4.4.2 Maize Pests / Maize stalk borer, Busseola fusca (Fuller 1901)![]()
Synonyms: Sesamia fusca (Fuller 1901)
Busseola sorghicida (Thurau 1904)
Taxonomic position: Insecta, Lepidoptera, Noctuidae
Authors: G. Ong’amo, N. Khadioli, B. Le Ru, N. Mujica, & P. Carhuapoma
Common names
Maize stalk borer, African maize stalk borer, sorghum stalk borer (English), Perceur de la tige du maïs (French), Maisstengelbohrer (German)
Hosts
Busseola fusca exhibits oligophagous feeding behavior on host plants belonging to the family Poaceae. Primary host plants are maize (Zea mays L.) and sorghum [Sorgum bicolor (L.) Moench]; finger millet [Eleusine coracana (L.) Gaertn] and pearl millet [Pennisetum glaucum (L.) Leeke] constitute secondary host plants. Wild hosts include several species of wild grasses such as wild Sudan grass [Sorghum verticilliflorum (Steud.) Stapf, Sorghum arundinaceum (Desv.) Stapf, Cymbopogon nardus (L.) Rendle, Panicum maximum Jacq., Pennisetum purpureum Schumach, and Arundo donax (L.)].
Detection and identification
Host-plant parts affected are leaves, stems, and cobs (maize) as well as inflorescences of maize, sorghum, millet, and wild grasses. Leaf damage. First instar larvae feed on young terminal leaf whorls producing characteristic patterns of small holes and “window panes” (patches of transparent leaf epidermis) where tissue has been eaten away (Photo 1A, B). Later, they eat into the growing points which may be killed so that the dead central leaves form characteristic dry, withered “dead hearts” (Photo 1D). Stem damage. Older larvae tunnel extensively into stems and eat out long frass-filled galleries, which may weaken stems and cause them to break (Photo 1C). If the plant dies, the larvae will move to another plant. If the plant survives, the later stages of the larva will bore into the stem and feed on the meristematic tissue of the stem. Some larvae bore in the cobs. Seed damage. When larvae feed on seeds in the cob, frass deposits are visible (Photo 1E, F).

Morphology
Egg
Eggs are 0.8–1 mm in diameter and are creamy-white when laid but darken just before emergence (Photo 2A). They are hemispherical and slightly flattened with radial ridges on the upper surface of each egg shell.
Larva
First instars are light colored at first with a dark head and markings on the body segments; the dark markings become more conspicuous up to the third instar (Photo 2B). The body of the mature larva is buff, dorsally suffused with pale purple-gray, and lack conspicuous hairs or markings. Head is medium brown, and a prothoracic shield usually paler than head; pinnacula brown, caudal plate matching prothoracic shield in color. Their spiracles are elongate-oval with black edges. Larvae have prolegs along the abdomen and are up to 40 mm long and 5 mm wide.
Pupa
The pupa is dark brown and measures about 25 mm long (Photo 2C).
Adult
The adult wingspan measures about 25–40 mm. The front wings and the body are usually dark brown; lighter-colored specimens are not uncommon (Photo 2D, E). Its hind wings are almost white with a smoky tinge and dark longitudinal lines indicating the veins.

Biology
Female B. fusca moths lay eggs in a row between the stem and leaf sheath (Photo 2A). Each female lays on average about 200 eggs over its short lifespan of several days; its exact duration depends on temperature and other factors. Egg-laying on maize is usually concentrated on plants that are less than 2 months old. The leaf sheath of the youngest unfolded leaf is the most attractive part for females to deposit eggs. The eggs hatch in 3–5 days and larvae move into the leaf whorls to feed. Third instar larvae tunnel into stems where they feed for 3–5 weeks before pupating within the stem tunnels. The adult moth emerges after a pupal period of 7–14 days from a hole that they produced before pupation. Adults mate soon after emergence. Under favorable conditions, the life cycle can be completed in 7–8 weeks. But during dry and/or cold weather conditions, the larvae can enter a period of suspended development (diapause) of 6 months or more in stems and other plant residues.
Temperature-dependent development
B. fusca successfully developed from egg to adult at temperatures of 15º–30ºC but failed at 12°C and 35ºC as no eggs hatched at these temperatures (see Annex 7.3.14). The total mean development time decreased with increasing temperature, from 223.9 days at 15°C to 72.5 days at 25°C, and then increased slightly to 81.3 days at 35ºC. The percentage mortality of eggs, larvae, and pupae decreased at temperatures of 15º–25ºC and increased thereafter. The minimum mortality for egg (37.4%), larvae (22.9%), and pupae (7.7%) was registered at 25ºC. The mean longevity significantly decreased with increasing temperature, with 12.4 and 11.7 days at 15ºC and 4.7 and 4.4 days at 30ºC for females and males, respectively. The maximum fecundity of 243.5 eggs was observed at 18ºC.
The models established to describe the development time, survivorship, and reproduction were compiled into an overall stochastic phenology model that allows the B. fusca life-table parameters to be estimated according to temperature (see Annex 7.3.14). The predicted intrinsic rates of natural increase (rm) indicate that populations develop within temperatures of 16º–31.5ºC. The negative r values below 16ºC and above 31.5ºC indicate that population size is decreasing due to high mortality or very low reproduction. The highest population growth can be expected at 24.5ºC (rm = 0.0496). Similarly, the finite rate of increase for B. fusca peaked at 29ºC (λ = 1.0509) and was smallest when exposed to 15.5ºC (λ = 0.996) and 35.5ºC (λ = 0.994); λ values of <1 indicate that the population is decreasing. At 24.5ºC doubling time (Dt) was shortest with 13.9 days. The highest values for reproductive parameters were at 21ºC for gross reproductive rate, with 274.7 female offspring/female. When mortality of females before reproduction was considered, the net reproductive rate (Ro) was highest at 22.5ºC, with around 44.9 female offspring/female. The mean generation time (T) decreased with temperature and was shortest at 26.5ºC with 70.3 days from egg to egg. These simulations indicate that B. fusca is able to establish in most tropical and subtropical maize production areas of the world.
Means of movement and dispersal
The B. fusca moth dispersal by flight was assumed to be mainly local with short distances; however, recent release-recapture experiments in Kenya indicated flight distances of >10 km/night (P. Campagne and B. Le Ru, unpublished data). Larvae and eggs may be transported on growing host plants. Long-distance spread is through translocation of stems and residues from one location to another where they are used as animal feed.
Economic impact
Larval feeding on leaves and stems may reduce grain production and kill the plant. Maize plants are less tolerant to stemborer attack than are sorghum and pearl millet, and hence losses of maize grain yields are greater. In South Africa, B. fusca infestation results in yield losses of 10–100%. In Tanzania and Kenya, losses of about 12% of maize grain for every 10% plants infested by B. fusca were reported. Recent studies in Kenya indicated that more than 10% of potential maize harvest is lost due to infestation by B. fusca in highland tropics, moist transitional zones, and moist mid-altitude zones.
Geographical distribution
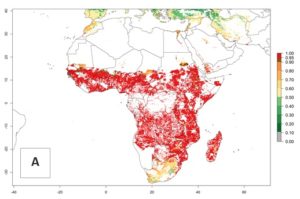
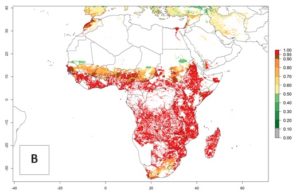
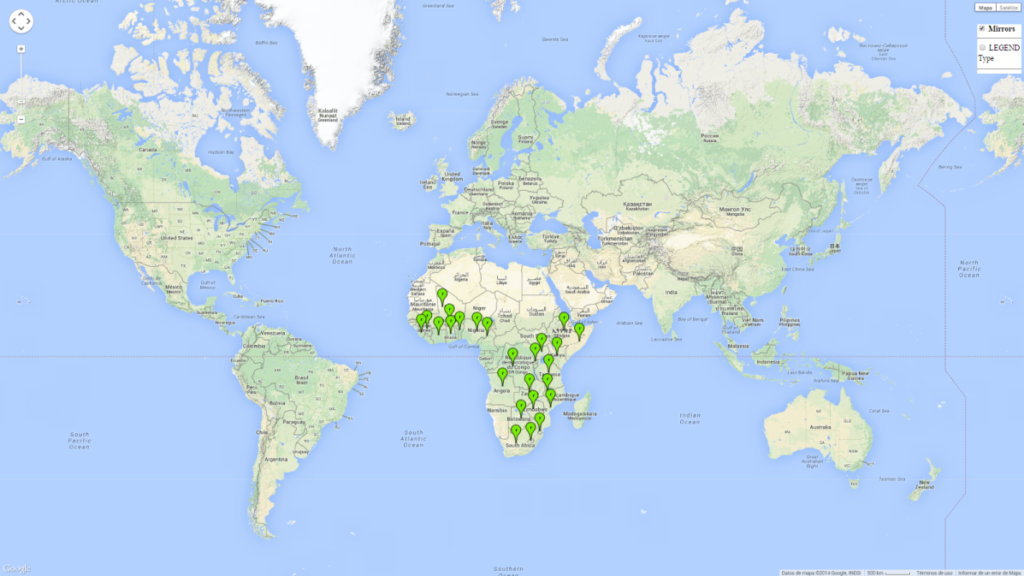
B. fusca is native to sub-Saharan Africa (SSA) and is not known to occur anywhere outside the African continent (Fig. 1).
| Africa | West Africa (at sea level to altitudes of 2,000 masl): Benin, Burkina Faso, Cameroon, Ghana, Guinea, Ivory Coast, Mali, Nigeria, Sierra Leone. East Africa (occurs at 600–2,700 masl): Ethiopia, Kenya, Somalia, Tanzania, Uganda. Central Africa (all altitudes): Congo, DR Congo, Gabon, Equatorial Guinea. Southern Africa (at elevations above 900 masl): Angola, Botswana, Lesotho, Malawi, Mozambique, Rwanda, South Africa, Swaziland, Zaire, Zambia, Zimbabwe. B. fusca is absent in the coastal areas of Kenya and Tanzania. |
Phytosanitary risks
B. fusca is listed as one of the important crop pests in the CABI Crop Protection Compendium. It is a pest of significance under surveillance by national plant health institutions in SSA countries that are obligated by the International Plant Protection Convention to discharge plant protection functions. Most of these countries have the Plant Protection Act that offers guidelines on actions to be taken to eradicate, reduce, or prevent the spread of such pests.
Risks mapping under current and future climates
Global Risks
Changes in establishment and future distribution
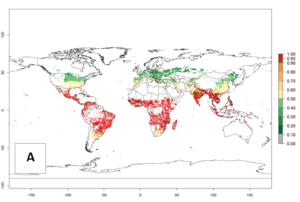
An establishment risk index (ERI)>0.95 indicates that the temperature conditions are suitable to support the permanent establishment of B. fusca populations, and an ERI<0.95 indicates that the likelihood of long-term establishment is reduced. The ERI>0.95 reflects the B. fusca current distribution in SSA (Fig. 2A). B. fusca also occurs in South Africa, with an ERI>0.7–0.8; this might be due to diapausing last instar larvae during the southern winter (diapause in insect life cycles cannot be considered in Insect Life Cycle Modeling because factors responsible for the diapause are unknown). Although never recorded outside of SSA, B. fusca has a high potential of establishment in most tropical areas of North, Central, and South America as well as Southeast Asia.
Figure 2. Changes in establishment and potential distribution of the maize stalk borer, Busseola fusca, in maize production systems worldwide according to model predictions, using the ERI for the years 2000 (A) and 2050 (B), and changes of the ERI between 2000 and 2050 (C). An ERI>0.95 is associated with potential permanent establishment. The pest has been also observed at an ERI>0.7–0.8, in which regions B. fusca enters into diapause to overcome unsuitable conditions.
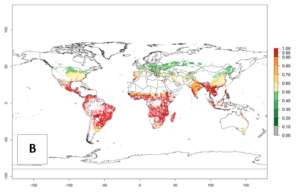
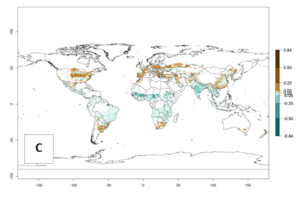
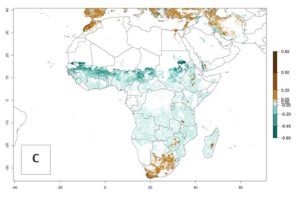
The predictions for 2050 indicate a decrease in the establishment potential of B. fusca in most lowland areas of West, East, and Central Africa, as well as outside its current range of distribution in Asia (e.g., India). Instead, a slight range expansion is potentially expected in highland maize production areas of East Africa and in Southern Africa (Fig. 2B, compare with Fig. 4C).
Changes in abundance
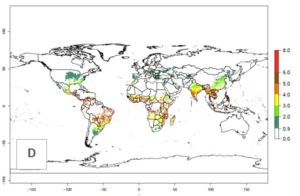
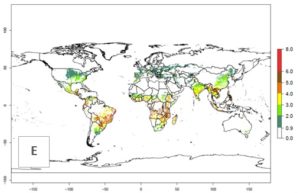
The mean number of generations that B. fusca might theoretically develop within a year in the year 2000 gave reasonable predictions when compared with generation numbers reported in the literature reaching up to 5 generations per year (Fig. 3A). The generation index (GI) change indicates that by the year 2050, an increase of up to 1 generation can be expected for most SSA countries in Central, East, and Southern Africa. However, higher changes are expected in midland and highland areas of East and Southern Africa (Fig. 3C). The activity index (AI) indicates the potential population growth throughout the year. A reduction by a factor of 1–2 is predicted for most parts of SSA lowlands, whereas an increase by a factor of 1–2 is predicted in most African highlands, particularly in East Africa (Fig. 3D–F).
| GI | AI | |
| 2000 | 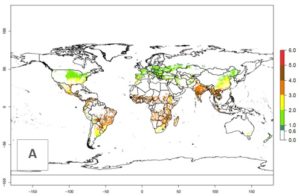 |
 |
| 2050 | 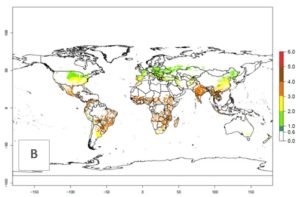 |
 |
| Index change (2000 – 2050) | 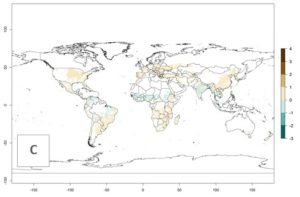 |
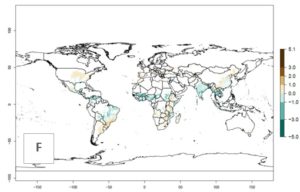 |
Figure 3. Changes in abundance (GI, damage potential) and activity (AI, potential population growth) of the maize stalk borer, Busseola fusca, in maize production systems worldwide according to model predictions, using the GI (A, B) and the AI (D, E) for the years 2000 and 2050, and the absolute index change (C, F).
Regional Risks for Africa
Changes in establishment and future distribution
The predictions for 2050 indicate a reduction in establishment in the Sahel of Mali, Burkina Faso, Chad, and Sudan. On the other hand, a slight range expansion is expected in most highland areas of East and Southern Africa (Fig. 4B). In the year 2050, B. fusca will continue to be a high risk pest (ERI>0.95) in most maize productions regions of Ethiopia, Kenya, Uganda, Rwanda, Burundi, Tanzania, Zambia, Angola, and Zimbabwe, whereas most of South Africa will become more suitable (Fig. 4C).
Figure 4. Changes in establishment and potential distribution of the maize stalk borer, Busseola fusca, in African maize production systems according to model predictions, using the ERI for the years 2000 (A) and 2050 (B), and changes of the ERI between 2000 and 2050 (C). An ERI>0.95 is associated with potential permanent establishment. The pest has been also observed at an ERI>0.7–0.8, in which regions B. fusca enters into diapause to overcome unsuitable conditions.
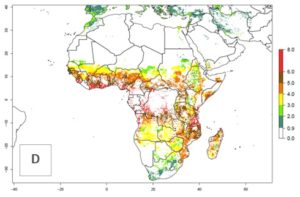
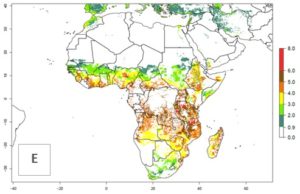
Changes in abundance
B. fusca theoretically develops 3–5 generations per year under the climate conditions of the year 2000 (Fig. 5A). The GI change indicates a decrease of up to 0.8 generations per year in countries of the Sahel in West and East Africa until the year 2050, whereas an increase of up to 1–2 generations per year is predicted for highlands of East Africa and most countries of Southern Africa (South Africa, Mozambique, Zimbabwe) (Fig. 5C). The AI indicates the potential population growth throughout the year. A reduction by a factor of 1–3 is predicted for most parts of SSA lowlands, whereas an increase by a factor of 1–3 is predicted in most African highlands, particularly in East Africa (Ethiopia, Kenya, Tanzania) and Angola (Fig. 5D–F).
| GI | AI | |
| 2000 | 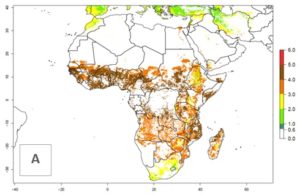 |
 |
| 2050 | 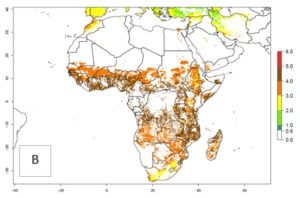 |
 |
| Index change (2000 – 2050) | 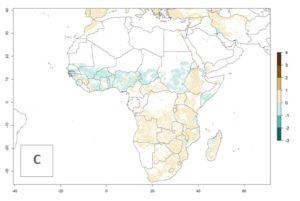 |
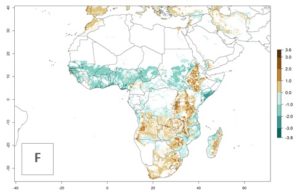 |
Figure 5. Changes in abundance (GI, damage potential) and activity (AI, potential population growth) of the maize stalk borer, Busseola fusca, in African maize production systems according to model predictions, using the GI (A, B) and the AI (D, E) for the years 2000 and 2050, and the absolute index change (C, F).
Country Risk Maps
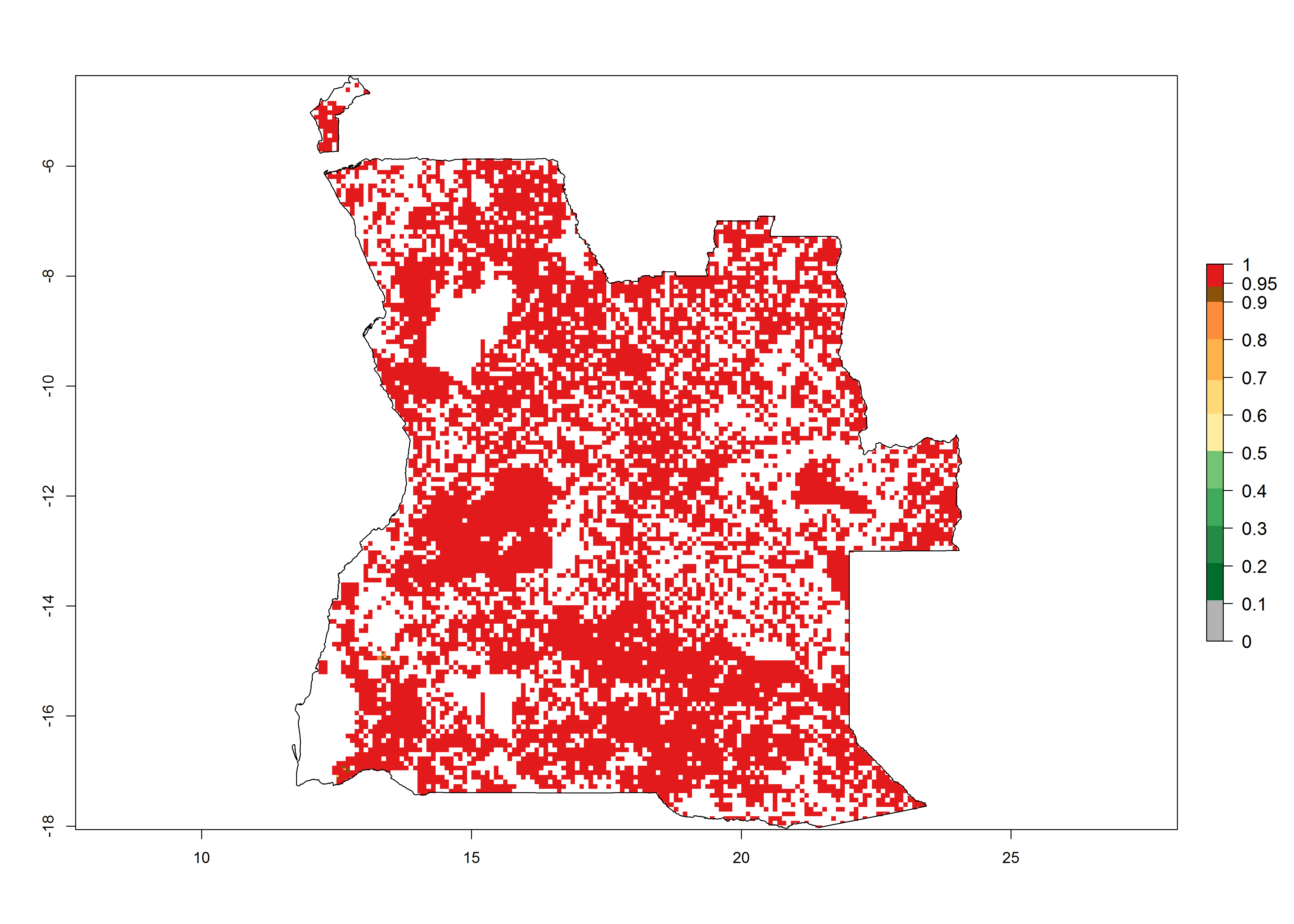
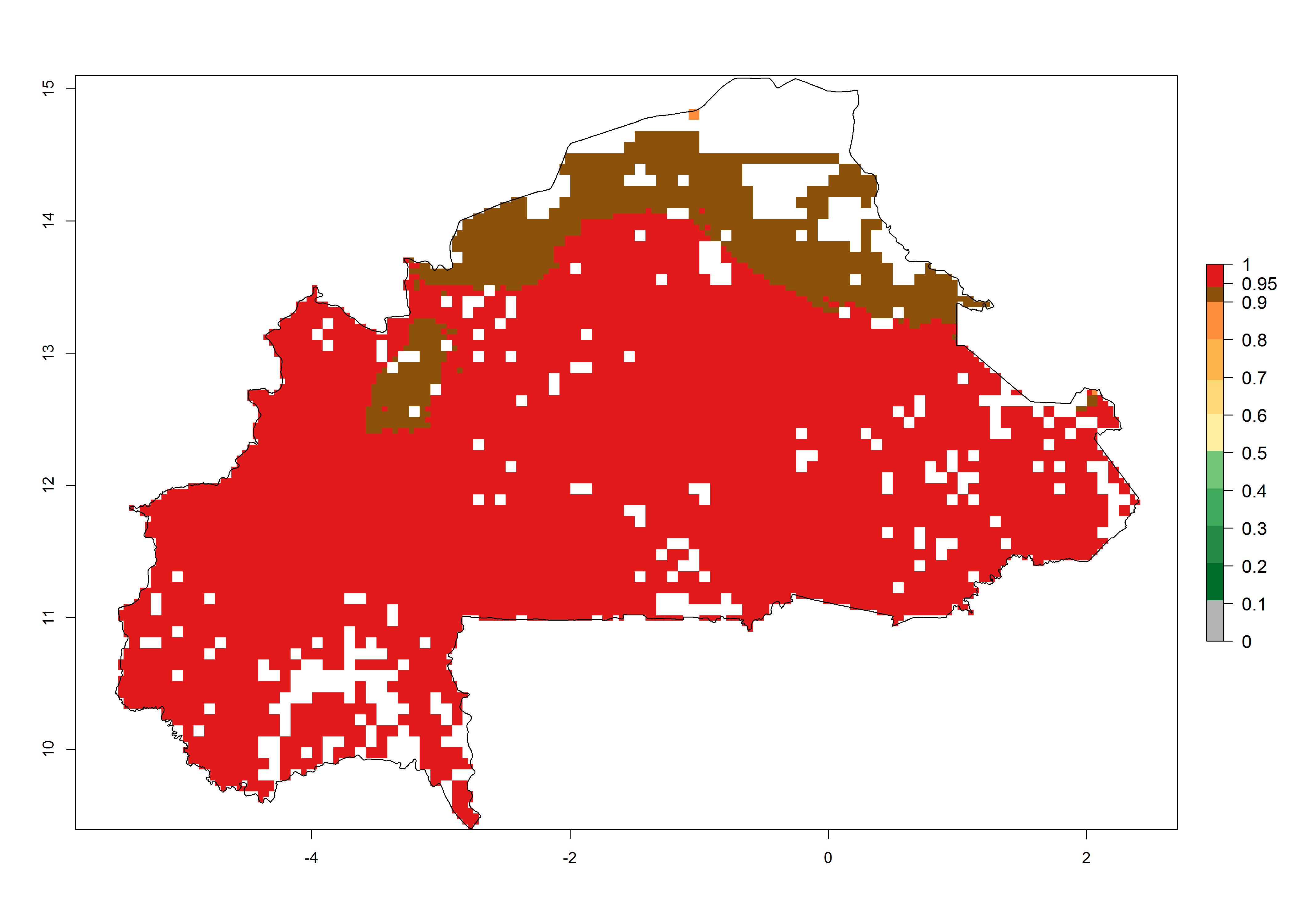
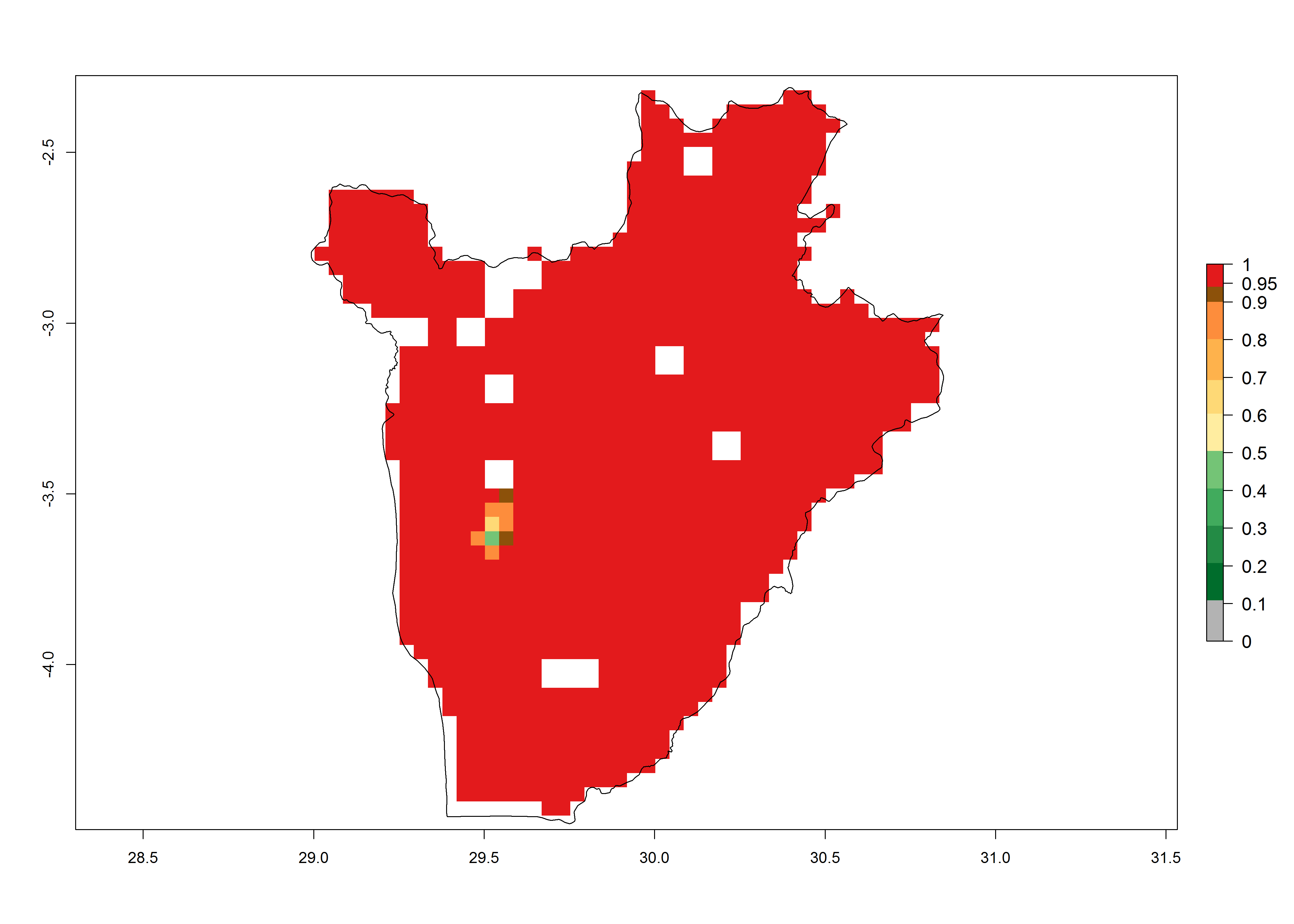
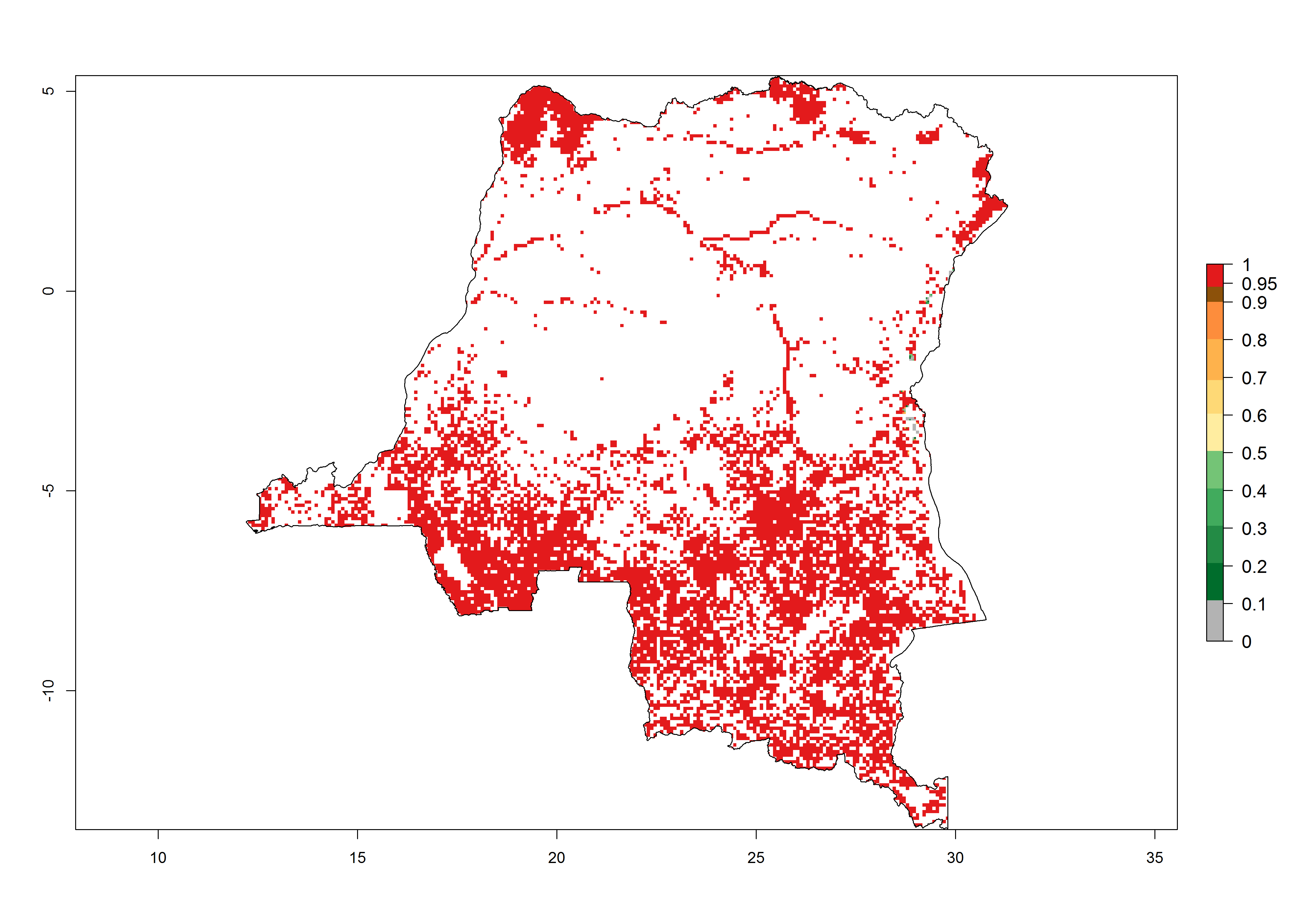
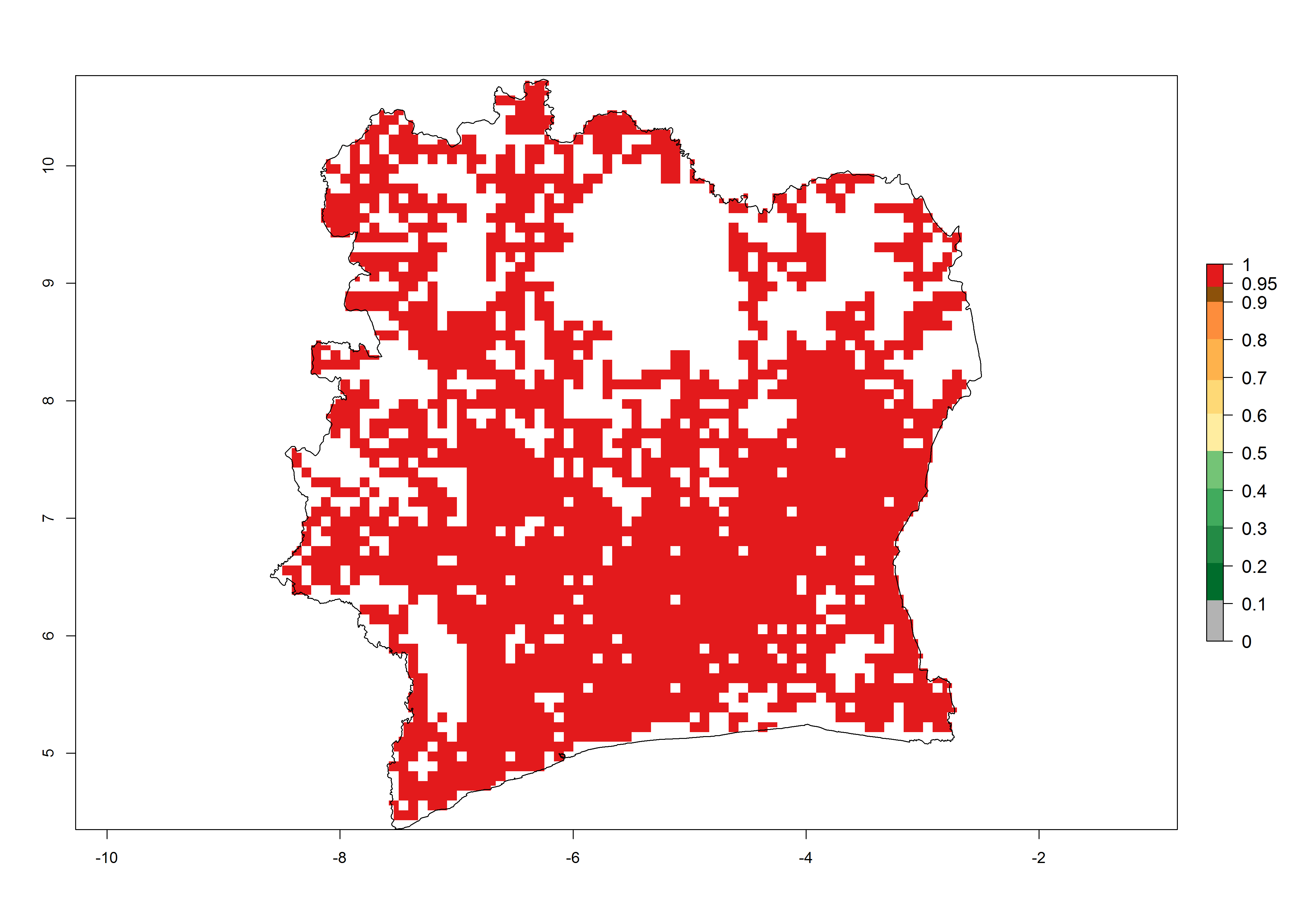
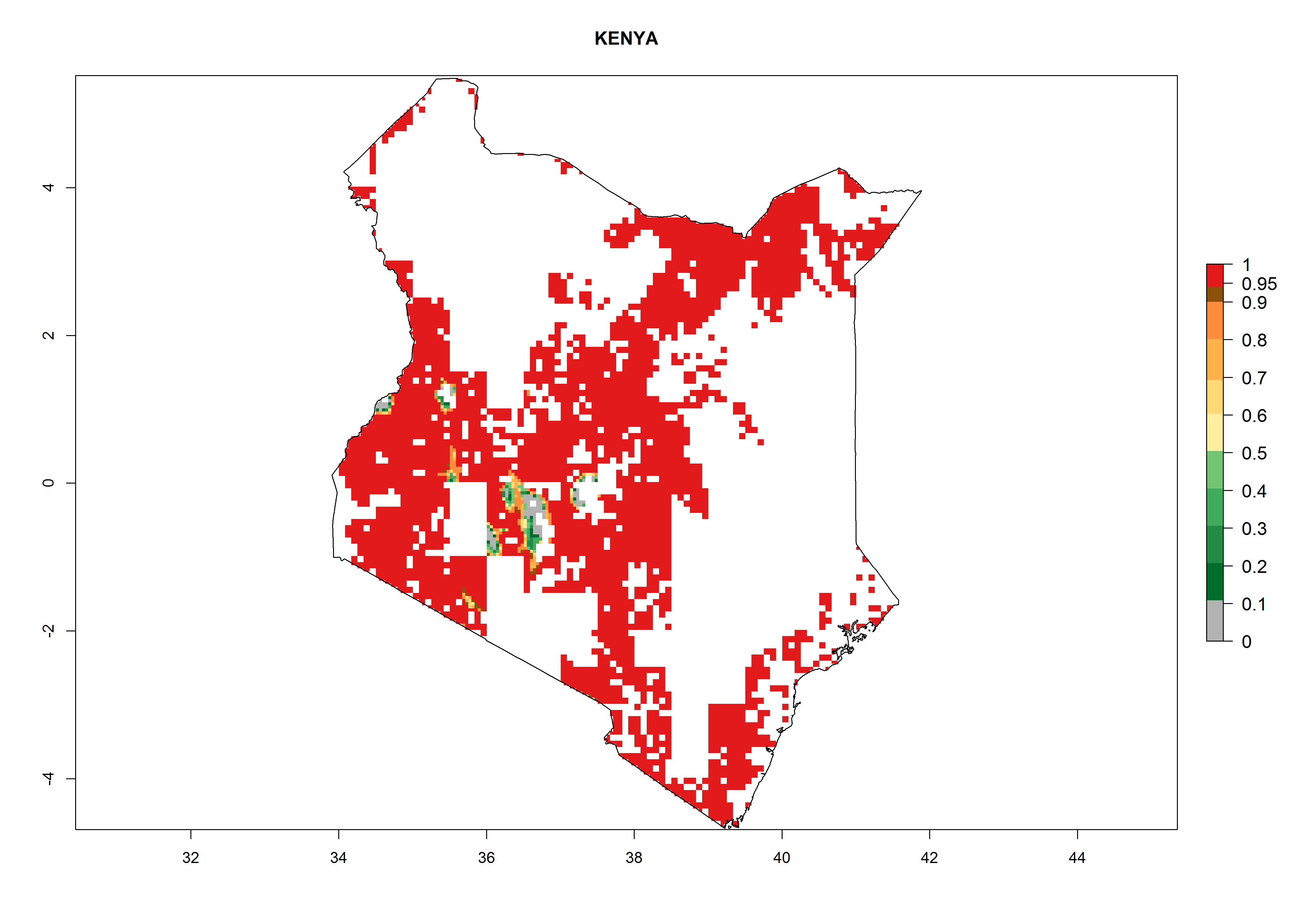
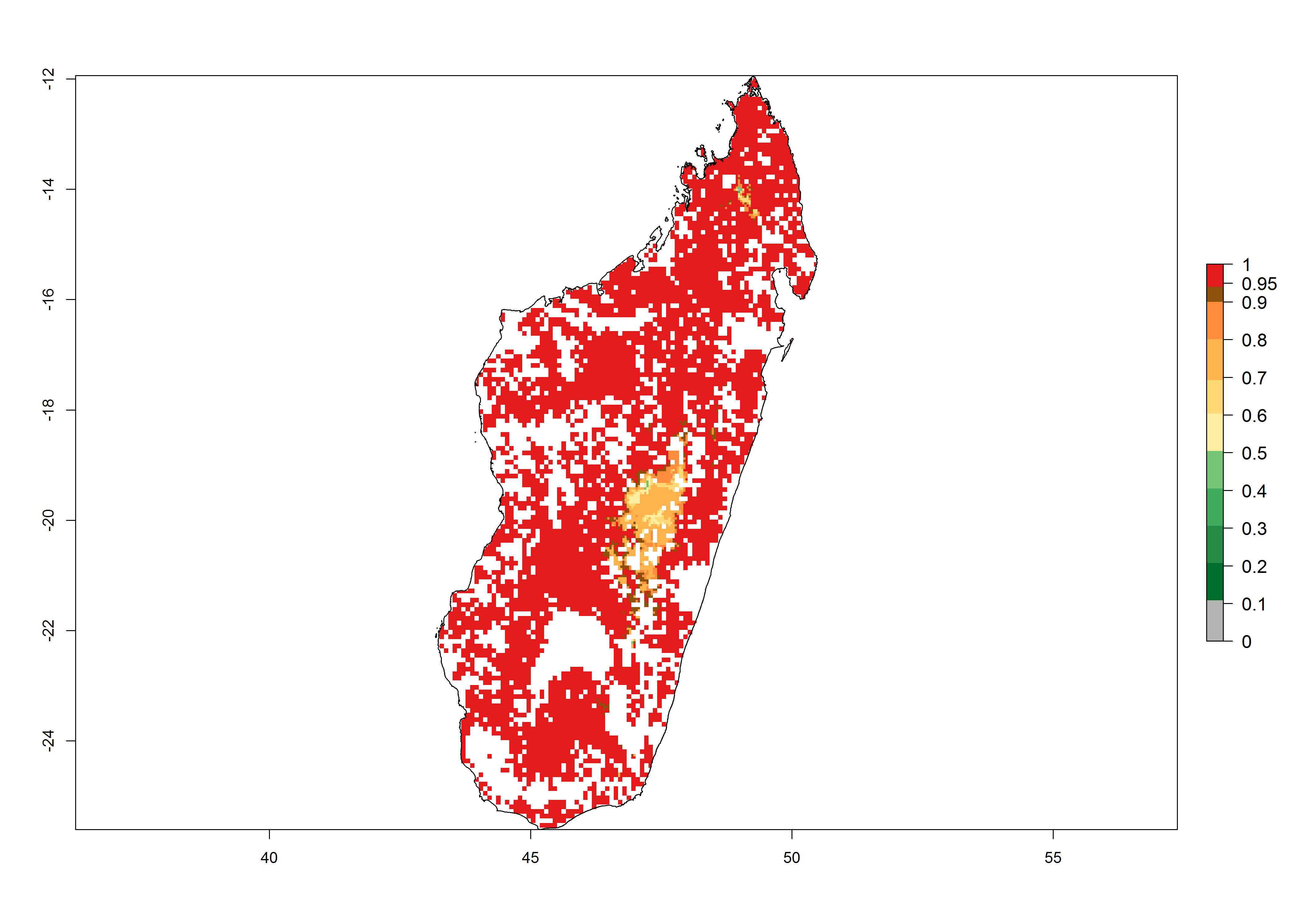
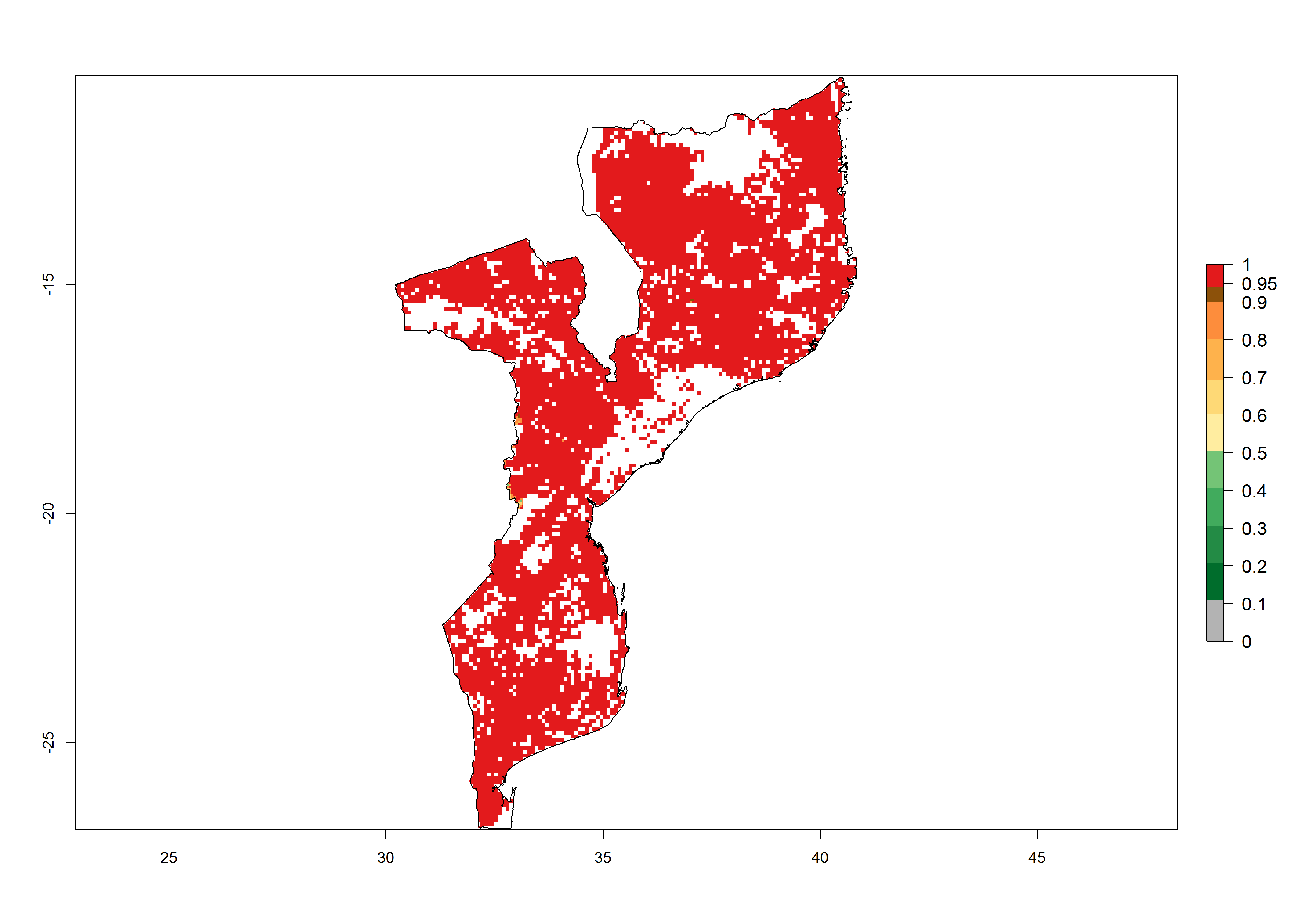
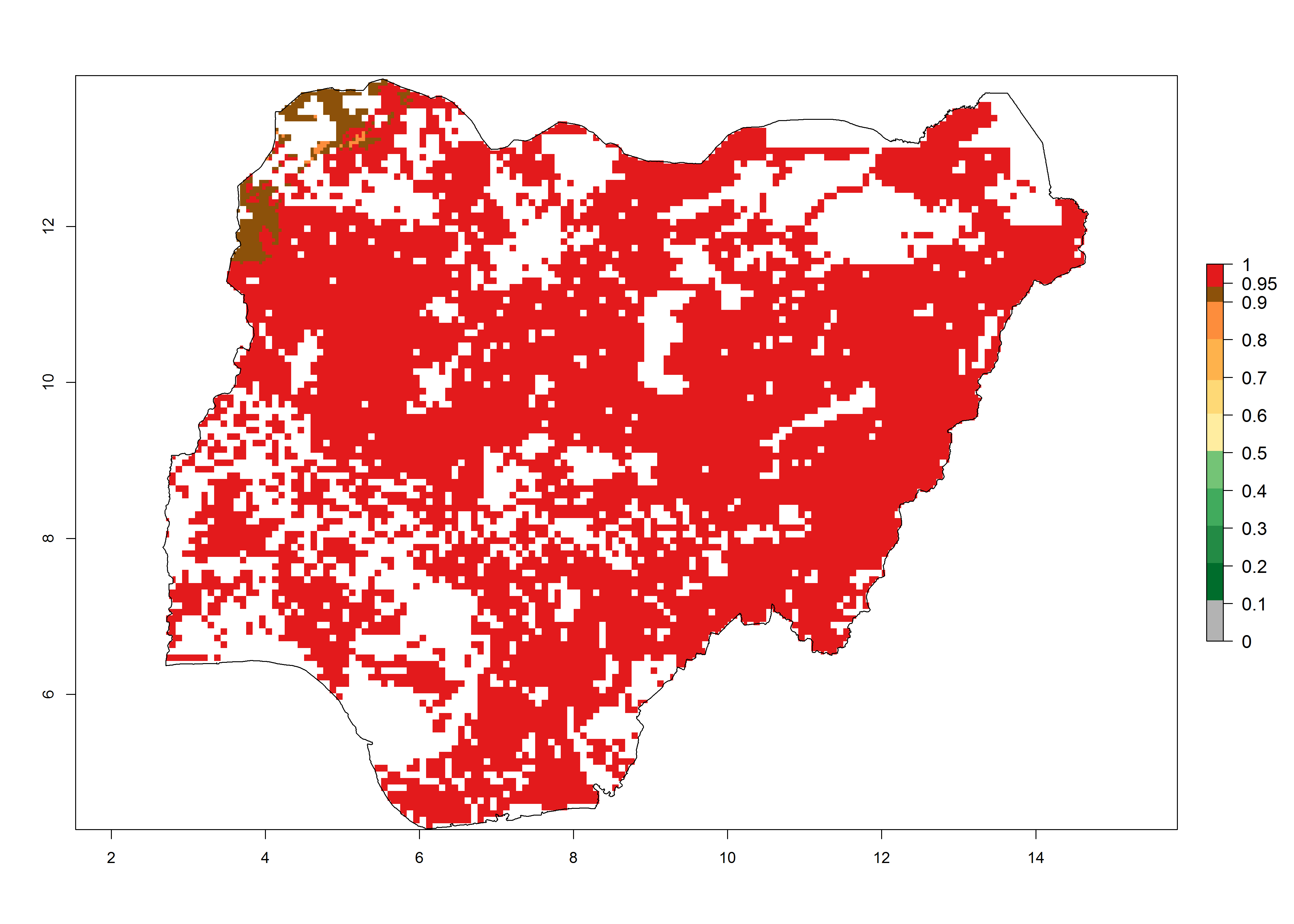
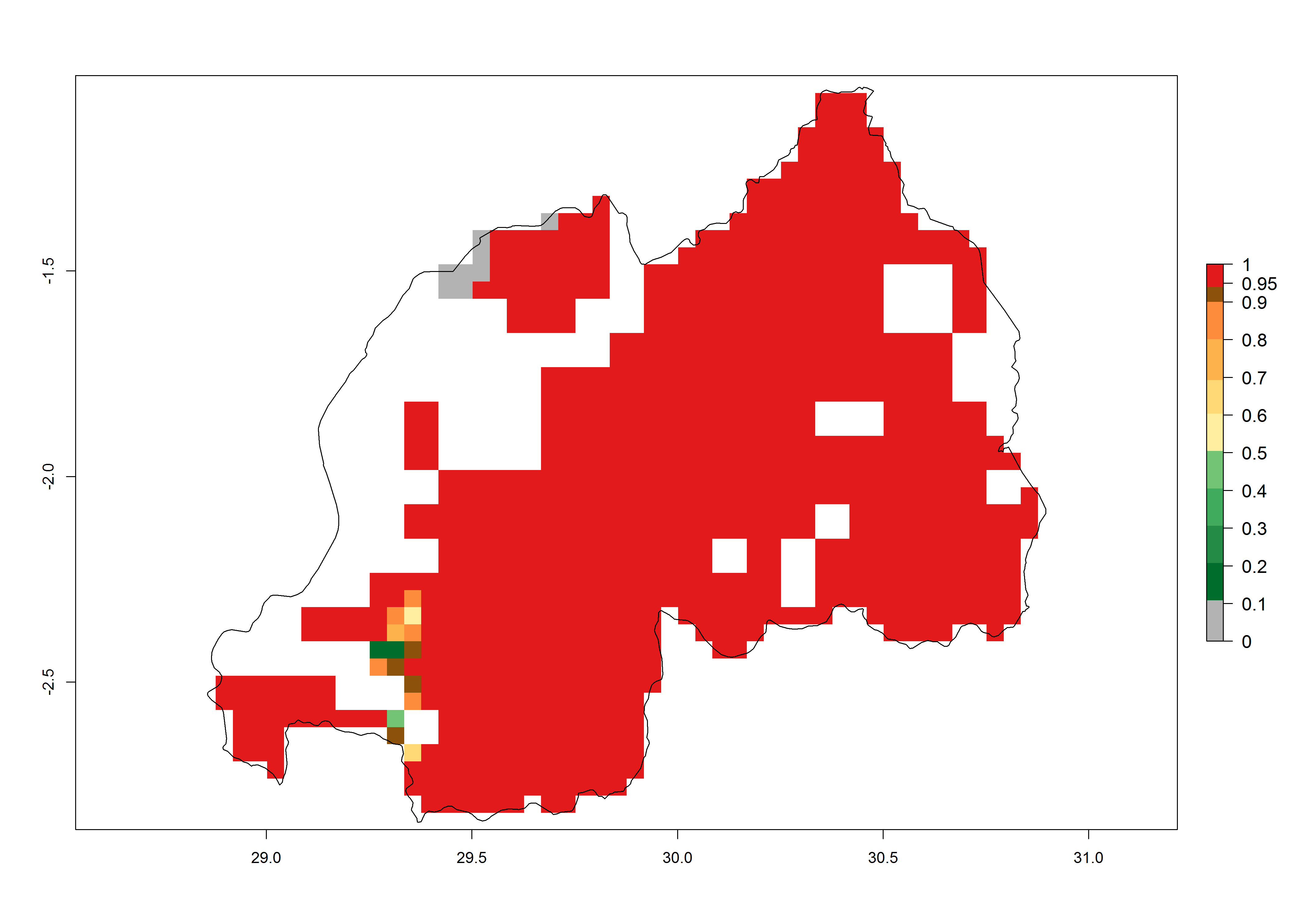
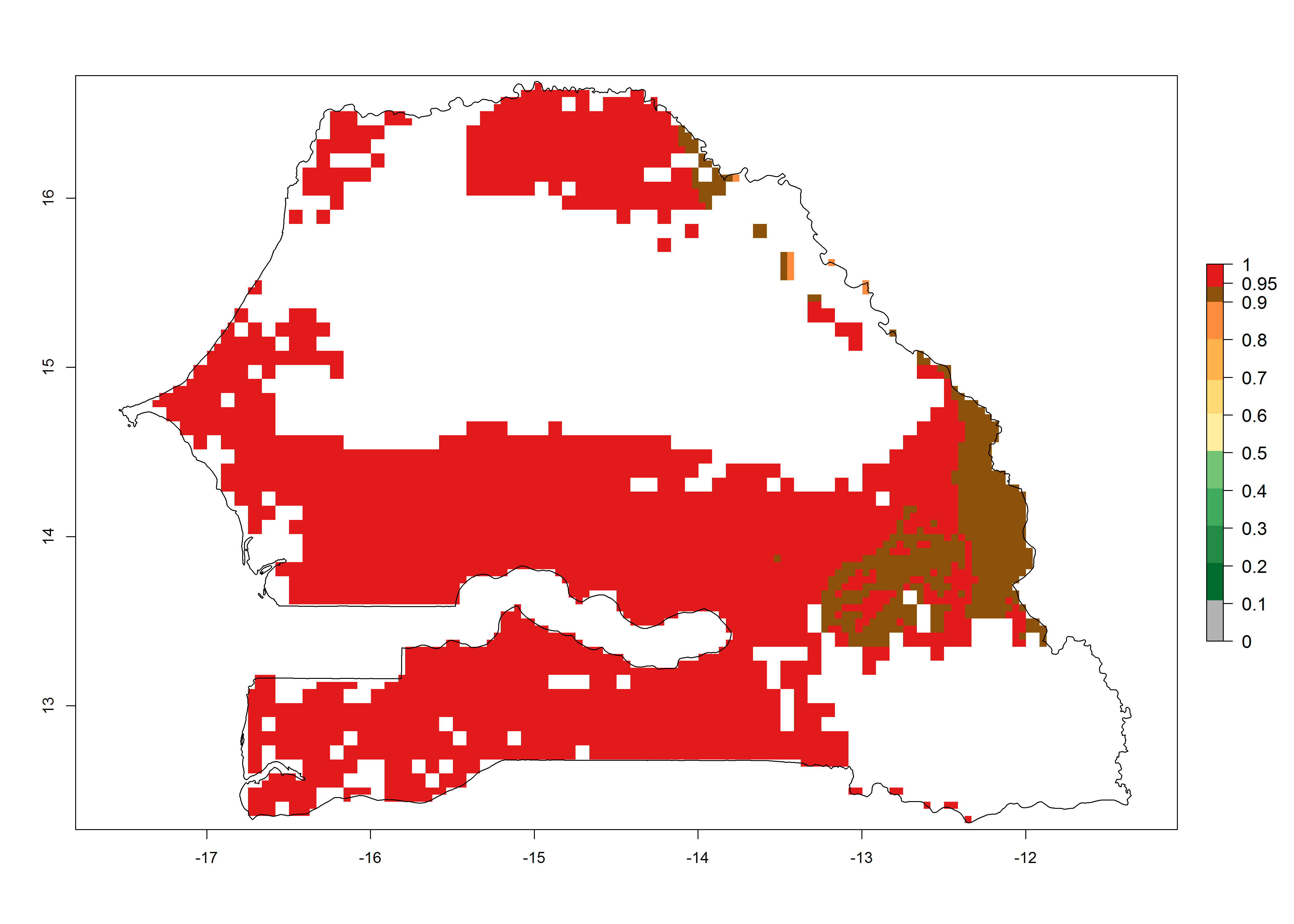
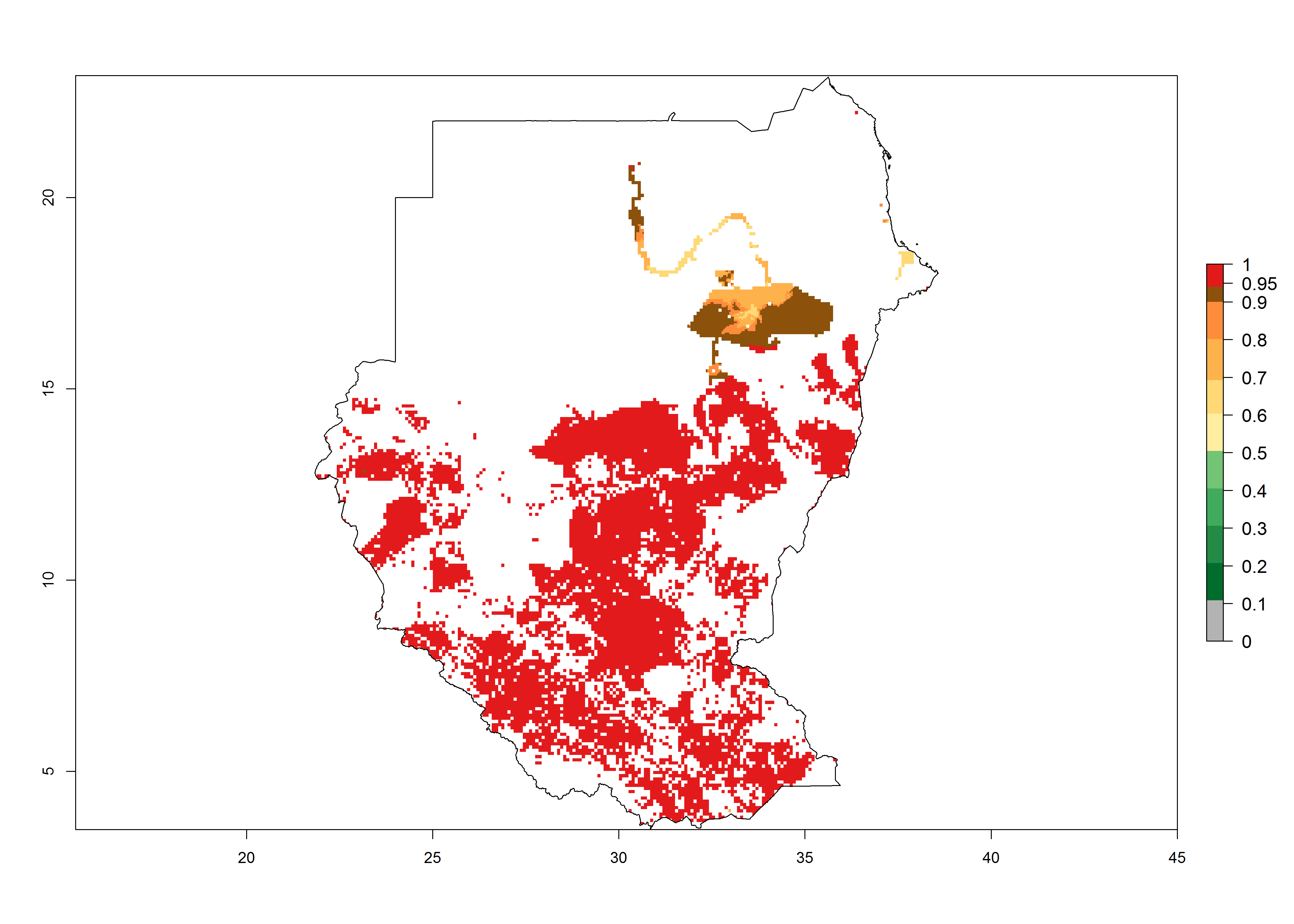
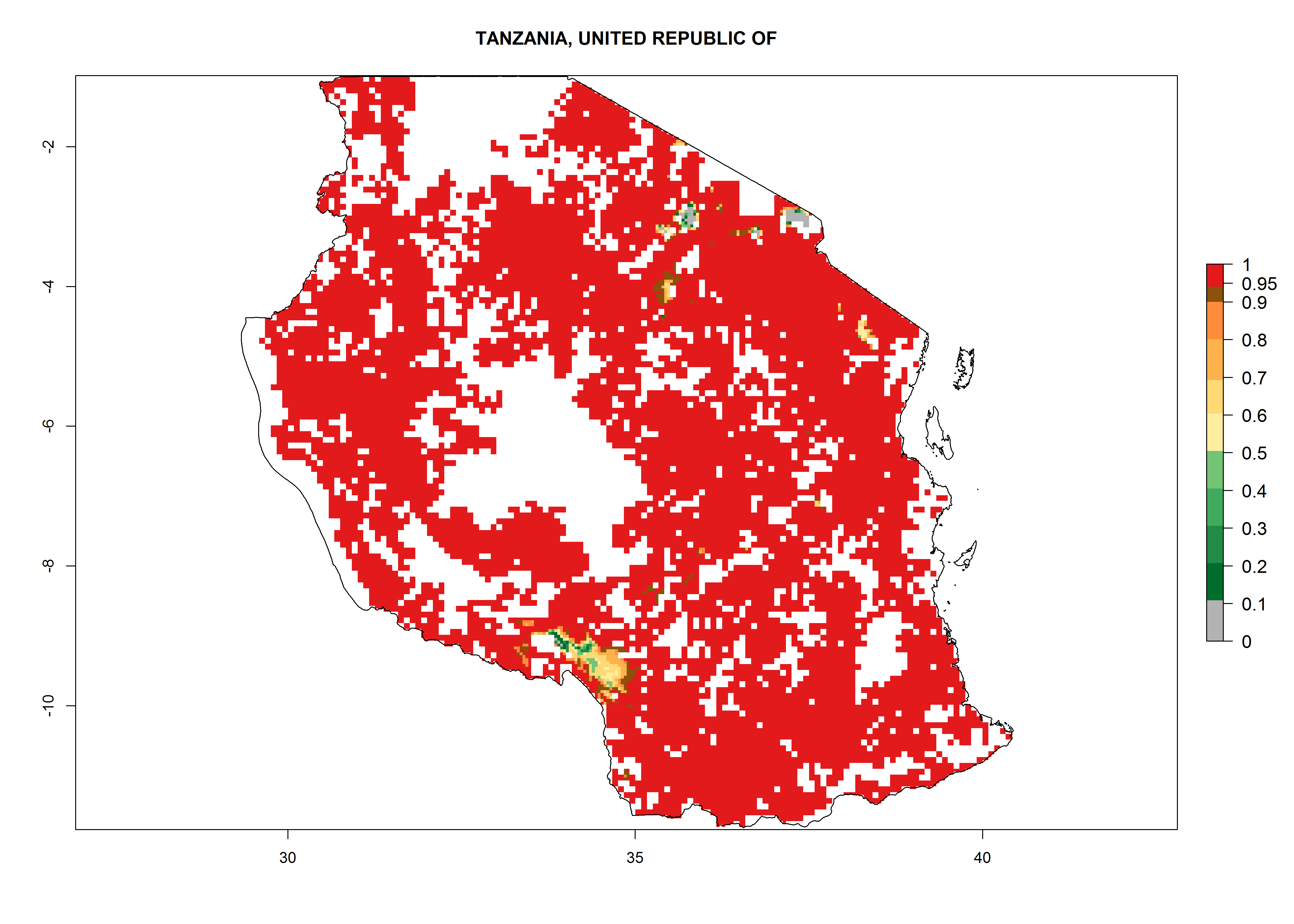
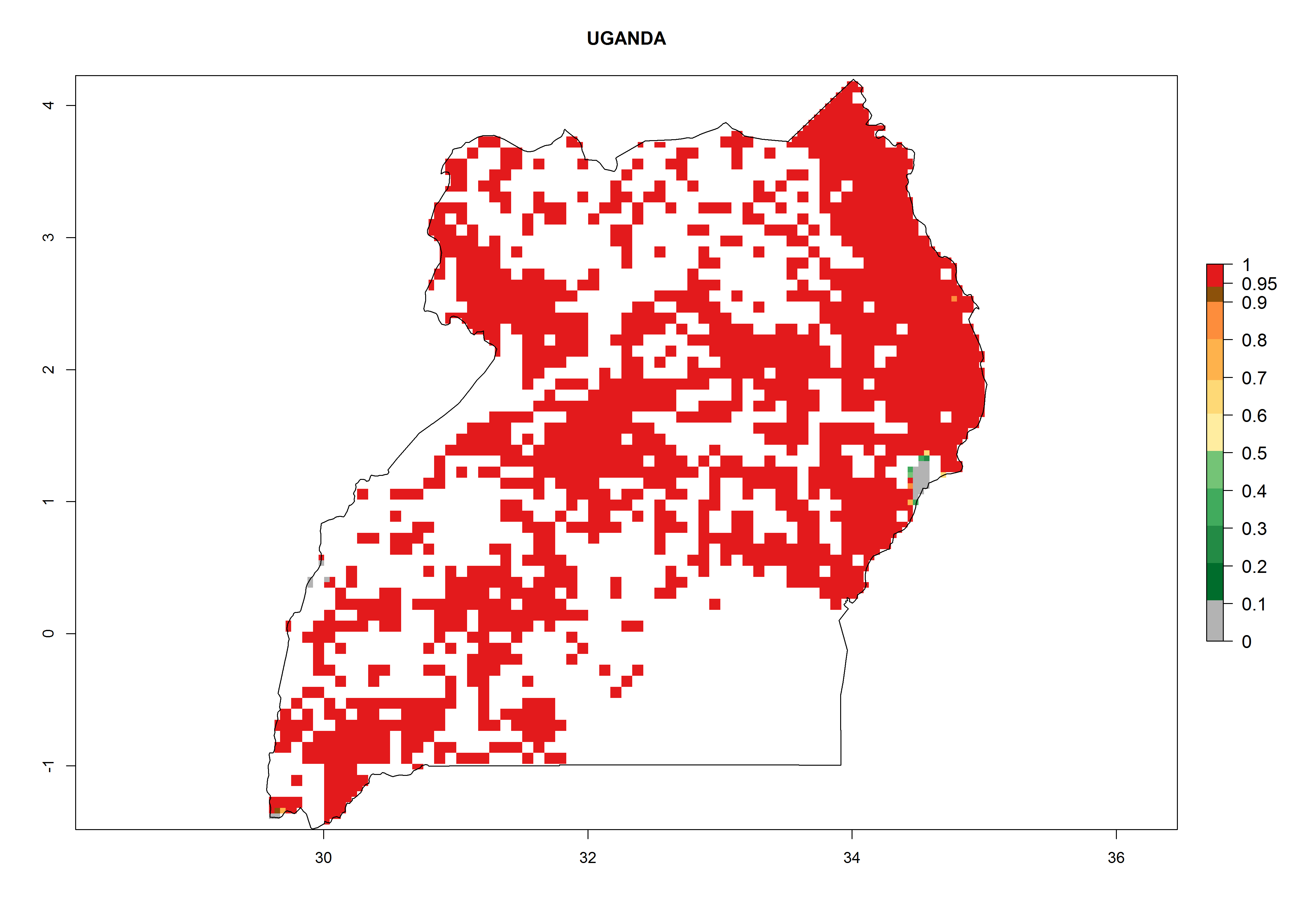
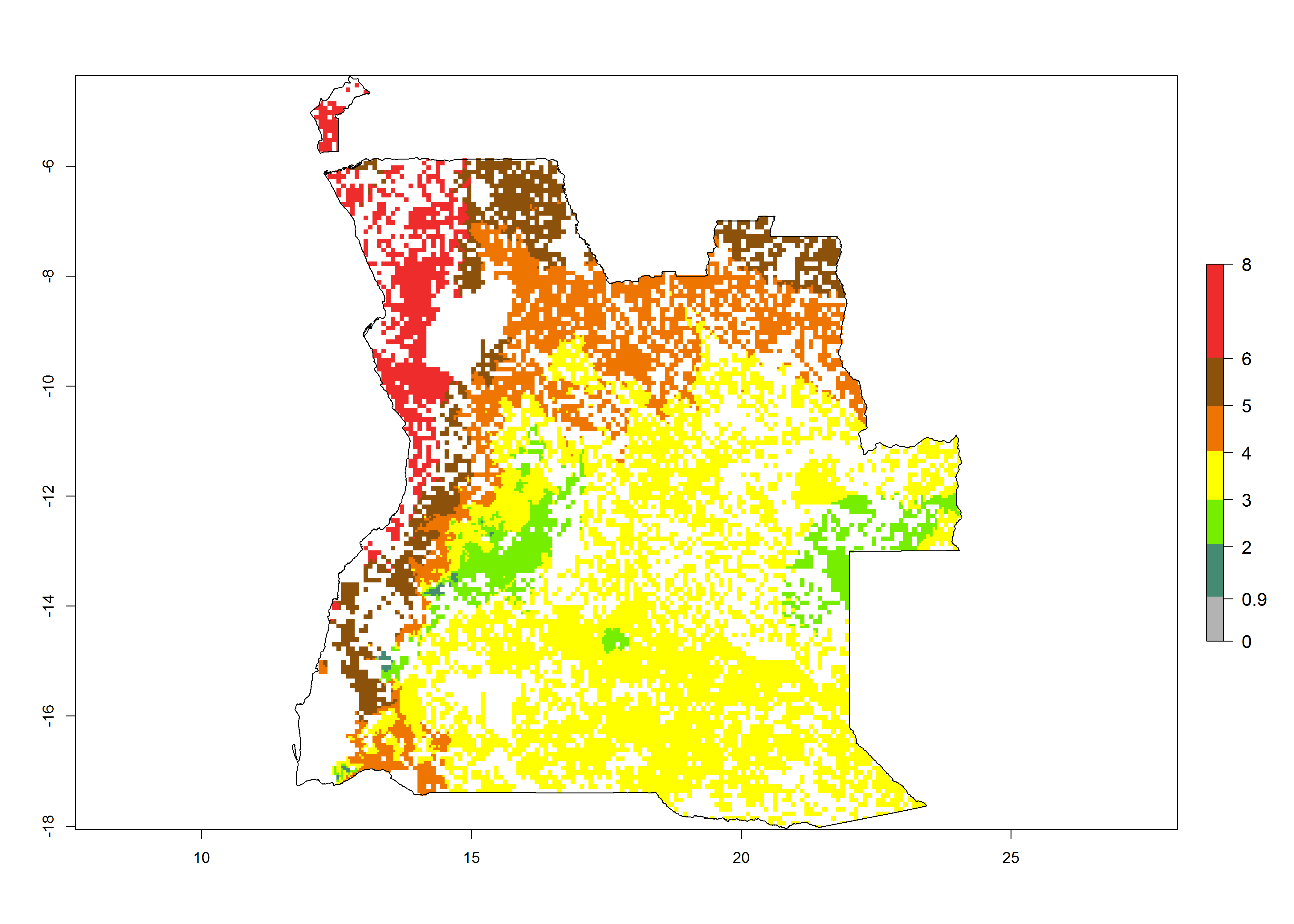
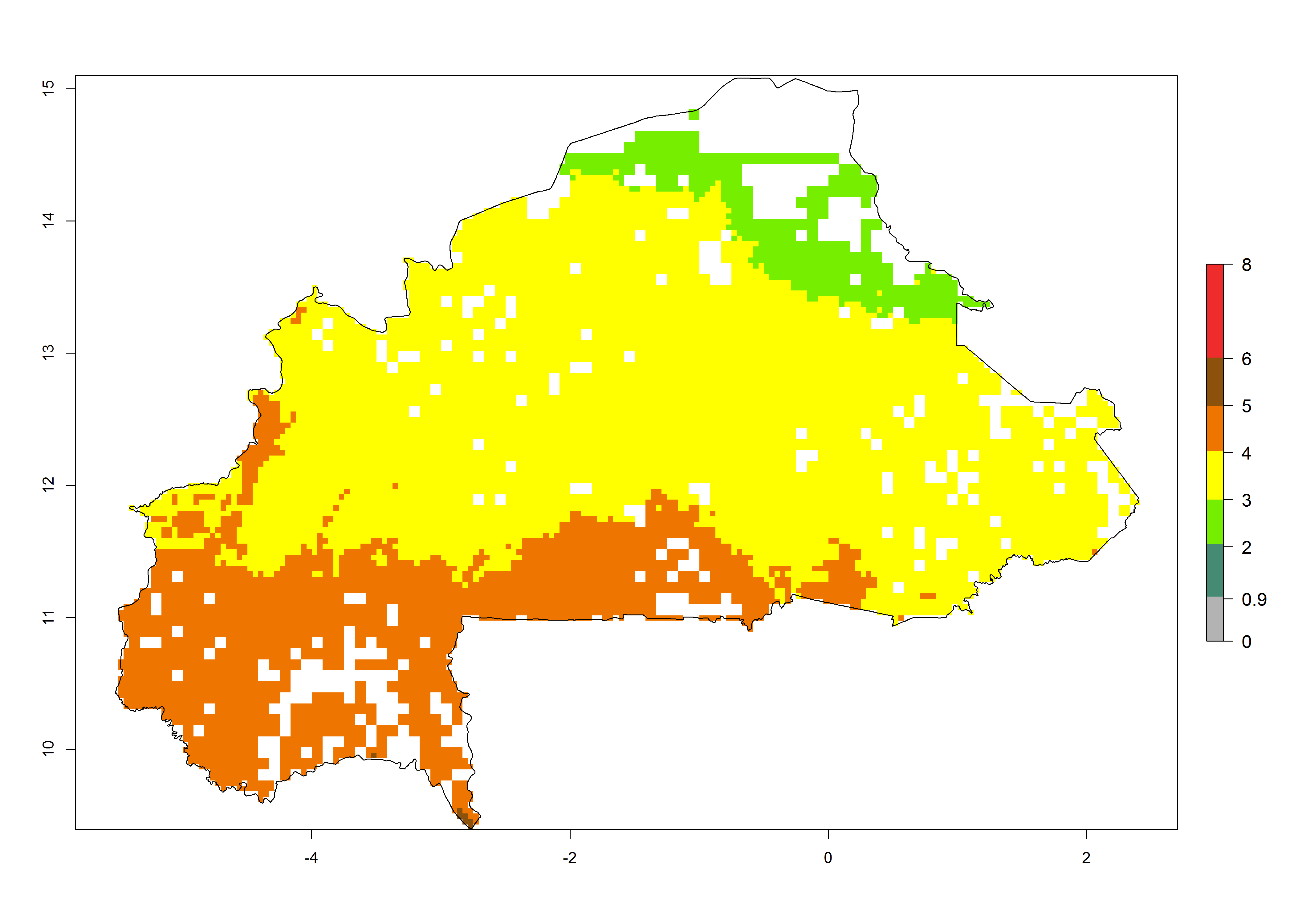
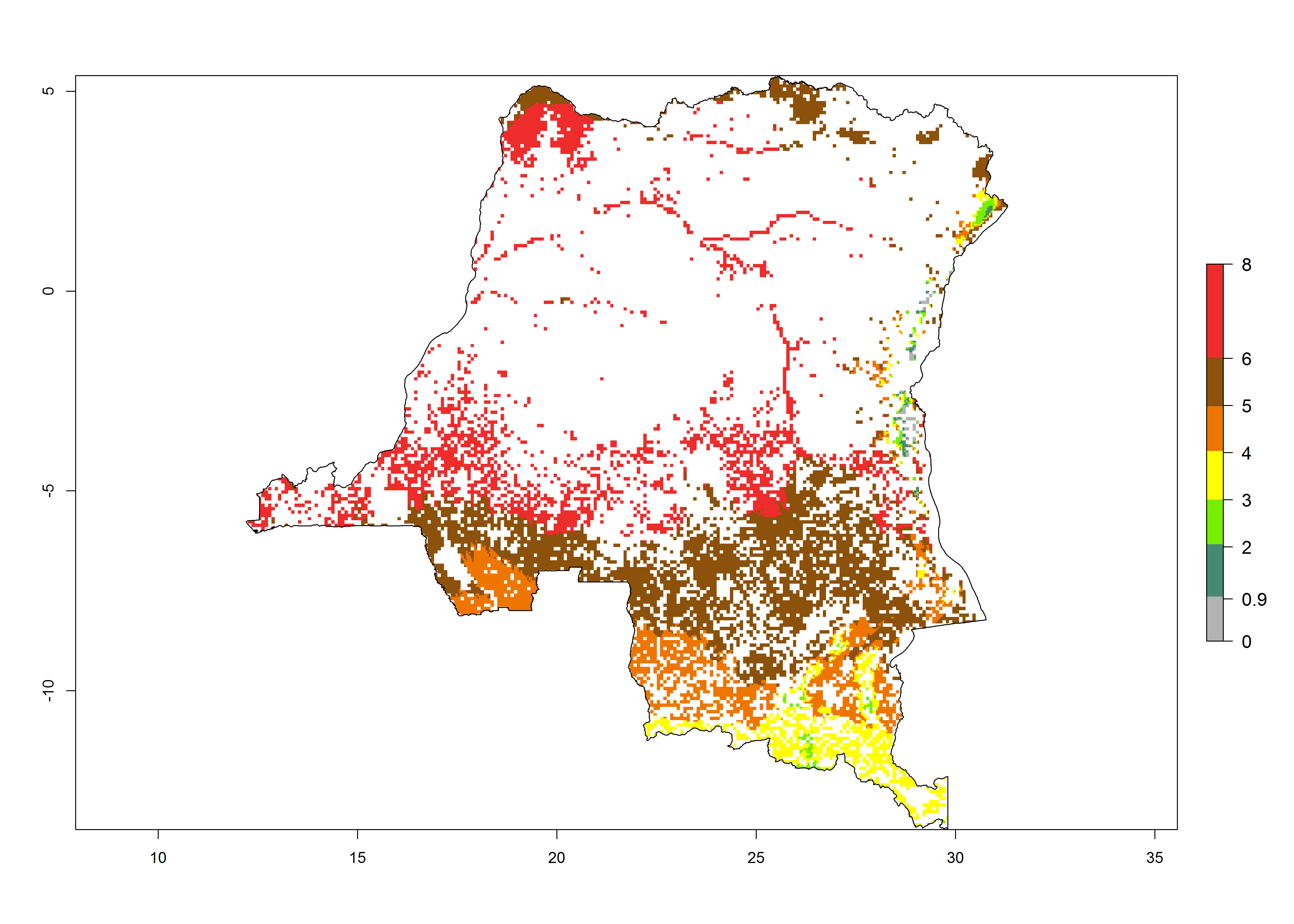
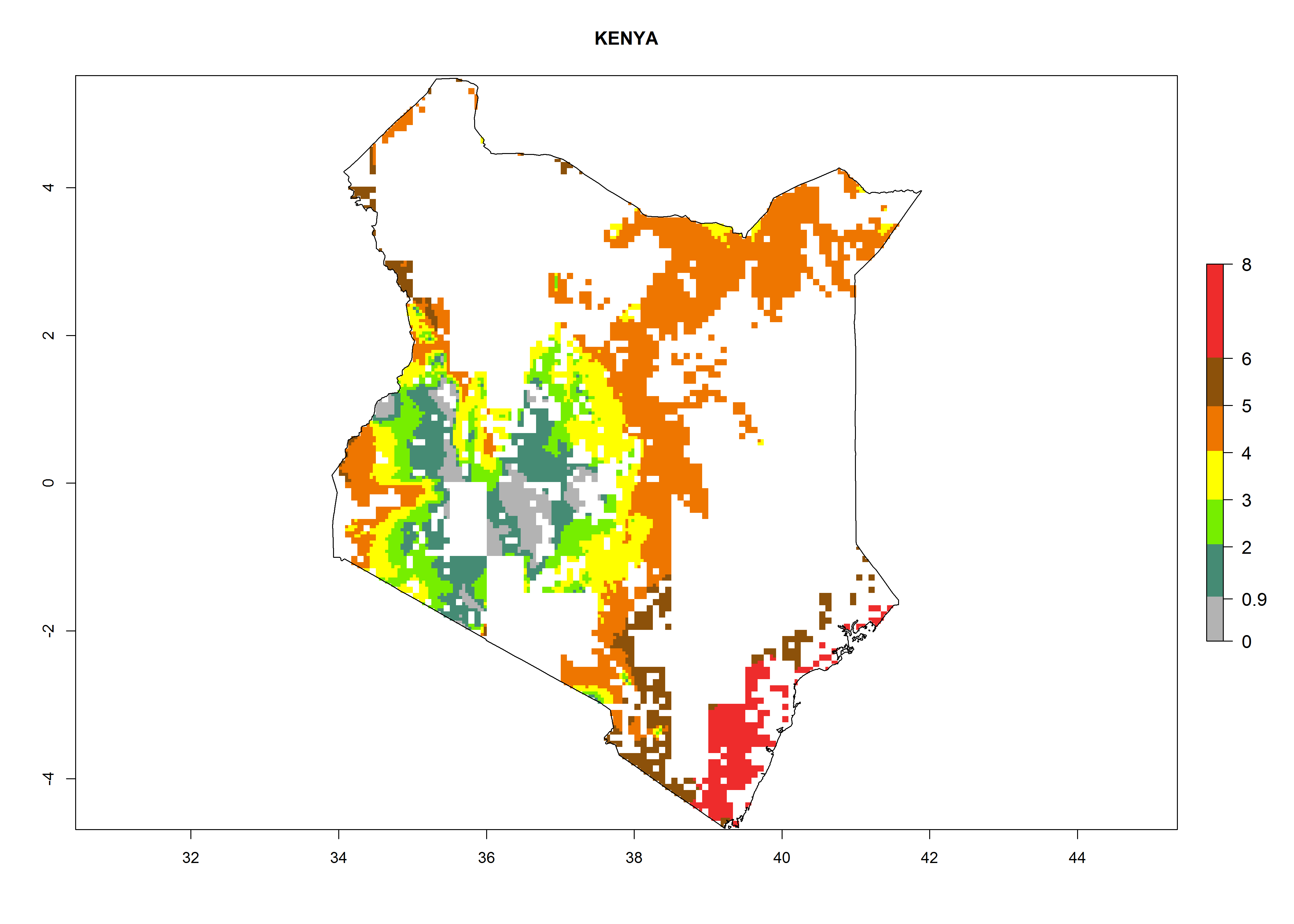
Establishment of B. fusca is possible in the main maize-growing zones (ERI>0.95) with 2–5 generations per year. The number of generations is projected to be low in high-altitude areas and is likely to increase in mid-altitude zones (GI>3). The annual population increase varies AI>0.9–9 (Fig. 6). In Uganda, an ERI>0.95 is indicated for the complete maize cultivation area, with 3–5 generations per year. The annual population growth potential varies AI>3–6 (Fig. 6). In Tanzania an ERI>0.95 is indicated for most of the maize cultivation areas except for the central part of the country and around Lake Victoria. The GI is projected to vary between 3 and 5 generations per year in most of the maize cultivation areas. The population growth potential is projected to vary AI>2–6 per year (Fig.6).
| ERI | GI | AI |
| a) Angola | 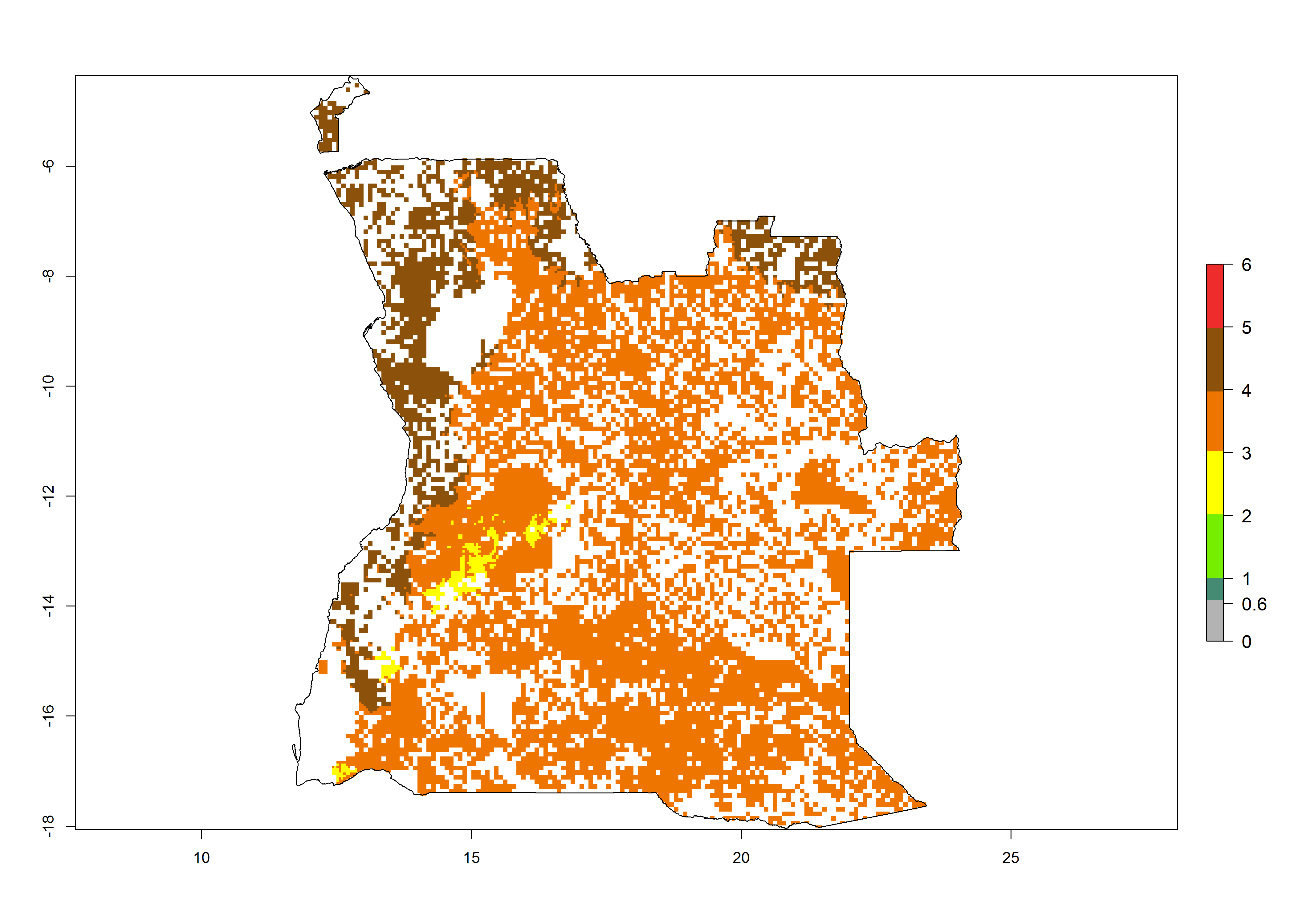 |
 |
| b) Burkina Faso | 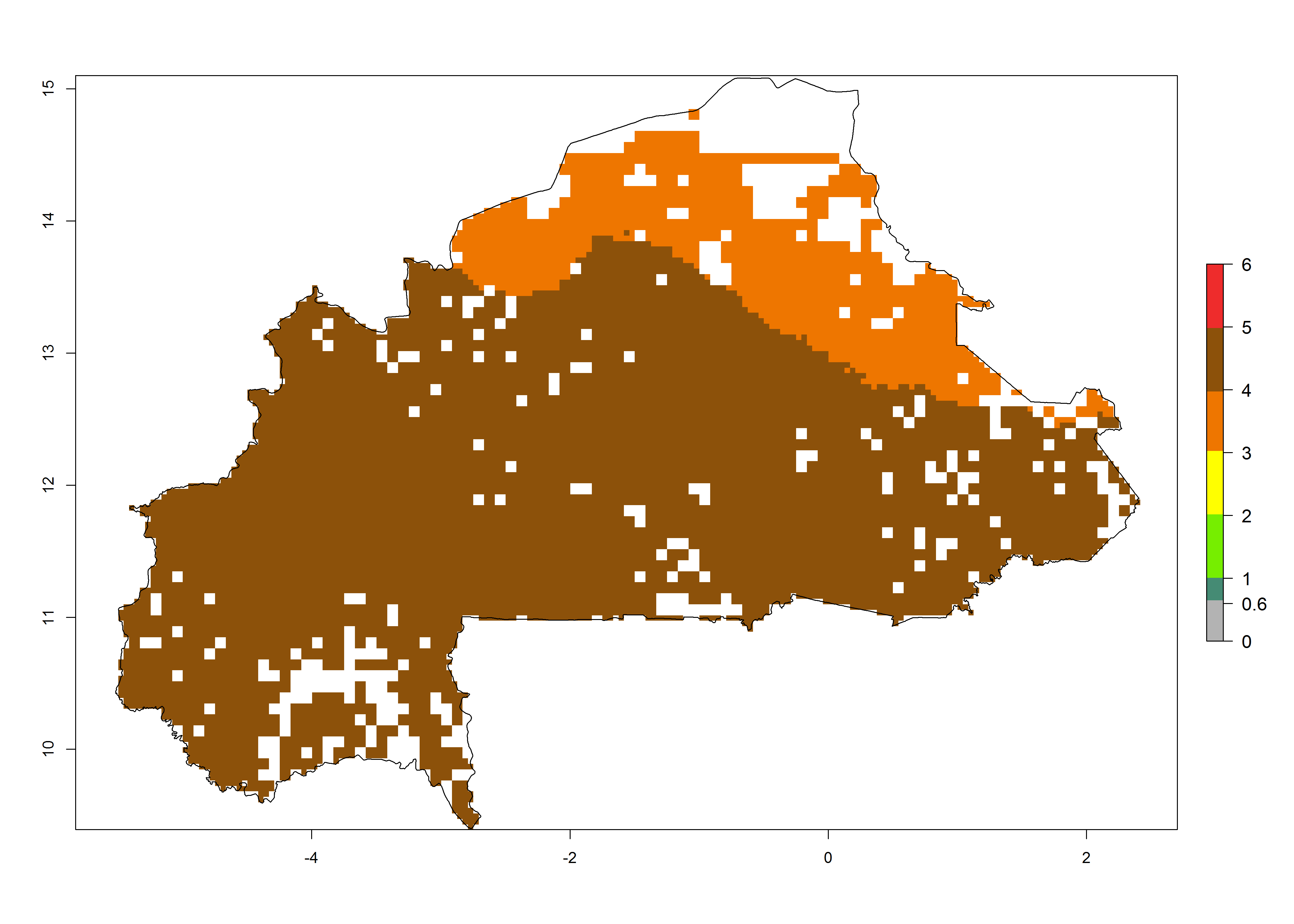 |
 |
| c) Burundi | 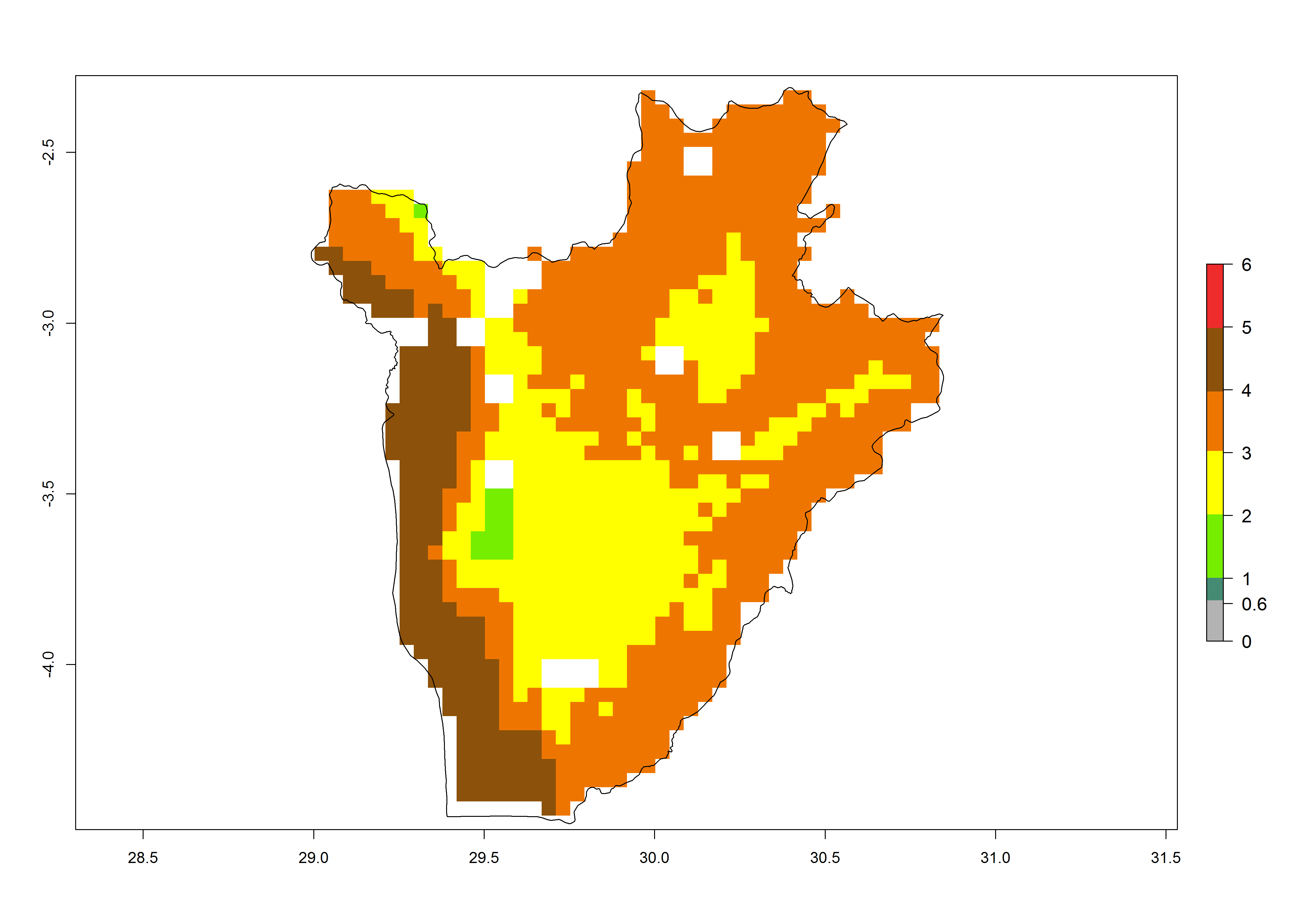 |
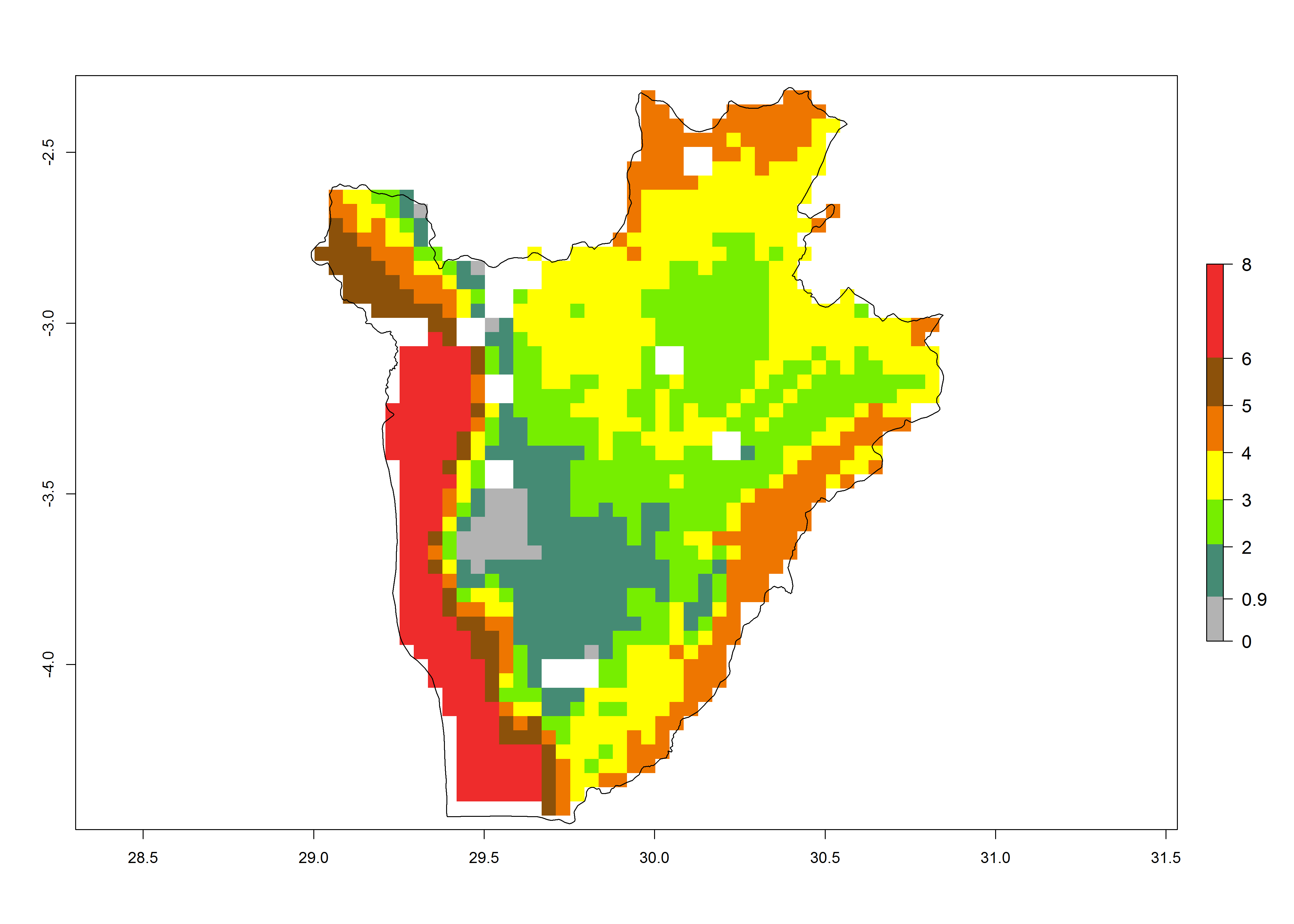 |
| d) DR Congo | 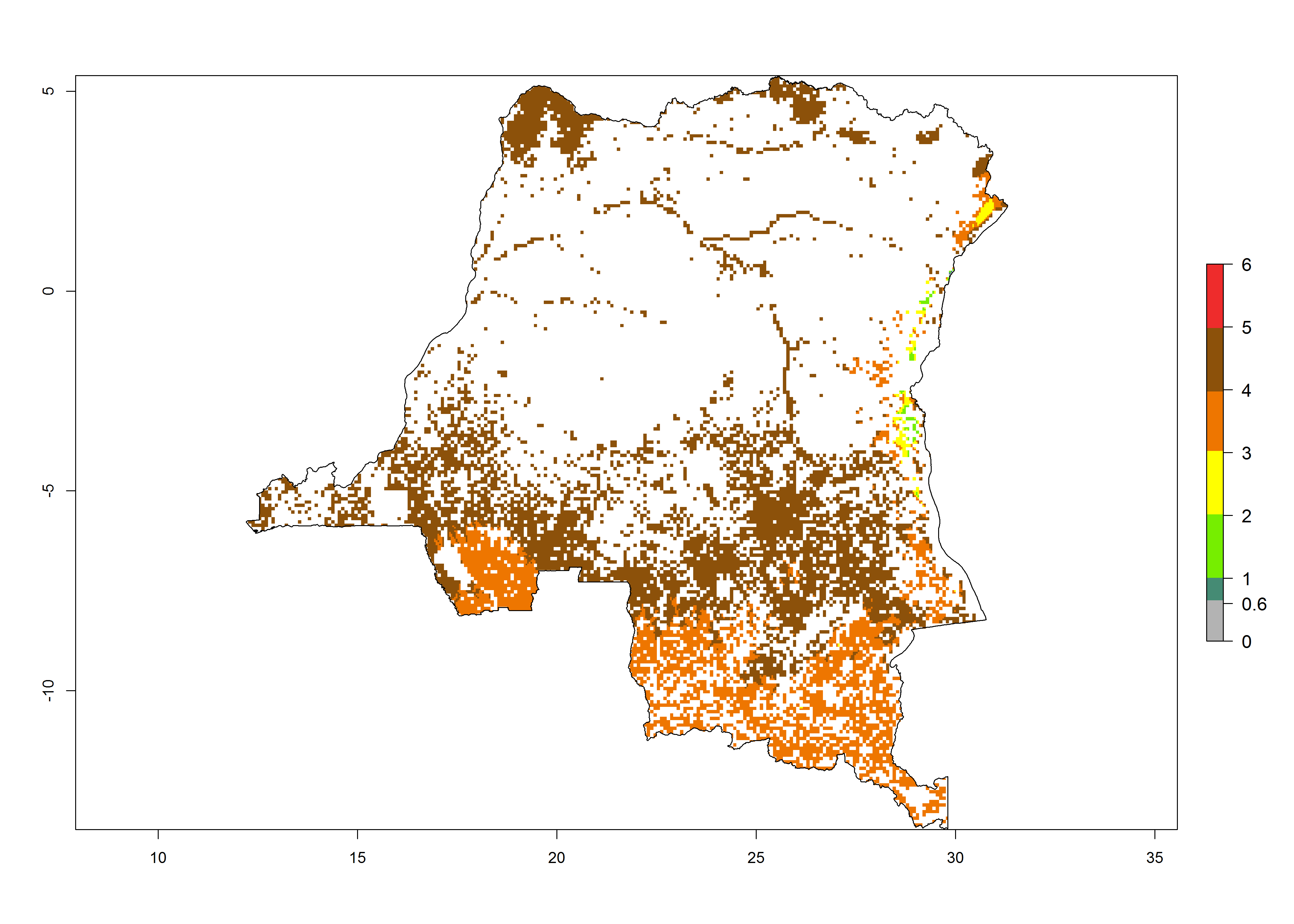 |
 |
| e) Ivory Coast |  |
 |
| f) Kenya | 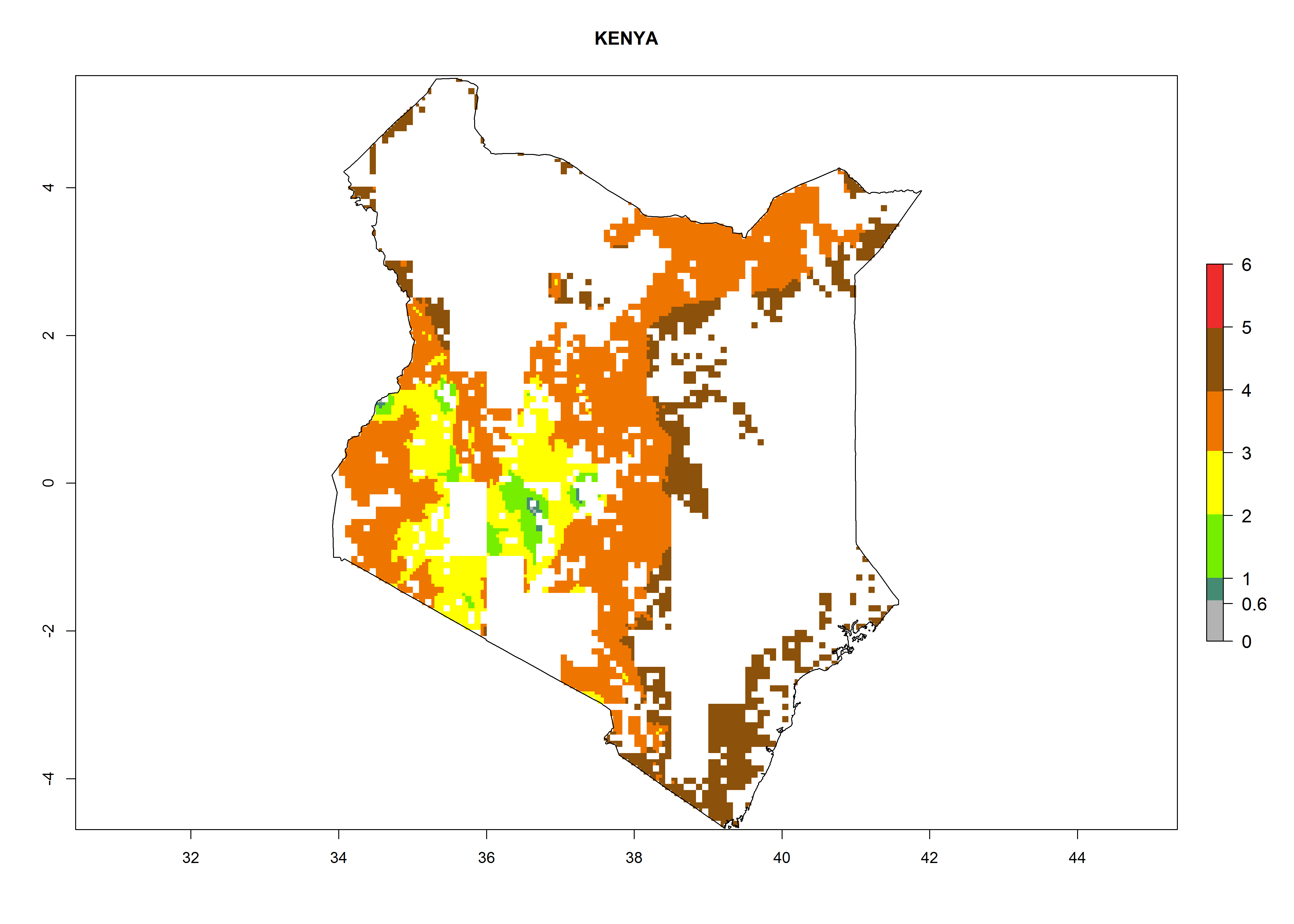 |
 |
| g) Madagascar | 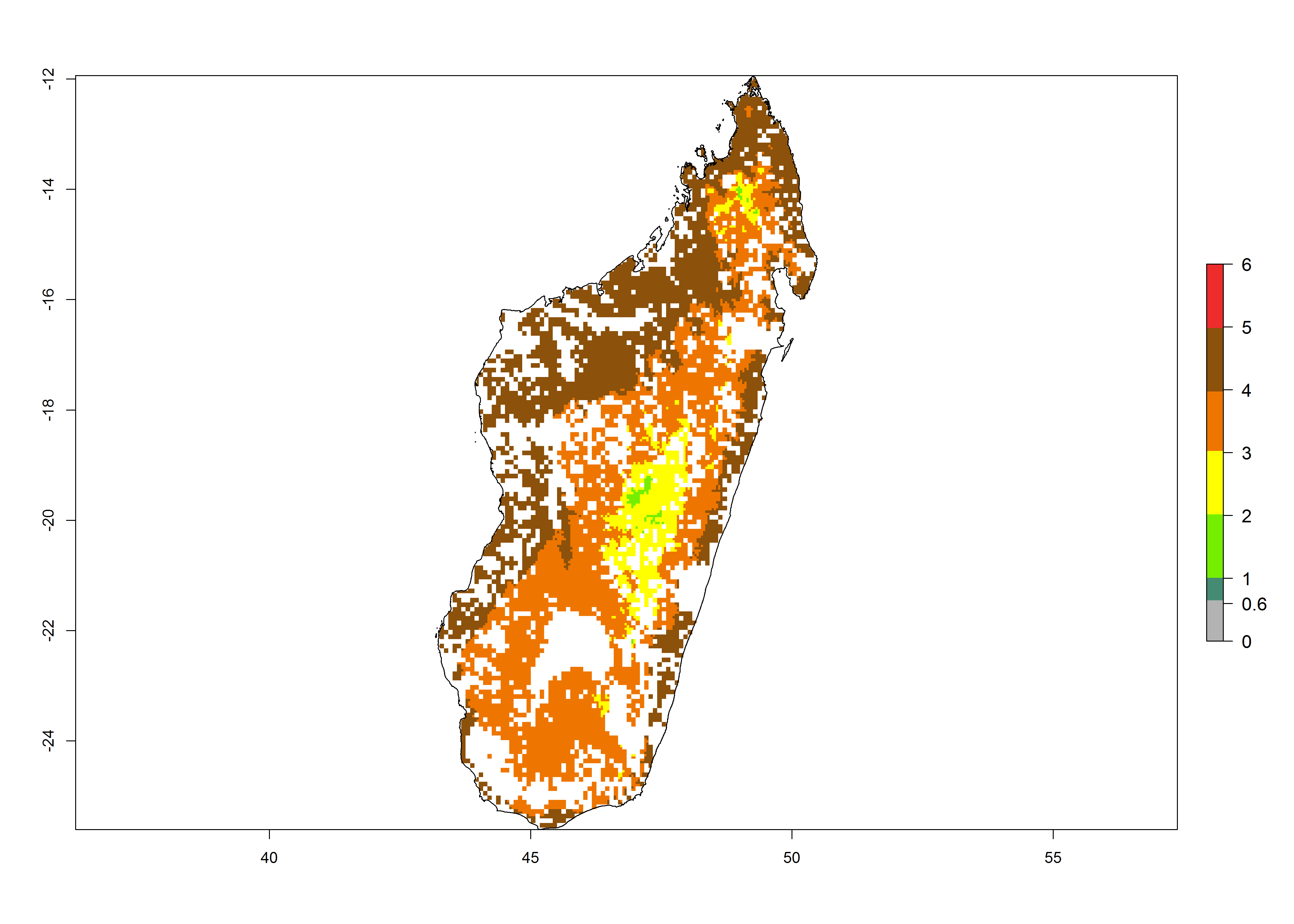 |
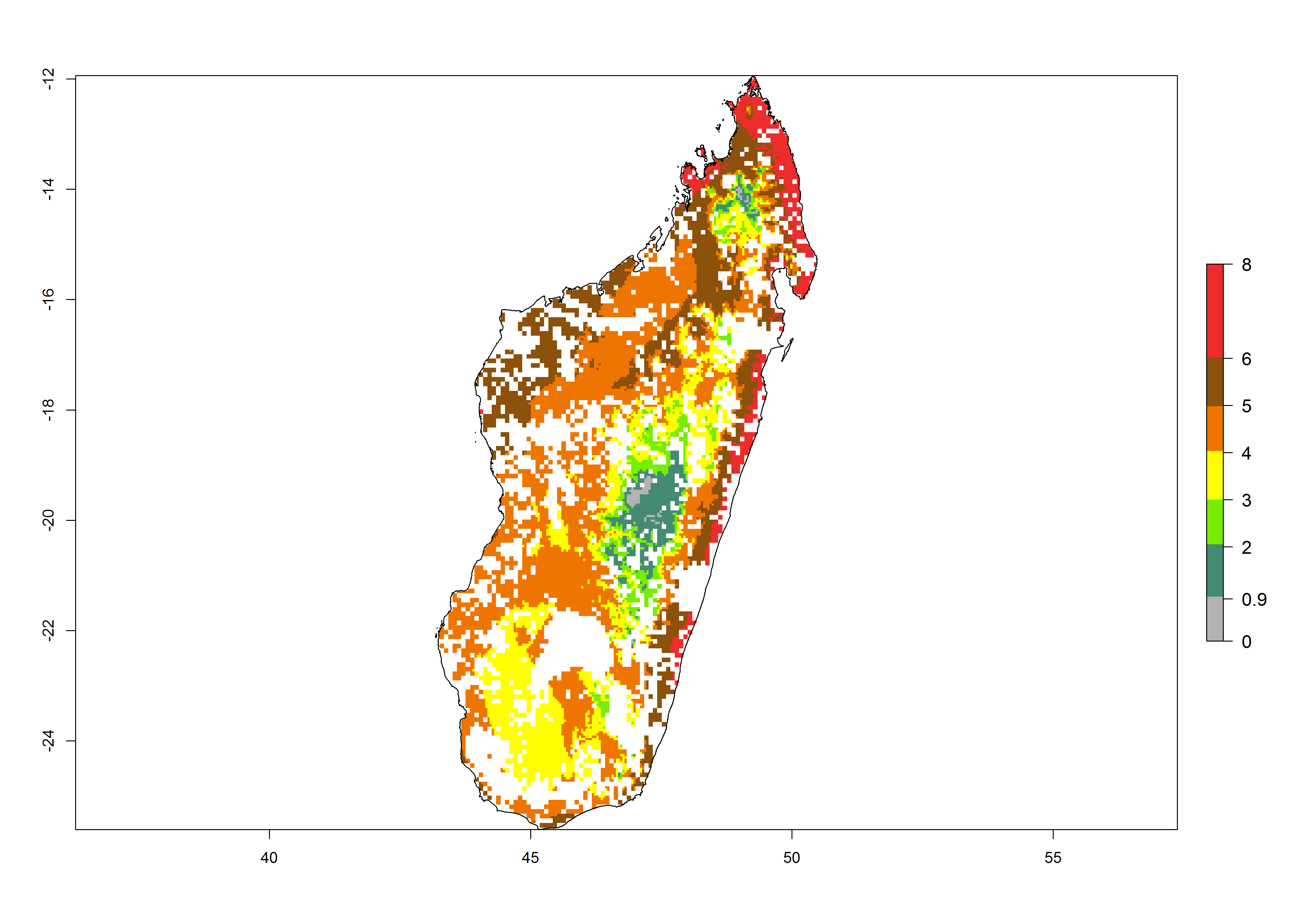 |
| h) Mozambique | 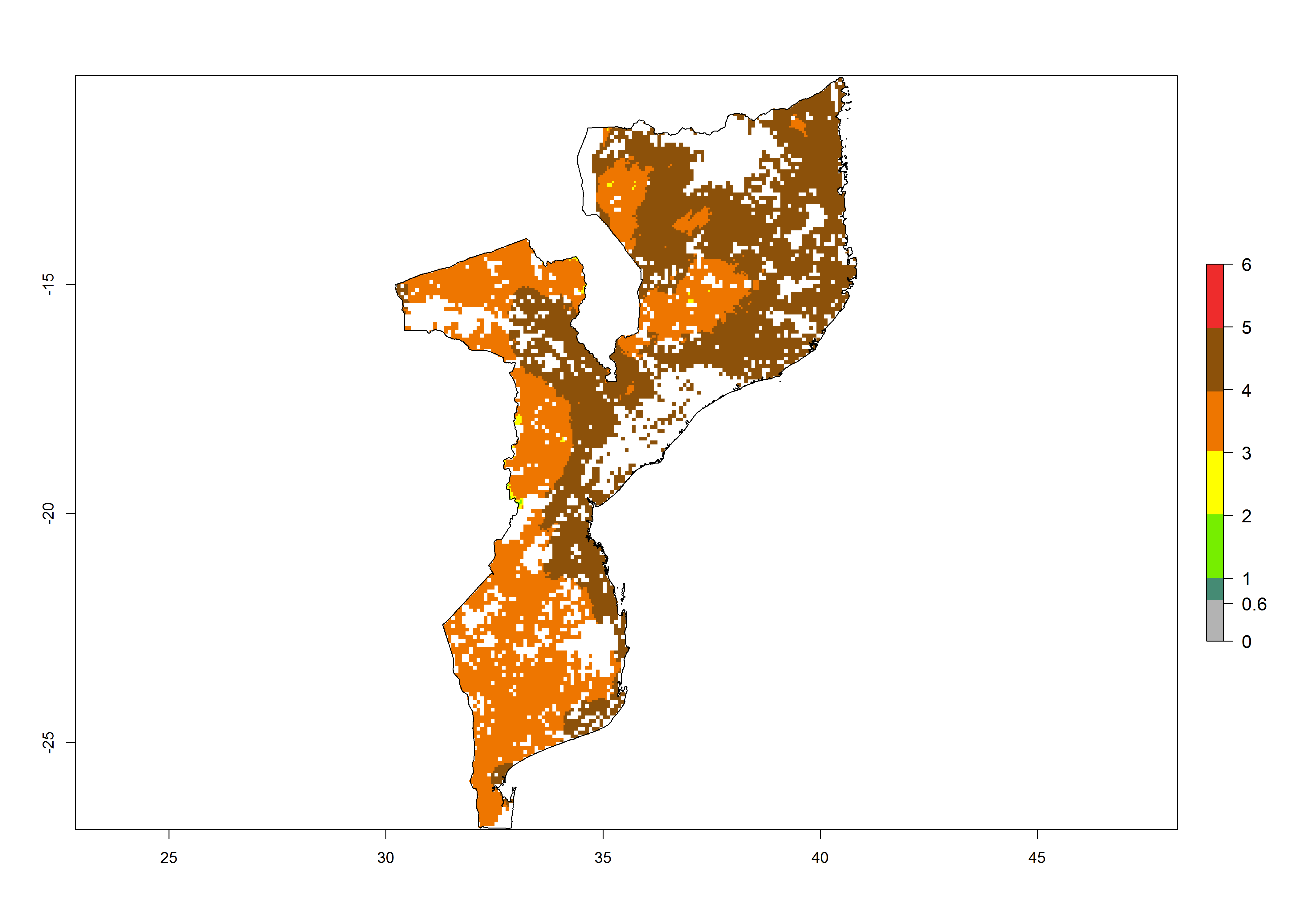 |
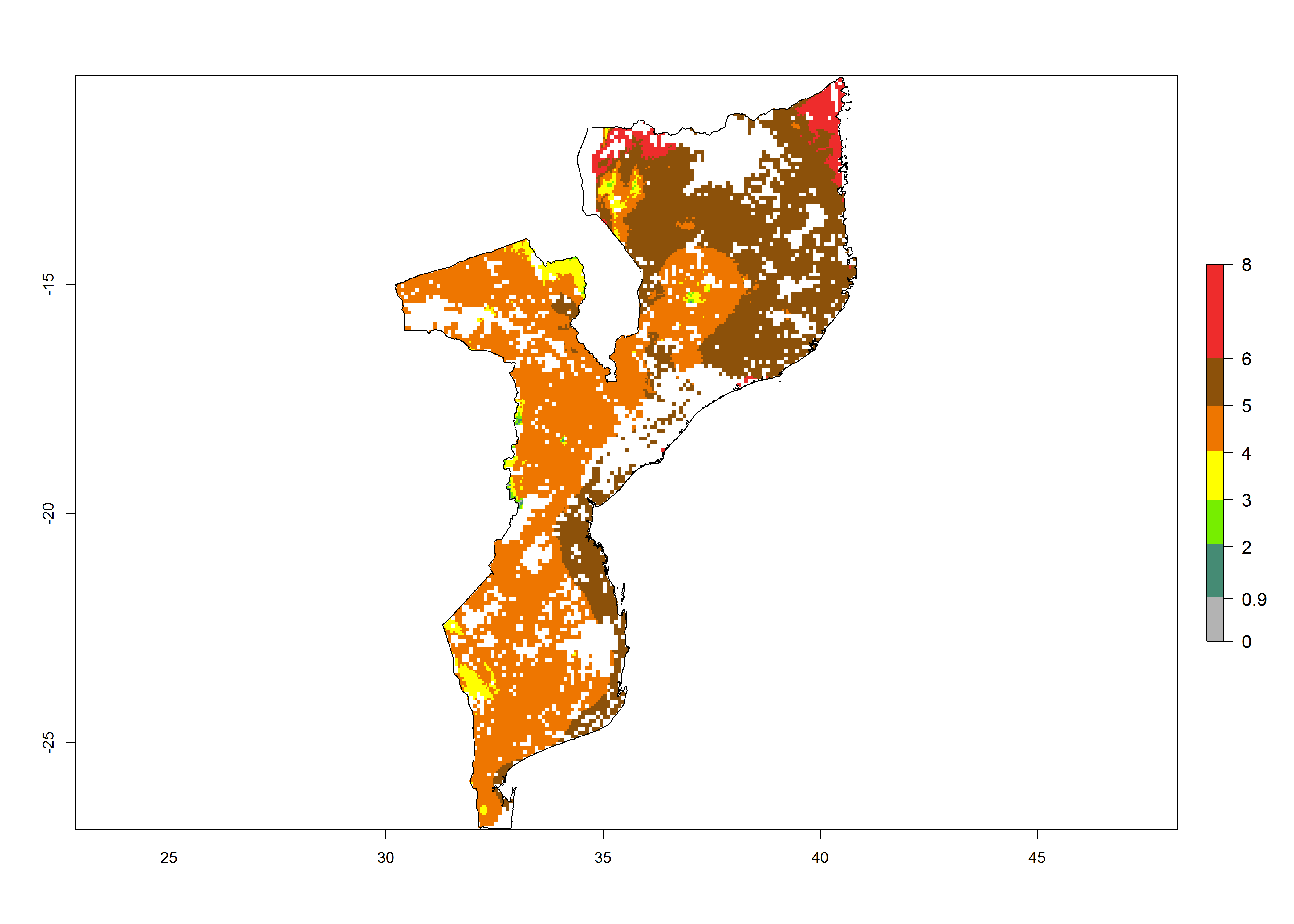 |
| i) Nigeria | 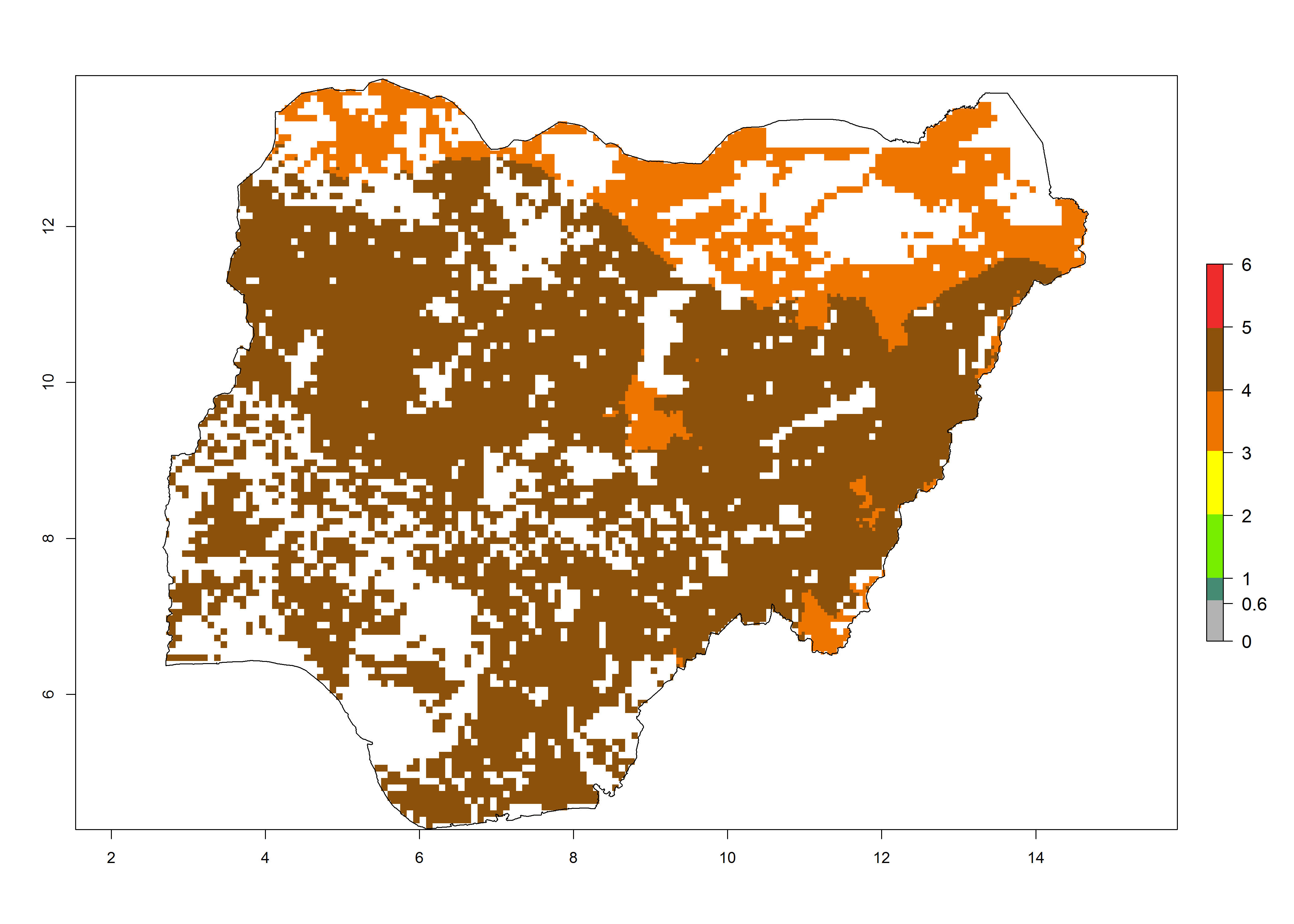 |
 |
| j) Rwanda | 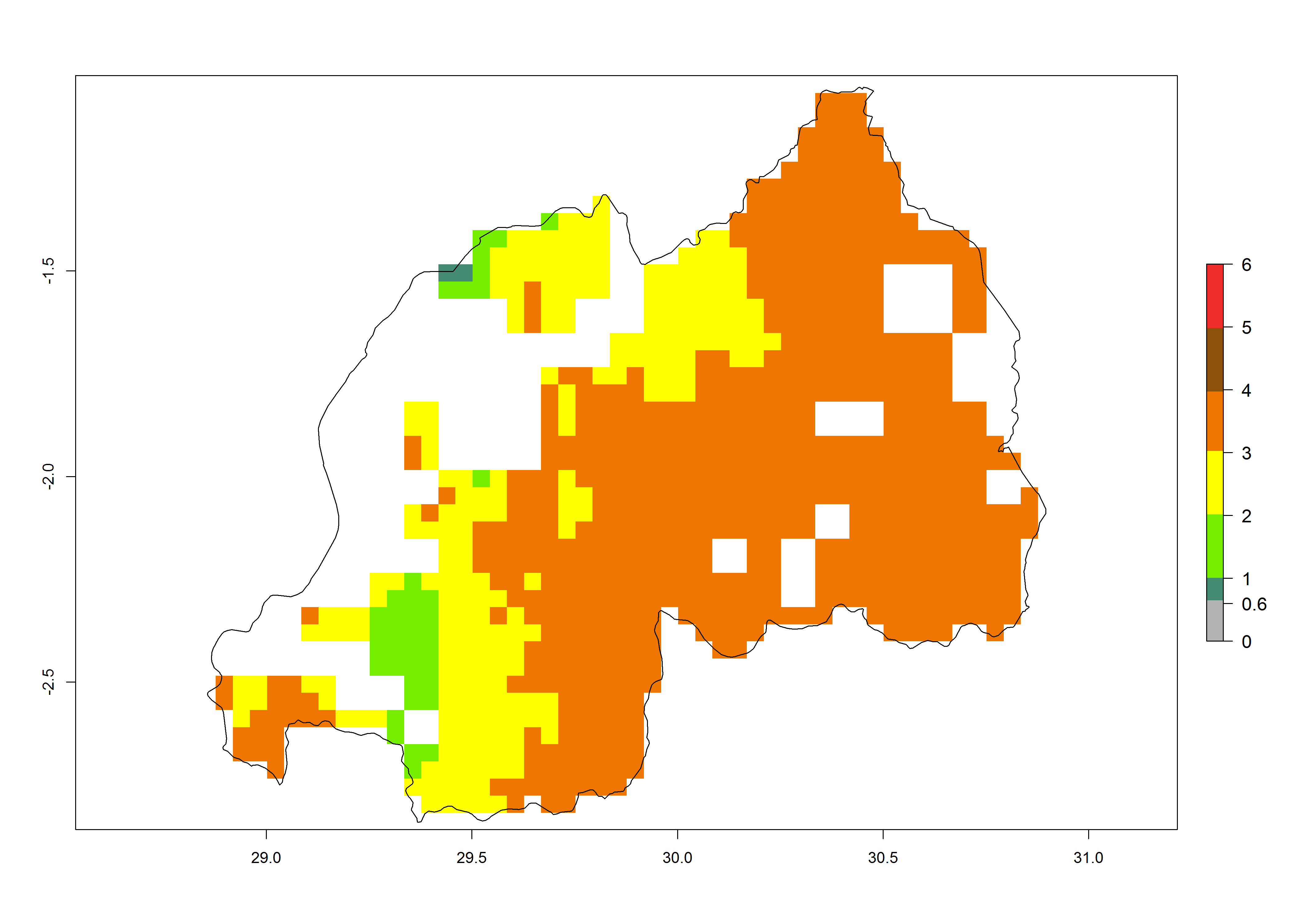 |
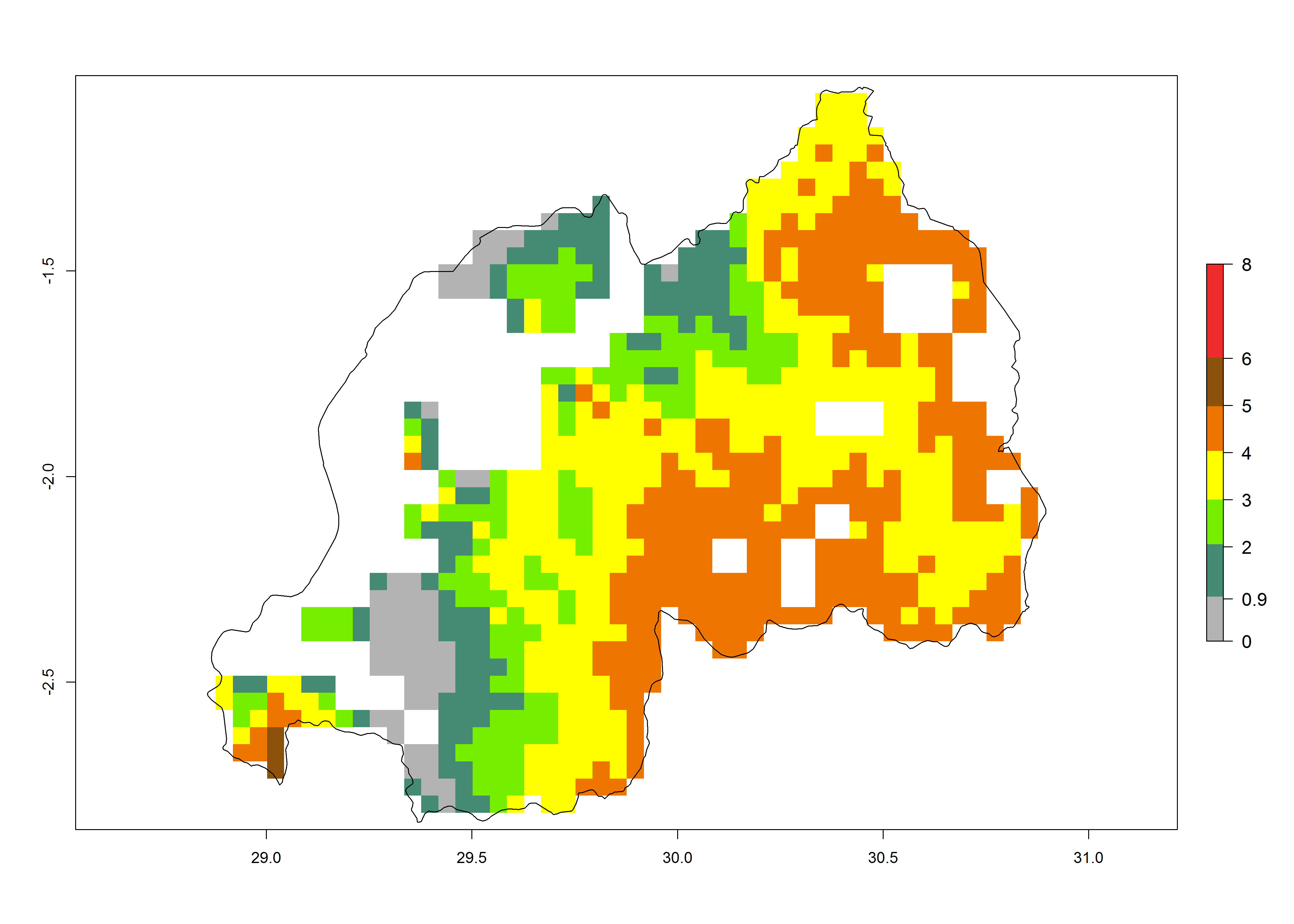 |
| k) Senegal |  |
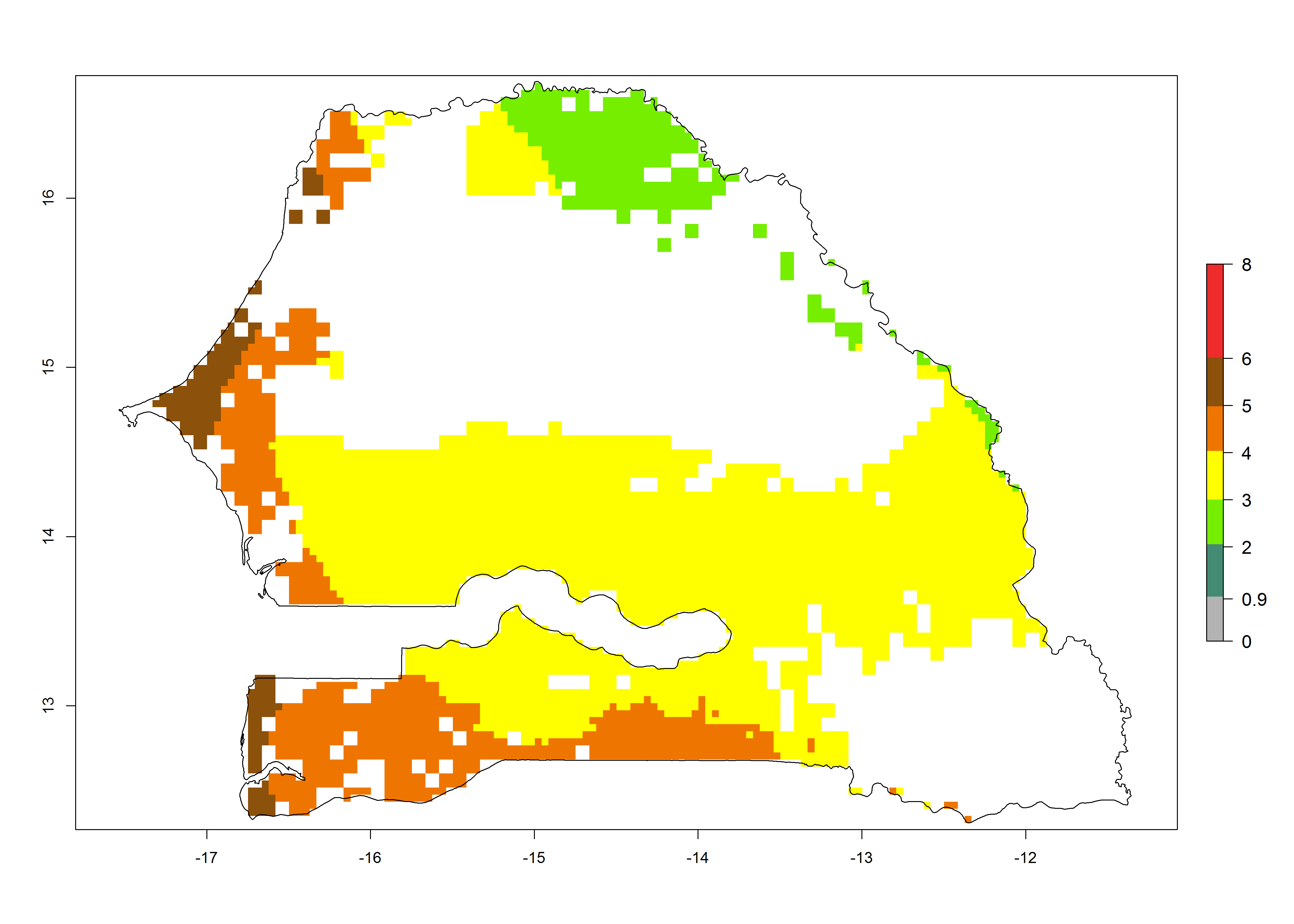 |
| l) South Africa |  |
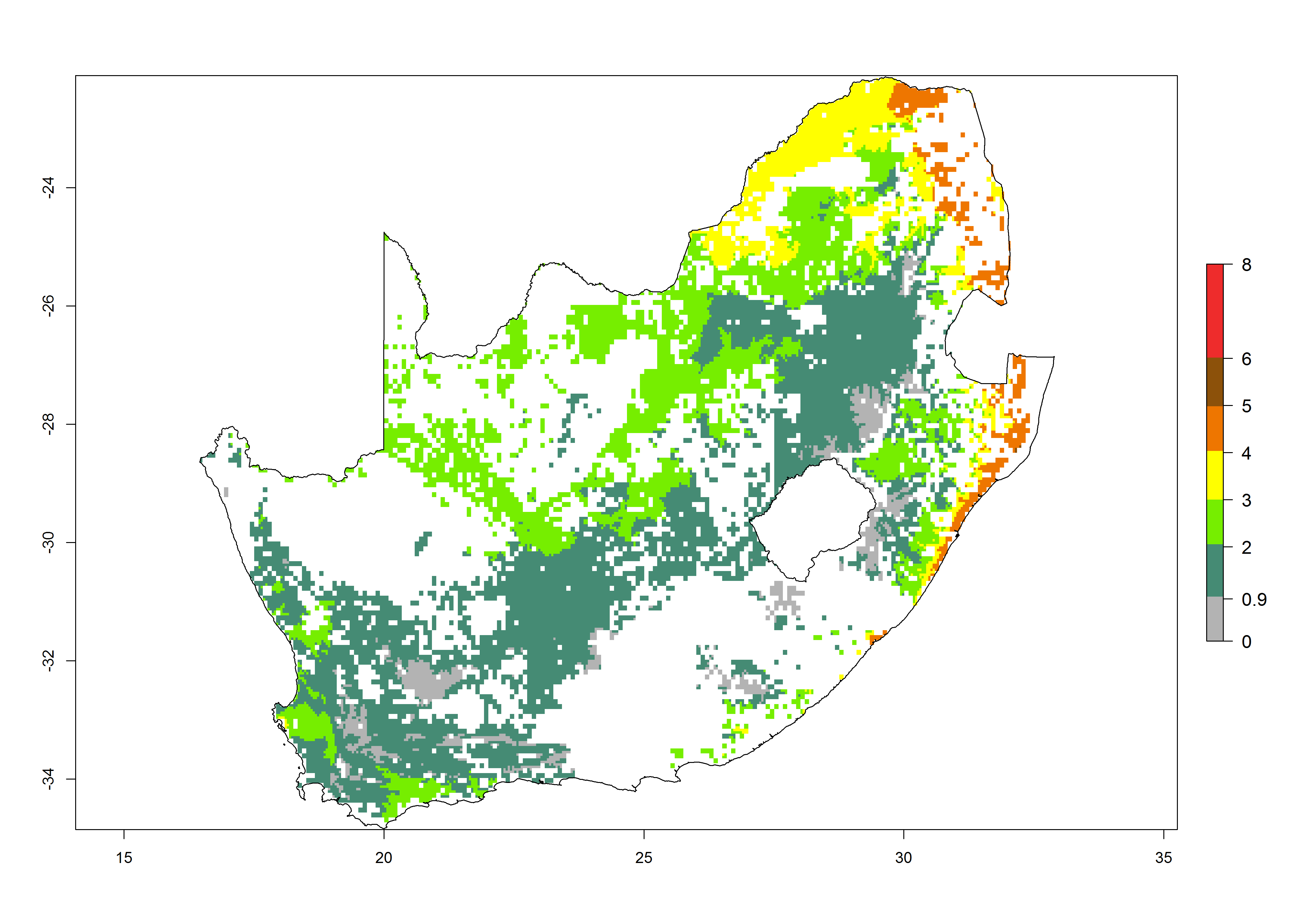 |
| m) Sudan | 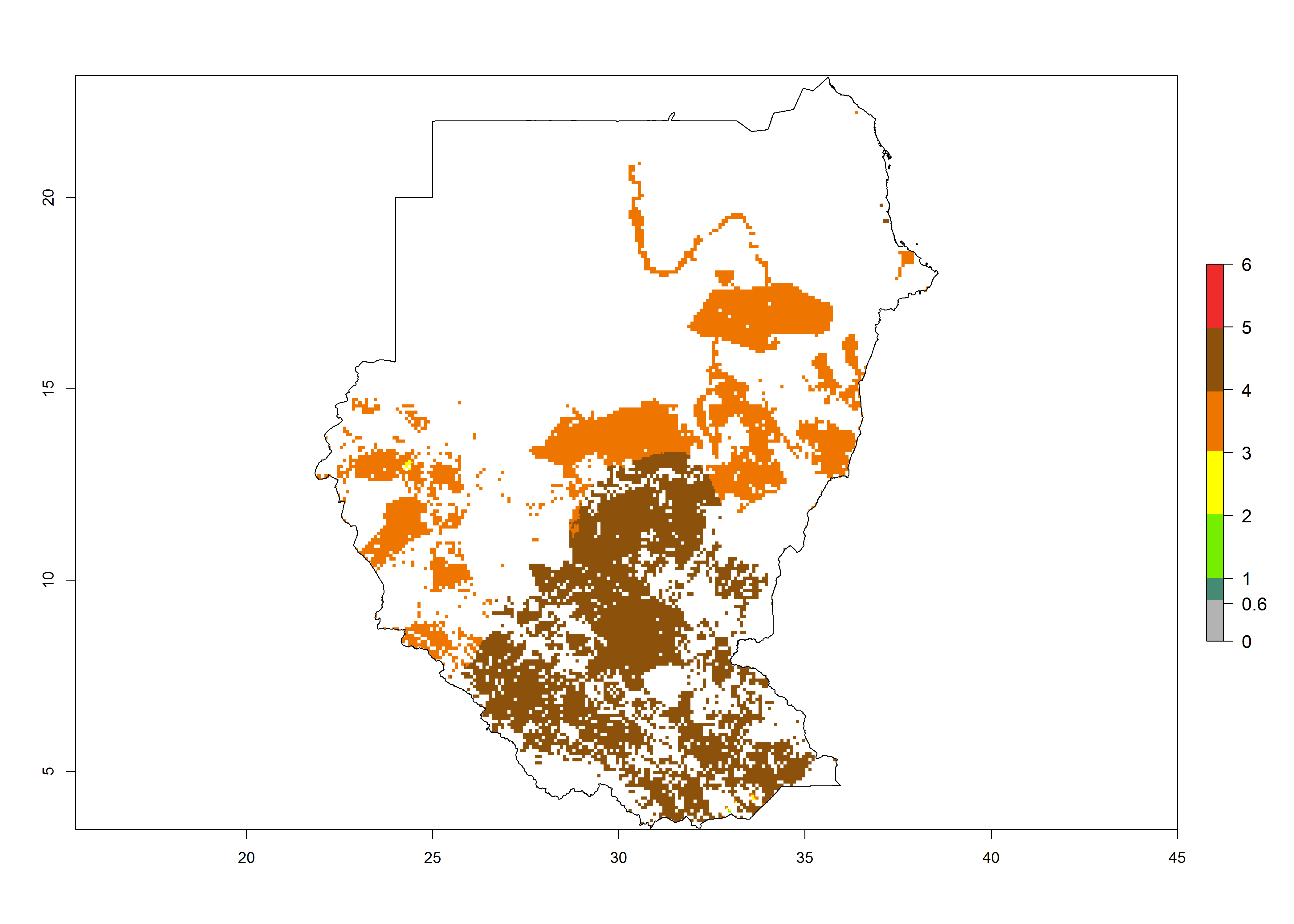 |
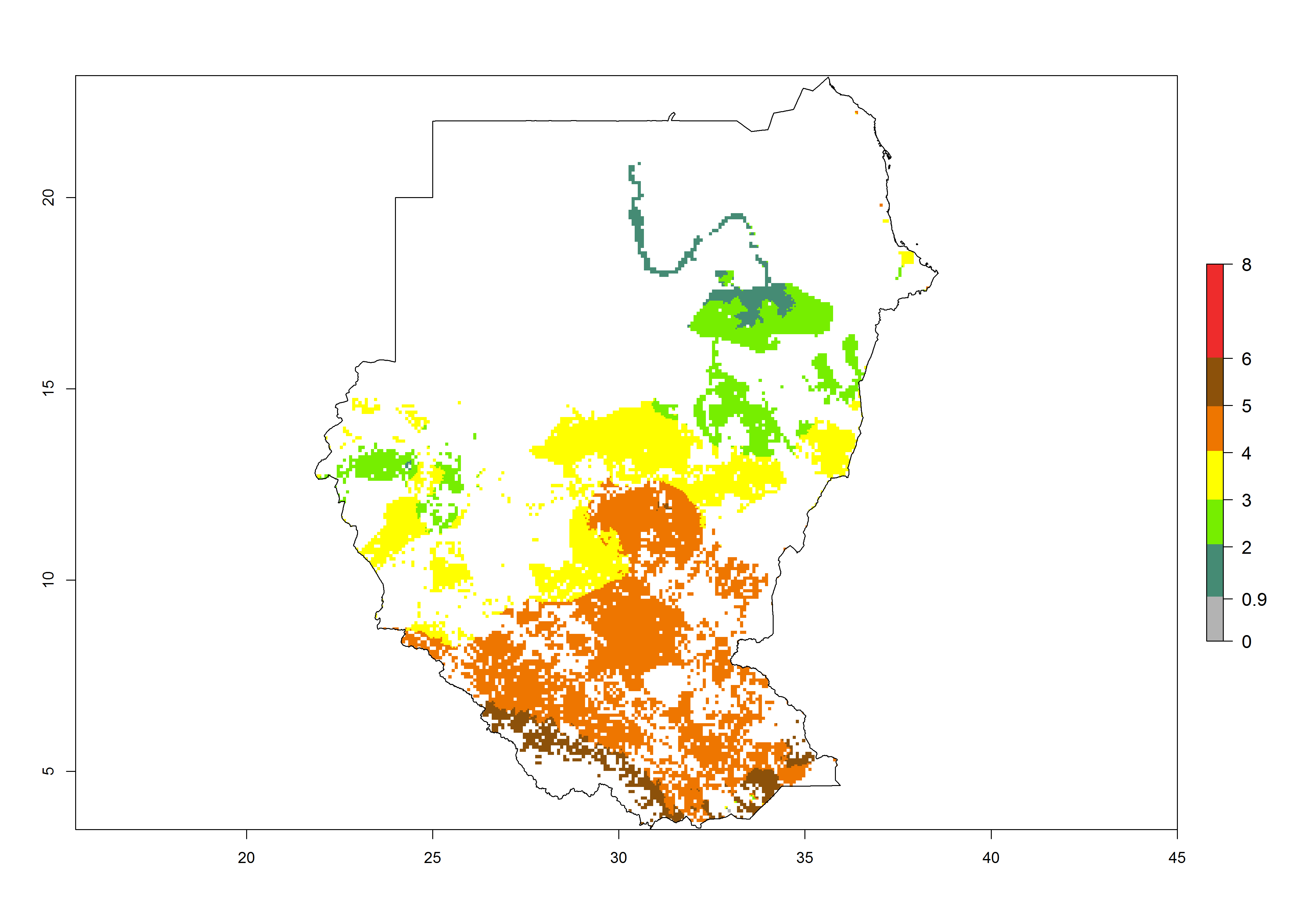 |
| n) Tanzania | 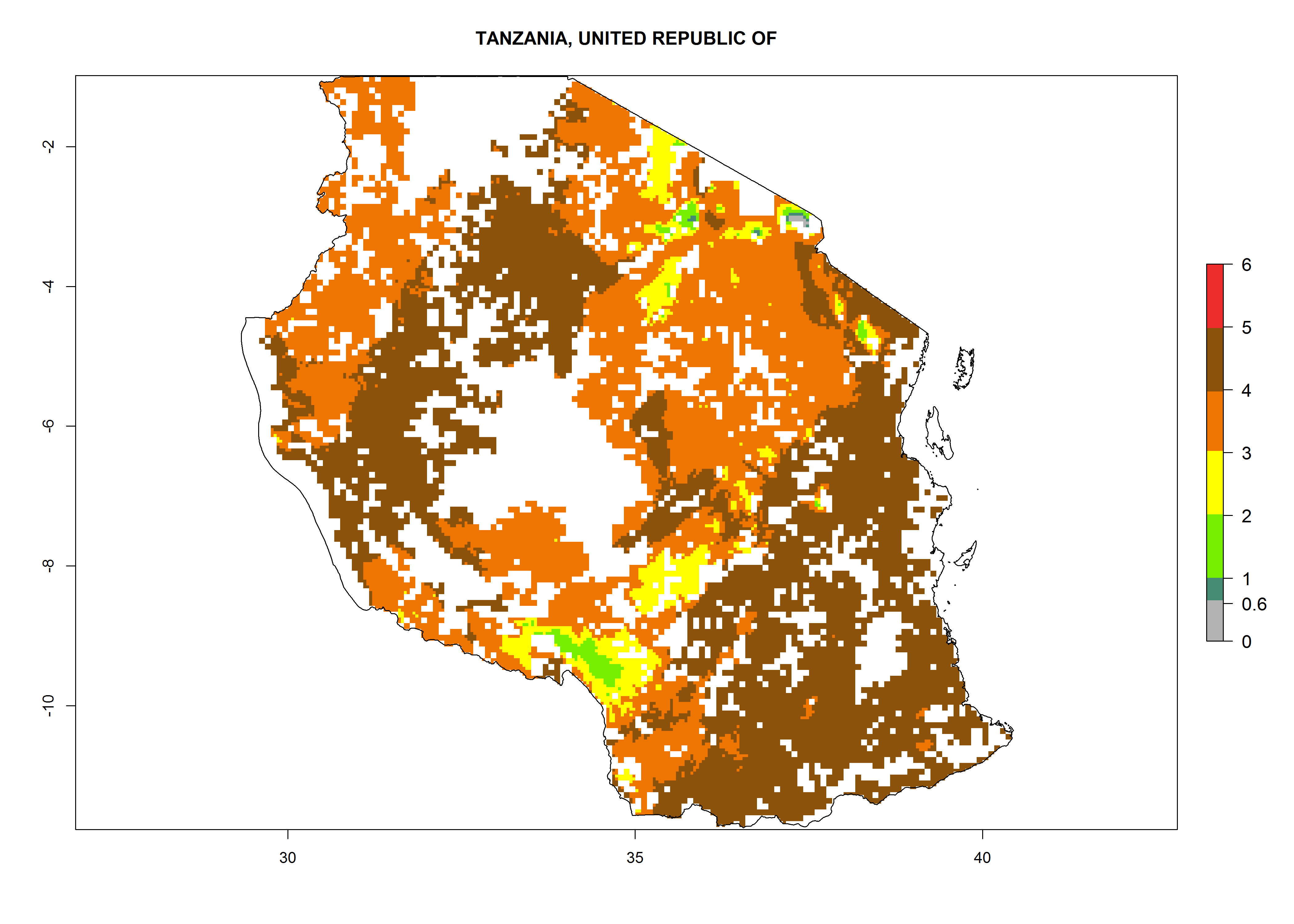 |
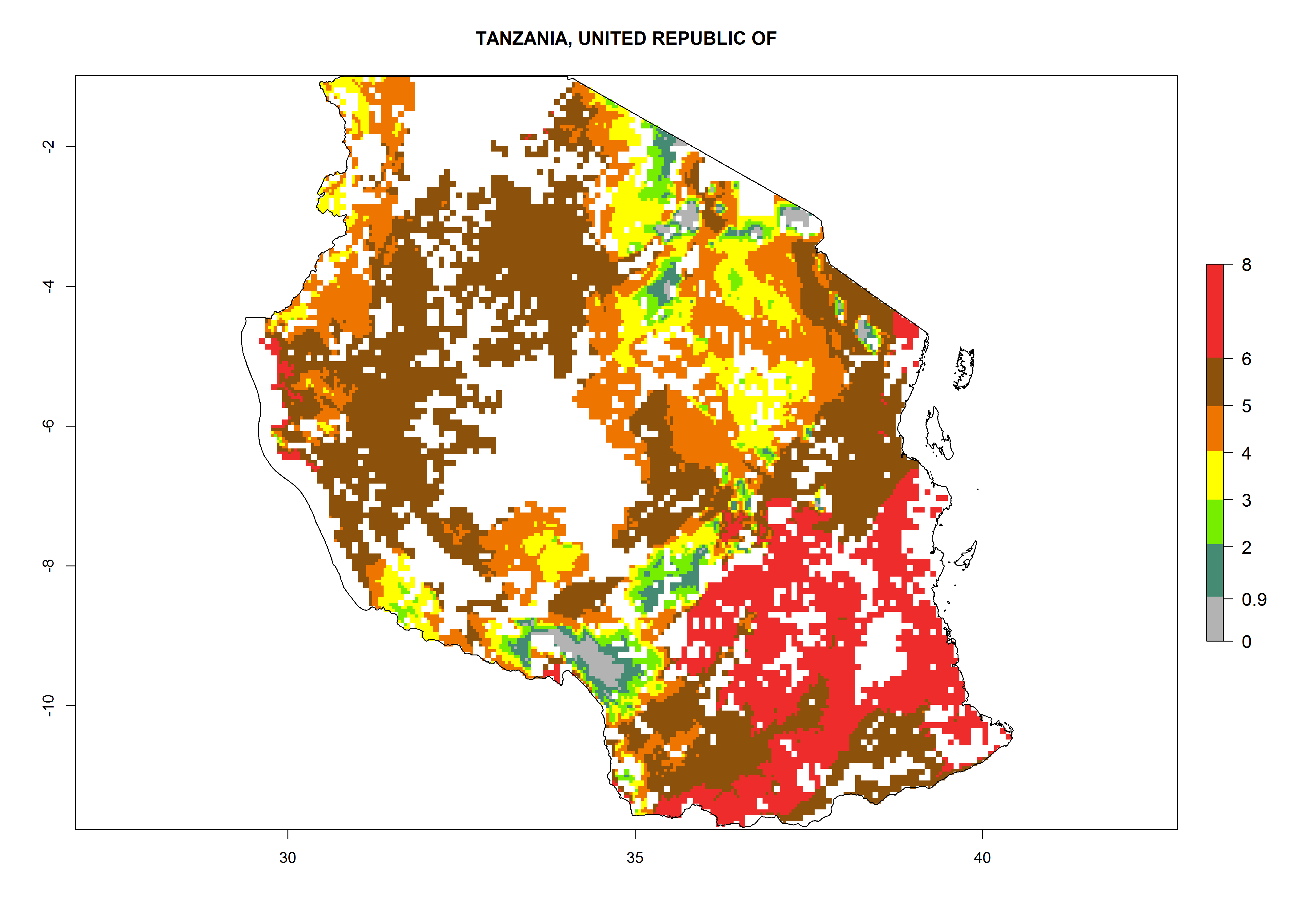 |
| o) Uganda |  |
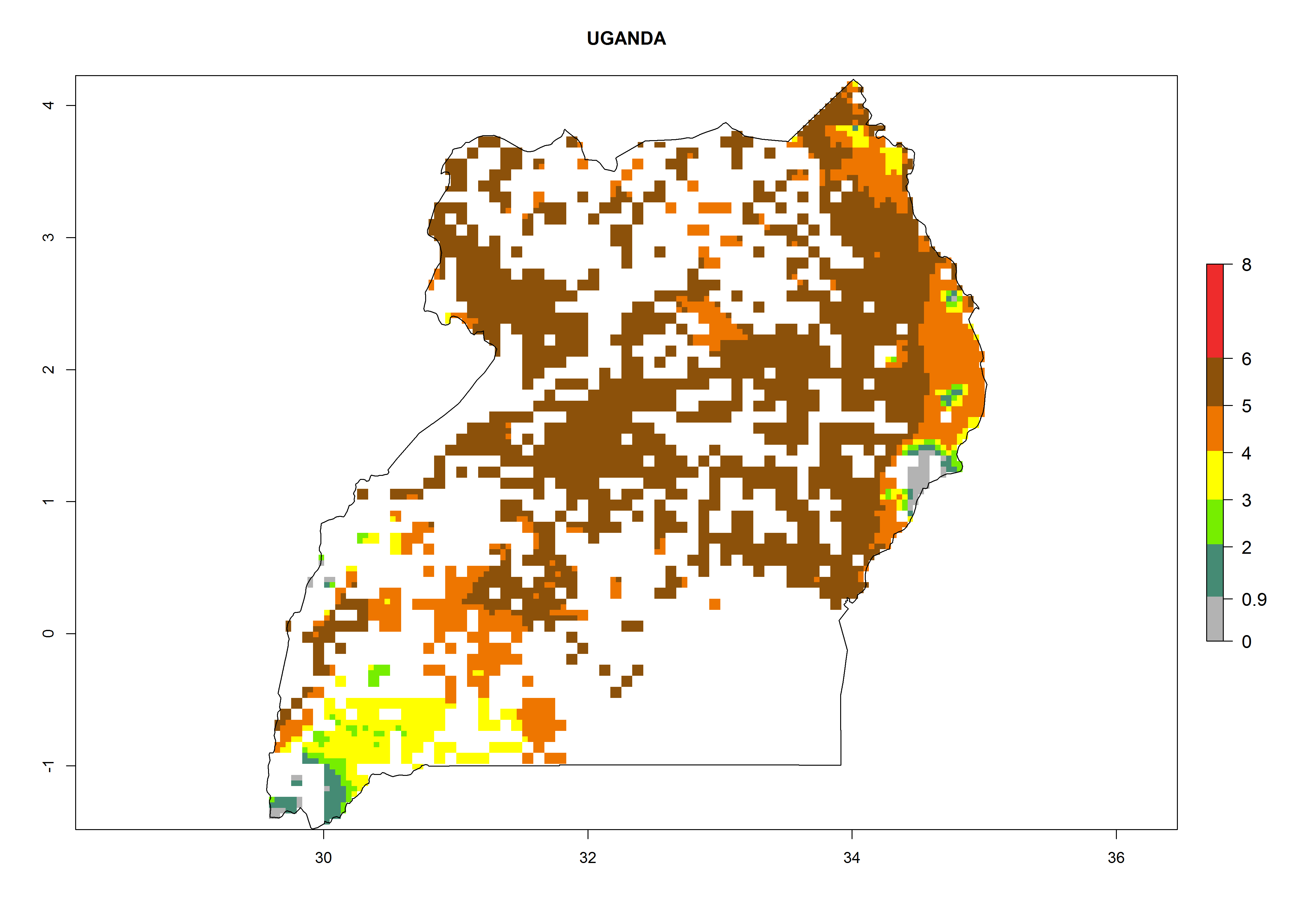 |
Figure 6. ERI, abundance (GI, potential damage), and activity (AI, potential population growth) of the maize stalk borer, Busseola fusca, in maize production systems of selected African countries according to model predictions for the year2000. An ERI>0.95 is associated with potential permanent establishment.
Phytosanitary measures
Complete burning of the stalks kills larval and pupal stages of the pest. Spreading out stalks thinly to dry can kill diapausing larvae.
Adaptation to risk avoidance at farm level
Monitoring pest population. African maize stalk borer infestations may be detected by walking through young crops looking for characteristic feeding marks on funnel leaves, the presence of dead hearts, and holes in tunneled stems. Fields must be inspected twice a week. Samples of affected stems are dissected to find caterpillars and pupae, which are best reared to the adult stage to identify the correct species as this is very difficult in immatures stages. In older plants caterpillars bore into stems and cobs. An action threshold of 10% infested plants has been recommended.
Cultural practices. Intercropping maize with non-hosts crops like cassava (Manihot esculenta Crantz) or legumes like cowpea (Vigna unguiculata L. Walp.) can reduce African maize stalk borer damage. Alternatively, maize can be intercropped with a repellent plant such as silver leaf desmodium [Desmodium uncinatum (Jacq.) DC.] and a trap plant, such as Napier grass (Pennisetum purpureum Schumach.) and molasses grass (Melinis minutiflora P. Beauv.) as a border crop to protect maize from stemborers. The trap plant attracts the adult female. More eggs are laid on the trap plant than on the crop, but the larvae develop poorly or not at all on the trap plant. This practice is known as “push-pull.” Cultural practices such as deep ploughing or tillage, use of crop residues for fodder and silage, harrowing, crop rotation, changes in sowing/planting dates, removal of dead hearts, removal of alternative host plants and volunteer crop plants, water management, fertilizer management, and intercropping are useful for reducing pest abundance and infestation. Early slashing of maize stubble and laying it out on the ground where the sun’s heat destroys the larvae and pupae within can also be done.
Biological control. Many natural enemies of the African maize stalk borer have been reported, but their impact is variable across regions, seasons, and crops. Two of the most abundant natural enemies of the African maize stalk borer are the larval parasitoids Cotesia sesamiae Cameron and Bracon sesamiae Cameron. They locate the stemborers inside of plant stems and lay eggs into them. Upon hatching, the larvae of the parasitic wasp feed internally in the stemborer, which cause it to die. Afterward, they exit to spin cocoons. The ability of indigenous natural enemies to control B. fusca is too low to prevent significant damage to maize. However, they help control to reduce pest densities as part of an integrated pest management approach that includes habitat management practices that conserve parasitoids and predators like ants and earwigs.
Chemical control. Chemical control can be achieved by applications of granules or dust into the leaf whorl at early crop growth to kill early larval instars. This method has limited effectiveness once the larvae bore into the stem. Neem (Azadirachta indica A. Juss.) products (powder from ground neem seeds) are reported to be effective and may be applied to the leaf whorl in a 1:1 mixture with dry clay or sawdust; 1 kg of powder should be sufficient to treat 1,500–2,000 plants. Rainwater dissolves the active substances in neem powder as it gathers in the funnel and washes out the powder. Where rainfall is irregular, a liquid neem seed extract can be sprayed into the funnel. Pesticides are poisons, so it is essential to follow all safety precautions on labels.
Physical control. Though still under study and development, the use of traps (pheromone t traps), baits, and mating disruption may be used in future.
Legislative control. In some areas, restricting maize cultivation to certain vegetation periods is exerted to lower infestation.
Further reading
CABI Crop Protection Compendium: http://www.cabi.org/cpc/ Accessed on 20/11/2013.
Chabi-Olaye, A., F. Schulthess, and C. Borgemeister. 2008. Effects of nitrogen and potassium combinations on yields and infestations of maize by Busseola fusca (Lepidoptera: Noctuidae) in the Humid Forest of Cameroon. Journal of Economic Entomology 101(1): 90–98.
Chabi-Olaye, A., C. Nolte, F. Schulthess, and C. Borgemeister. 2005a. Effects of grain legumes and cover crops on maize yield and plant damage by Busseola fusca (Lepidoptera: Noctuidae) in the humid forest of southern Cameroon. Agriculture, Ecosystems & Environment 108: 17–28.
———. 2005b. Relationships of intercropped maize, stem borer damage to maize yield and land-use efficiency in the humid forest of Cameroon. Bulletin of Entomological Research 95: 417–427.
———. 2006. Relationships of soil fertility and stem borers damage to yield in maize-based cropping system in Cameroon. Ann. Soc. Entomol. Fr. 42: 471–479.
Dryer, M. 1987. Field and laboratory trials with simple neem products as protectants against pests of vegetables and field crops in Togo. Proc. 3rd International Neem Conference (Nairobi, Kenya, 1986). pp. 431–447.
Khadioli, N., Z.E.H Tonnang, G.O. Ong’amo, T. Achia, I. Kipchirchir, J. Kroschel, and B. Le Ru. 2014. Effect of temperature on the life history parameters of noctuid lepidopteran stem borers, Busseola fusca (Fuller) and Sesamia calamistis Hampson. Annals of Applied Biology 165(3): 373–386.
Khan, Z.R., F.N. Muyekho, E Njuguna, J.A. Pickett, L.J. Wadhams, N.Dibogo, A. Ndiege, G.Genga, and C. Luswetti. 2005. A primer on planting and managing ‘push-pull’ fields for stemborer and striga control in maize – a step-by-step guide for farmers. International Centre of Insect Physiology and Ecology (ICIPE). ICIPE Biennial Scientific Report 2004–2005. Biocontrol cereal pests in Africa. Pdf. www.icipe.org
Le Ru, B.P., G.O. Ong’amo, P. Moyal, L. Ngala, B. Musyoka, Z. Abdullah, D. Cugala, B. Defabachew, T.A. Haile, T. Kauma Matama, et al. 2006. Diversity of lepidopteran stem borers on monocotyledonous plants in eastern Africa and the islands of Madagascar and Zanzibar revisited. Bulletin of Entomological Research 96: 555–563.
Lusweti, A., E. Wabuyele, P. Ssegawa, and J.R. Mauremootoo. 2011. Invasive plants of East Africa (Kenya, Uganda and Tanzania), Lucid v. 3.5 key and fact sheets. National Museums of Kenya, Makerere University, BioNET-EAFRINET, CABI & The University of Queensland (September 2011) keys.lucidcentral.org/keys/v3/EAFRINET. http://keys.lucidcentral.org/keys/v3/eafrinet/maize_pests/key/maize_pests/Media/Html/Busseola_fusca_(Fuller_1901)_-_African_Maize_Stalkborer.htm. Accessed on 20/11/2013.
Mally, C.W. 1920. The maize stalk borer, Busseola fusca Fuller. Bulletin, Department of Agriculture, Union of South Africa, No. 3. 111pp.
Mathee, J.J. 1974. Pests of graminaceous crops in South Africa. Entomology Memoir 40, Department of Agricultural Technical Services, Republic of South Africa 24pp.
Ong’amo, G.O., B.P. Le Ru, S. Dupas, P. Moyal, P.A. Calatayud, and J.F. Silvain. 2006. Distribution, pest status and agro-climatic preferences of lepidopteran stem borers of maize in Kenya. Annales de la Société Entomologique de France 42: 171–177. doi:10.1080/ 00379271.2006.10700620.
Walker, P.T. 1960. The relation between infestation by the stalk borer Busseola fusca and yield of maize in East Africa. Annals of Applied Biology 48(4): 780–786.
Walker, P.T., and M.J. Hodson. 1976. Developments in maize stem borer control in East Africa, including the use of insecticide granules. Annals of Applied Biology 84: 111–114.