Hand-picking 1–3 instar larvae when they are still in a batch can also reduce A. acerata populations in case sufficient labor is available.
Chemical control. It has been inevitable for farmers to use contact insecticides during outbreaks. However, calendar insecticide spraying should be avoided in years without outbreaks as this would kill natural enemies (parasitoids) and hence increase the pest status. No resistance to insecticides is known in A. acerata, so less-toxic contact insecticides such as permethrin, cypermethrin, or dimethoate can be used, considering all safety measures during application and storage of the insecticide.
Further reading
Azerefegne, F., and C. Solbreck. 2010. Oviposition preference and larval performance of the sweet potato butterfly Acraea acerata on Ipomoea species in Ethiopia. Agricultural and Forest Entomology 12: 161–168.
Larsen, T.B. 1991. The Butterflies of Kenya and their Natural History. Oxford, New York: Oxford University Press.
Larsen, T.B. 2005. Butterflies of West Africa (Vols. I and II). Stenstrup, Denmark: Apollo Books.
Lugojja, F., M.W. Ogenga-Latigo, and N.E.J.M. Smit. 2001. Impact of defoliation on the agronomic performance of sweetpotato in Uganda. African Crop Science Journal 9(1): 103–108.
Matanmi, B.A., and T.J. Hassan. 1987. The life history and habits of Acraea eponina (Cramer) with notes on Acraea acerata Hewiston (Lepidoptera: Nymphalidae). Revue de Zoologie Africaine 101: 371–377.
Okonya, J.S., and J. Kroschel. 2013a. Incidence, abundance and damage by the sweetpotato butterfly (Acraea acerata Hew.) and the African sweet potato weevils (Cylas spp.) across an altitude gradient in Kabale district, Uganda. International Journal of AgriScience 3(11): 814–824.
Okonya, J.S., and J. Kroschel. 2013b. Pest Status of Acraea acerata Hew. and Cylas spp. in sweet potato (Ipomoea batatas (L.) Lam.) and incidence of natural enemies in the Lake Albert Crescent agro-ecological zone of Uganda. International Journal of Insect Science 5: 41–46.
Smit, N.E.J.M, F. Lugojja, and M.W. Ogenga-Latigo. 1997. The sweetpotato butterfly (Acraea acerata Hew., Nymphalidae): A review. International Journal of Pest Management 43: 275–278.