4.4.3 Maize Pests / African pink stemborer, Sesamia calamistis (Hampson 1910)![]()
Synonyms: Sesamia mediastriga (Bethune-Baker 1911)
Taxonomic position: Insecta, Lepidoptera, Noctuidae (Apameini)
Authors: G. Ong’amo, N. Khadioli, B. Le Ru, N. Mujica, & P. Carhuapoma
Common names
African pink stemborer, Pink stalk borer, Pink stalk borer of sugarcane, African pink borer of sugarcane, Mauritius pink borer of sugarcane, southern pink borer of sugarcane (English), sésamie du maïs (French), afrikanischer Stengelbohrer (German).
Hosts
Sesamia calamistis is a polyphagous species found on about 55 host plants belonging to the families Poaceae (Andropogoneae, Arundineae, Cynodonteae, Eragrostideae, Orizeae, Paniceae) and Cyperaceae. Primary host plants are maize (Zea mays L.), sorghum [Sorgum bicolor (L.) Moench], pearl millet [Pennisetum glaucum (L.) R. Br.], rice (Oriza sativa L.), sugarcane (Saccharum officinarum L.), and wheat (Triticum aestivum L.). Wild hosts include many species of wild grasses such as Cymbopogon nardus (L.) Rendle, Echinochloa pyramidalis (Lam.) Hitchc. & Chase., Eriochloa fatmensis (Hochst. & Steud.) W. D. Clayton, Hyparrhenia dissoluta (Nees ex Steud.) C.E.Hubb., H. rufa (Nees) Stapf, H. dregeana (Nees) Stent, Jardinea congoensis Franch. ex Hack., Megathyrsus maximus (Jacq.) B.K. Simon & S.W.L. Jacobs, Paspalum virgatum L., Pennisetum polystachyon (L.) Schult., P. purpureum Schumach., Phragmites mauritianus Kunth., Rottboellia cochinchinensis (Lour.) W.D. Clayton, Setaria homonyma (Steud.) Chiov., S. incrassata (Hochst.) Hack., S. verticillata (L.) P.Beauv., wild sudan grass [Sorghum verticilliflorum (Steud.) Stapf)], Sorghum arundinaceum (Desv.) Stapf, Sporobolus sp., Urochloa mosambicensis (Hack.) Dandy, Vossia cuspidata (Roxb.) Griff. and sedges, such as Cyperus articulatus L., C. dives Del., C. exaltatus Retz., C. involucratus Rottb., Pycreus mundtii Nees, Schoenoplectus corymbosus (Roem. & Schult) J. Ryan. It has never been found on Typhaceae, contrary to Sesamia nonagrioides Lefèbvre.
Detection and identification
Host-plant parts affected by feeding larvae are leaves and stems. The most serious damage occurs at early plant stages. Leaf damage. Damage caused by larvae reduces yield by killing the growing point (“dead heart”) (Photo 1A), causing early leaf senescence and reduced translocation and lodging due to weakened stems. Larval feeding may cause the entire plant to die back. S. calamistis infestation increases the incidence and severity of stalk rot. Stem damage. Larvae can tunnel into stems, which results in holes, broken stems, and eventual death of the growing point of maize. Frass is also deposited in stems (Photo 1B). Seed damage. When larvae feed on seeds in the cob, frass deposits are visible (Photo 1C).

Morphology
Egg
Eggs are about 1 mm in diameter, hemispherical, and slightly flattened with radial ridges. They are creamy-white when laid but darken as they develop (Photo 2A).
Larva
The larva looks smooth and shiny and lacks obvious hairs or markings (Photo 2B). Larval color varies but is usually creamy-white with a distinctive pink suffusion. The head and prothoracic shield are brown; the dorsal part of the last abdominal segment bearing the anus is yellow-brown. Spiracles are elongated oval with black surrounds. Mature larvae are 30–40 mm long.
Pupa
Pupae are up to 18 mm long and brown to yellowish-brown with a wrinkled frontal region of the head. Pupa has a terminal “tail” (cremaster) with four large and two small spines (Photo 2C).
Adult
The wingspan in females is 20–30 mm and in males a little less (Photo 2D). The forewings are pale-brownish with variable but generally inconspicuous darker markings along the margin and an overall silky appearance. Hindwings are pearly white with a yellowish margin.

Biology
Female moths lay eggs between the lower leaf sheaths and the stem in batches of 10–40 eggs arranged in two to four contiguous rows. On average, each female lays around 300 eggs in 5 days. Egg-laying occurs from the time plants are 2 weeks old until flowering. The most serious damage, however, occurs at early plant stages. Most larvae penetrate the stem shortly after they emerge from their eggs. Larval feeding might result in dead hearts, and the tunneling and girdling activity of the larvae often break the stalk. During the ear-filling period, the majority of the larvae occur in the ears. Development of the larvae takes 4–6 weeks. Most larvae pupate within the stem or cobs. In tropical regions, S. calamistis develops throughout the year and has no period of suspended development (diapause). However, under drought or cold temperatures, development slows down and the pest is less abundant, occurring mainly on alternative host grasses. Mature larvae become inactive from the start of the dry season and remain in this stage until the rains begin. Under irrigation, development is uninterrupted.
Temperature-dependent development
S. calamistis successfully develops from egg to adult between 15ºC and 30ºC but fails at 12°C–35ºC, as no eggs hatched at these temperatures (see Annex 7.3.15). The total mean development time decreased with increasing temperature, from 184.8 days at 15°C to 51.9 days at 30°C. The percentage mortality of immature stages decreased with increasing temperatures of 15º–25ºC for egg and larvae, and 15º–28ºC for pupae. The minimum mortality for egg (41.8%) and larvae (29.3%) was observed at 25ºC and for pupae at 28ºC (3.2%). The mean longevity significantly decreased with increasing temperature, with 8.4 days at 15ºC for both sexes, and 6.7 and 7.4 days at 30ºC for female and male, respectively. The maximum fecundity of 242.9 eggs was observed at 25ºC.
The models established to describe the development time, survivorship, and reproduction were compiled into an overall stochastic phenology model that allows the S. calamistis life-table parameters to be estimated according to temperature (see Annex 7.3.15). The predicted intrinsic rate of natural increase (rm) indicates that populations develop within the range of 16º–31ºC. The negative r values below 16ºC and above 31ºC indicate that population size is decreasing due to high mortality or very low reproduction. The highest population growth can be expected at 26.5ºC (rm = 0.06519). Similarly, the finite rate of increase for the African pink stemborer peaked at 29ºC (λ = 1.06736) and was <1 when exposed to 15.5ºC (λ = 0.996) and 31.5ºC (λ = 0.995); λ values of <1 indicate that the population is decreasing. At 26.5ºC, doubling time (Dt) was shortest with 10.63 days. The highest gross reproductive rate was reached at 23ºC, with 290 female offspring/female. When mortality of females before reproduction was considered, the net reproductive rate (Ro) was highest at 24.5ºC, with around 53 female offspring/female. The mean generation time (T) decreased with temperature and was shortest at 29.5ºC, with 54.2 days from egg to egg. These simulations indicate that C. partellus is able to establish in most tropical and subtropical maize production areas of the world.
Means of movement and dispersal
S. calamistis moths are nocturnal and can fly long distances; however, the most likely means of entry into new areas is by introduction of infested planting material. There are risks of transporting eggs and actively feeding larvae on host plants. The long-distance spread is through translocation of stems from one location to another where they are used as animal feed.
Economic impact
S. calamistis is generally less important than Busseola fusca and Chilo partellus (see sections 4.4.1 and 4.4.2) as a pest of cereal crops in East and Southeast Africa but may be locally abundant; it is only a serious pest of cultivated cereals in Central and West Africa. S. calamistis accounted for 7–12% of stemborers during long rains and up to 23% of stemborers during short rains in Kenya. Further, surveys have shown that losses associated with S. calamistis account for less than 5% of potential harvest.
Geographical distribution
S. calamistis is found in sub-Saharan Africa (SSA) and some of the islands in the Indian Ocean (Mauritius, Reunion, Madagascar, Comoros) (Fig. 1). In East Africa, it occurs from sea level to 2,400 masl.
| Africa | Angola, Benin, Botswana, Burkina Faso, Cameroon, Comoros, Congo, DR Congo, Ethiopia, Gambia, Ghana, Ivory Coast, Lesotho, Madagascar, Malawi, Mozambique, Niger, Nigeria, Senegal, South Africa, Sudan, Swaziland, Zambia, Zimbabwe |
Phytosanitary risks
The European Plant Protection Organisation (EPPO) CODE assigned to S. calamistis is SESACA. It is also listed as one of the important crop pests in the CABI Crop Protection Compendium. S. calamistis is readily transmitted on infected plant material, thus its spread potential is considered high unless strict controls are imposed over movement of infested material. It is also a pest of significance under surveillance by national plant health institutions in SSA countries that are obligated by the International Plant Protection Convention to discharge plant protection functions.
Risks mapping under current and future climates
Global Risks
Changes in establishment and future distribution
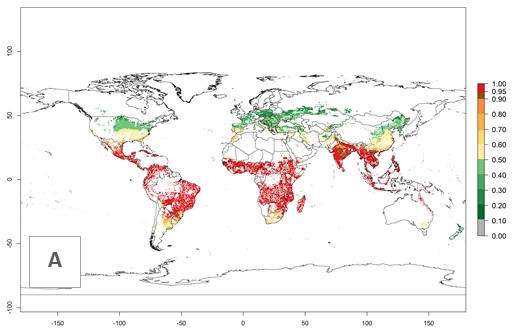
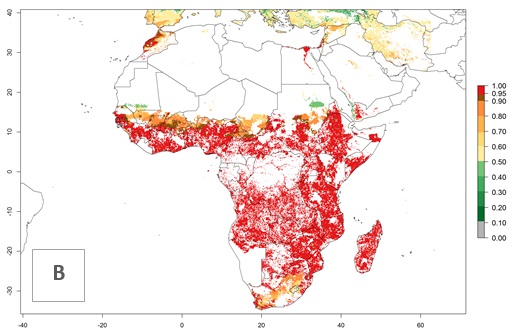
The establishment risk index (ERI) of 0.95–1 reflects well the current global distribution of S. calamistis under the current climate of the year 2000 (Fig. 2A); those regions include most SSA countries (compare with Fig. 1). Therefore, regions with an ERI>0.95 are considered to be at high risk of S. calamistis establishment as tropical regions of Central and South America, the Caribbean, Asia, and Oceania.
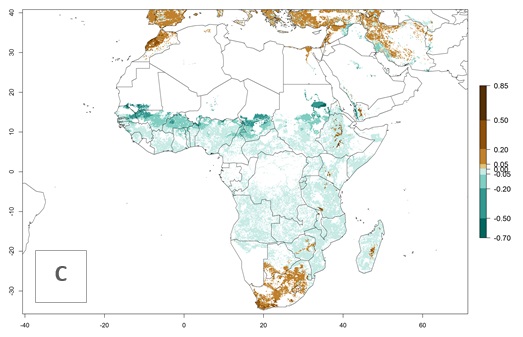
Figure 2. Changes in establishment and potential distribution of the A African pink stemborer, Sesamia calamistis, in maize production systems worldwide according to model predictions, using the ERI for the years 2000 (A) and 2050 (B), and changes of the ERI between 2000 and 2050 (C). An ERI>0.6 is associated with potential permanent establishment.
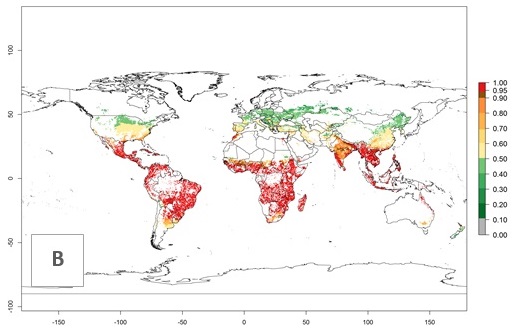
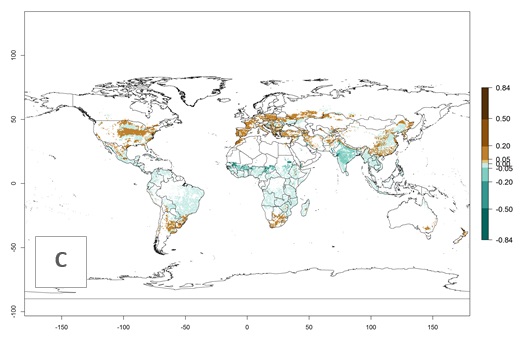
In zones where the ERI drops below the maximum number of 1, the likelihood of long-term establishment is reduced as in subtropical or temperate zones. S. calamistis also occurs in restricted maize-growing areas with an ERI>0.6–0.9 as in South Africa. S. calamistis has not been reported in regions with a low likelihood of establishment (ERI<0.6). Global predictions for 2050 indicate that S. calamistis will remain a high risk pest (ERI>0.9) for most tropical countries (Fig. 2B, C). A slight increase in the spread of S. calamistis but with a middle establishment potential (ERI>0.6–0.9) is predicted for subtropical regions of Asia (southern China); South America (northern Argentina, Uruguay, southern Brazil); North America (southern U.S.); northern Mexico; and Southern Africa (South Africa). A slight range expansion to temperate zones is observed, but with a generally low establishment potential (ERI<0.6) (Fig. 2B, C).
Changes in abundance
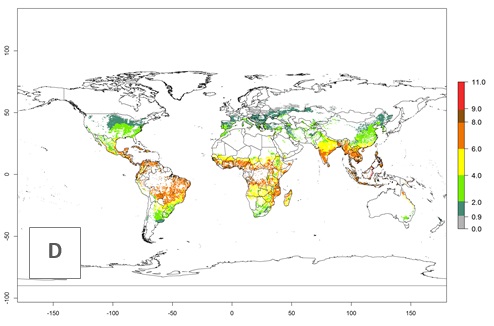
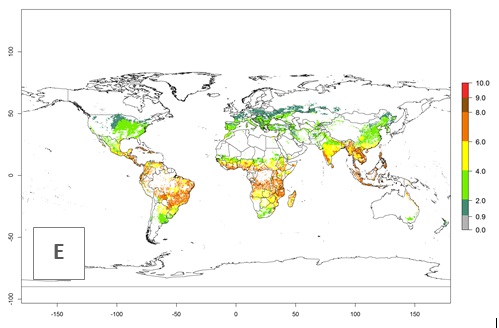
The mean number of generations that might theoretically develop within a year in the year 2000 and the year 2050 climate change scenario are visualized in Figure 3A, B, respectively. The generation index (GI) for the year 2000 estimates 5–7, 2–5, and 1–2 generations per year in most maize production systems of tropical, subtropical, and temperate regions worldwide, respectively. Globally, simulated GIs gave reasonable predictions when compared with generation numbers reported in the literature for S. calamistis (e.g., under tropical conditions in West Africa, 5–6 generations have been reported). Estimations made by the GI are consistent with these data (Fig. 3A, B). Predicted changes in the GI between the years 2000 and 2050 indicate that most maize-growing areas in tropical and subtropical regions will experience an increase by 1 generation per year. Global maps of the activity index (AI) in the year 2000 estimate a high activity of S. calamistis by a factor of 6–11 in most tropical regions, and by a factor of 2–6 for most subtropical regions of South America, East and Central Africa, and Southeast Asia, and up to 2 in temperate zones (Fig. 3D).
| GI | AI | |
| 2000 |  |
 |
| 2050 | 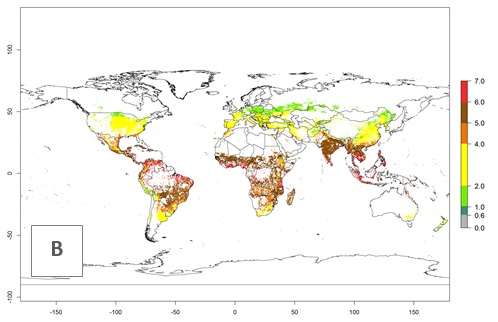 |
 |
| Index change (2000 – 2050) | 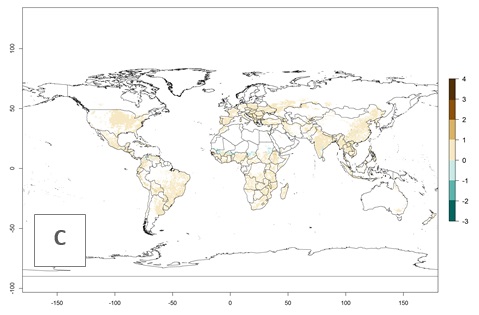 |
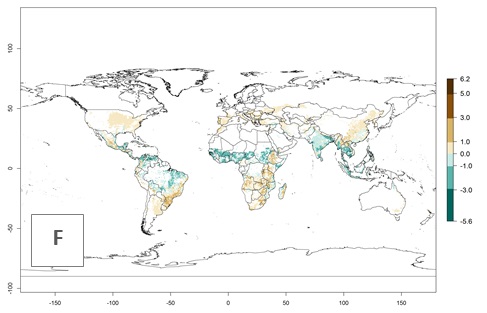 |
Figure 3. Changes in abundance (GI, damage potential) and activity (AI, potential population growth) of the African pink stemborer, Sesamia calamistis, in maize production systems worldwide according to model predictions, using the GI (A, B) and the AI (D, E) for the years 2000 and 2050, and the absolute index change (C, F).
Predictions of changes for the year 2050 temperature scenario show a decrease in the potential growth of S. calamistis in tropical regions of Central and South America, Africa (West region), and Asia (Fig. 3E, F). In most of subtropical regions the AI increases by values up to 5.
Regional Risks for Africa
Changes in establishment and future distribution
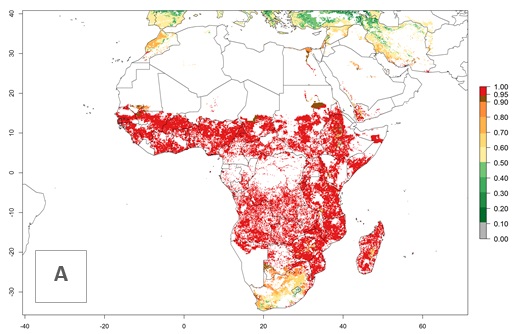
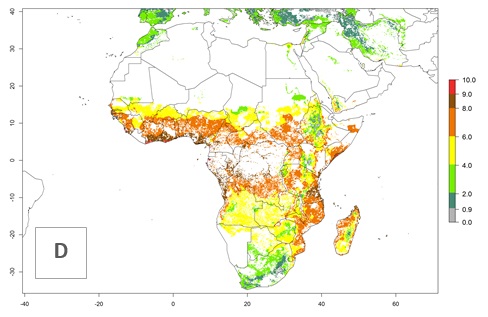
S. calamistis is already present in most countries of East, West, and Central Africa, with an ERI>0.95 indicating a high risk in the maize production areas (Fig. 4A). S. calamistis also occurs in restricted maize-growing areas with an ERI>0.6–0.9 as in South Africa. The predictions for the year 2050 indicate a slight (<0.05) reduction of the ERI in most lowland areas in West, East, and Central Africa, but S. calamistis will remain a high risk pest (ERI>0.9) for most tropical countries. The reduction, however, will be much more marked in the Sahelian region of Senegal, Mali, Burkina Faso, Chad, and Sudan and remain at a medium risk (0.7–0.9). On the other hand, a slight range expansion is expected in most highland regions of East and Southern Africa (Fig. 4B). Future predictions indicate that S. calamistis will represent a high risk (ERI>0.95) in most highland regions of Ethiopia, Kenya, Uganda, Rwanda, Burundi, Tanzania, Zambia, Angola, and Zimbabwe. Many other areas in South Africa and south Botswana will become more suitable (Fig. 4C).
Figure 4. Changes in establishment and potential distribution of the African pink stemborer, Sesamia calamistis, in African maize production systems according to model predictions, using the ERI for the years 2000 (A) and 2050 (B), and changes of the ERI between 2000 and 2050 (C). An ERI>0.6 is associated with potential permanent establishment.
Changes in abundance
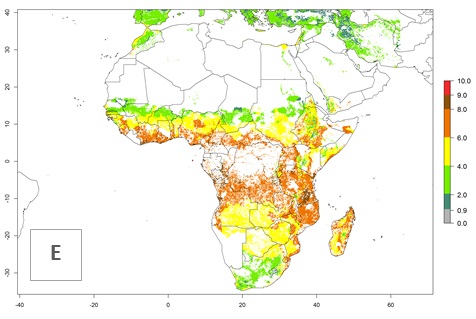
Under the current climate of the year 2000, 2–7 generations per year are estimated for most of the maize production areas in Africa (Fig. 5A). The highest number of generations per year (5–7) is predicted for West African countries.
| GI | AI | |
| 2000 | 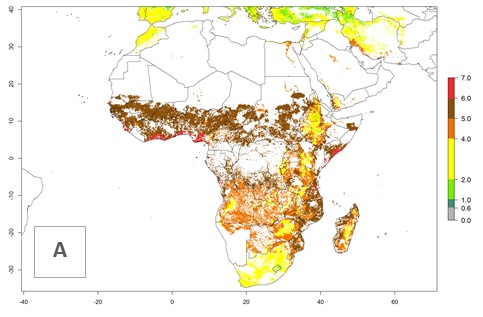 |
 |
| 2050 | 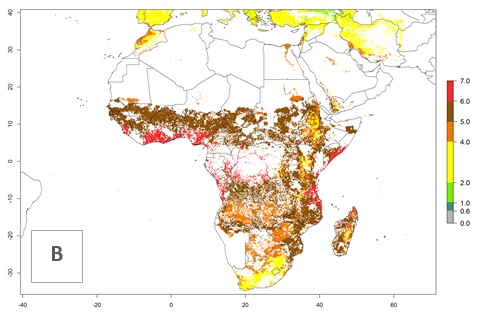 |
 |
| Index change (2000 – 2050) | 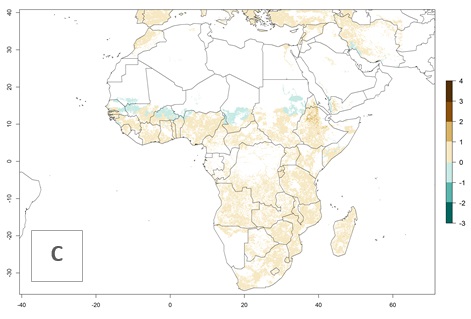 |
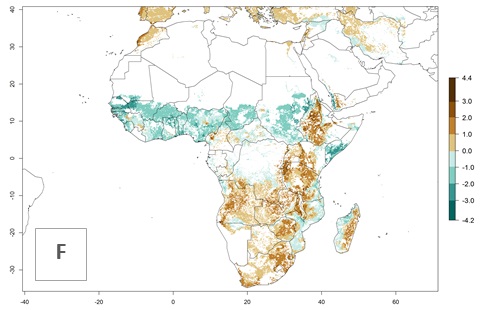 |
Figure 5. Changes in abundance (GI, damage potential) and activity (AI, potential population growth) of the African pink stemborer, Sesamia calamistis, in African maize production systems according to model predictions, using the GI (A, D) and the AI (B, E) for the years 2000 and 2050, and the absolute index change (C, F).
The GI change between the year 2000 and 2050 indicates a decrease of 1 generation per year in some northern lowland areas in the Sahelian belt in Senegal, Mali, Chad, and Sudan, whereas an increase of 1 generation per year can be expected in all other countries in SSA—Central, East, and Southern Africa (Fig. 5C). The highest activity (AI>8) of S. calamistis for the year 2000 is mainly estimated in the coastal areas of West (Sierra Leone, Ivory Coast, Ghana, Togo, Benin, Nigeria); Central (Gabon, Congo, northern coast of Angola); and East (southern Somalia, Kenya, Tanzania) Africa (Fig. 5D). For the year 2050, a reduction of the AI by a factor of 1–3 is predicted for most parts of SSA lowlands, whereas an increase by a factor of 1–4 is predicted for most African highlands, particularly in East Africa, northern South Africa, and Angola highlands (Fig. 5D–F).
Country Risk Maps
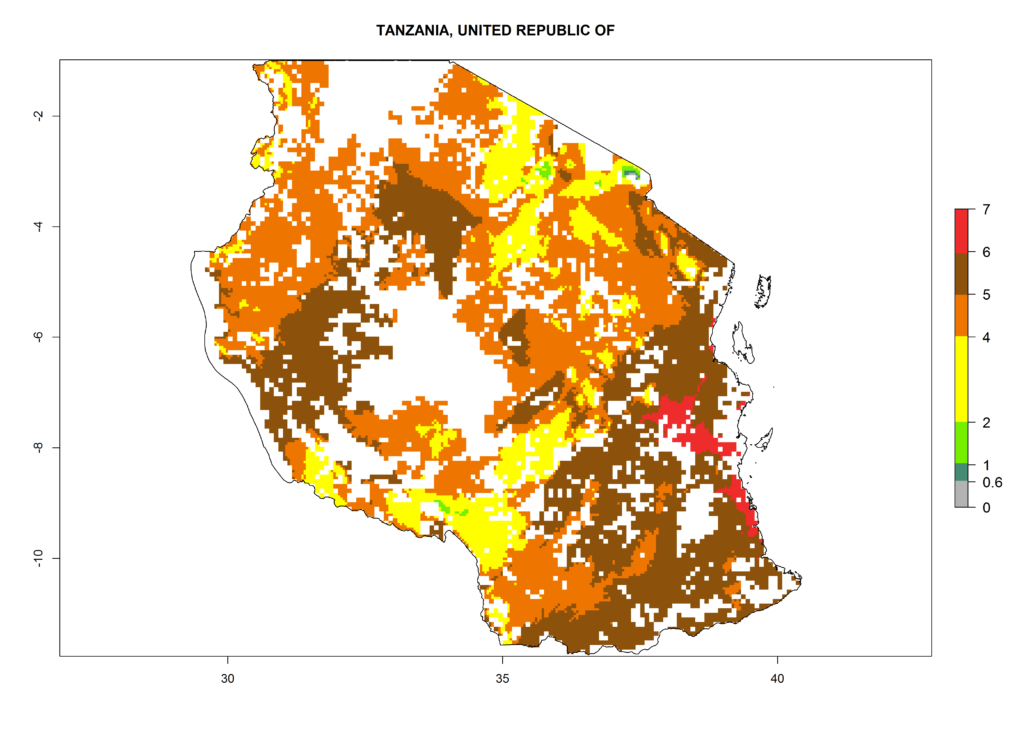
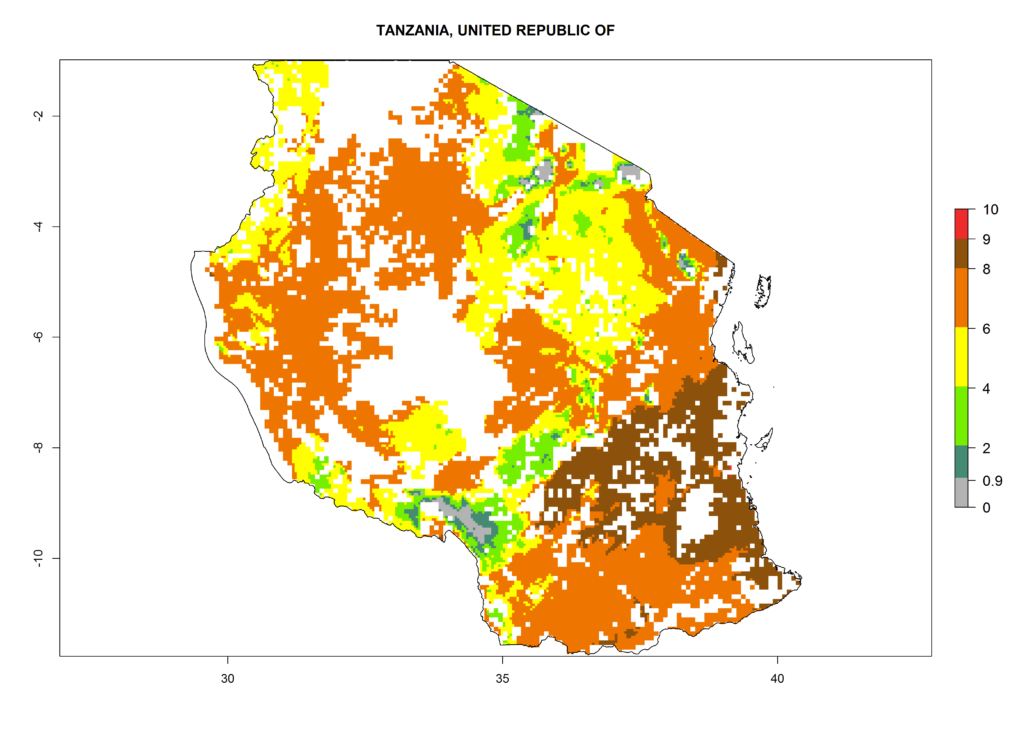
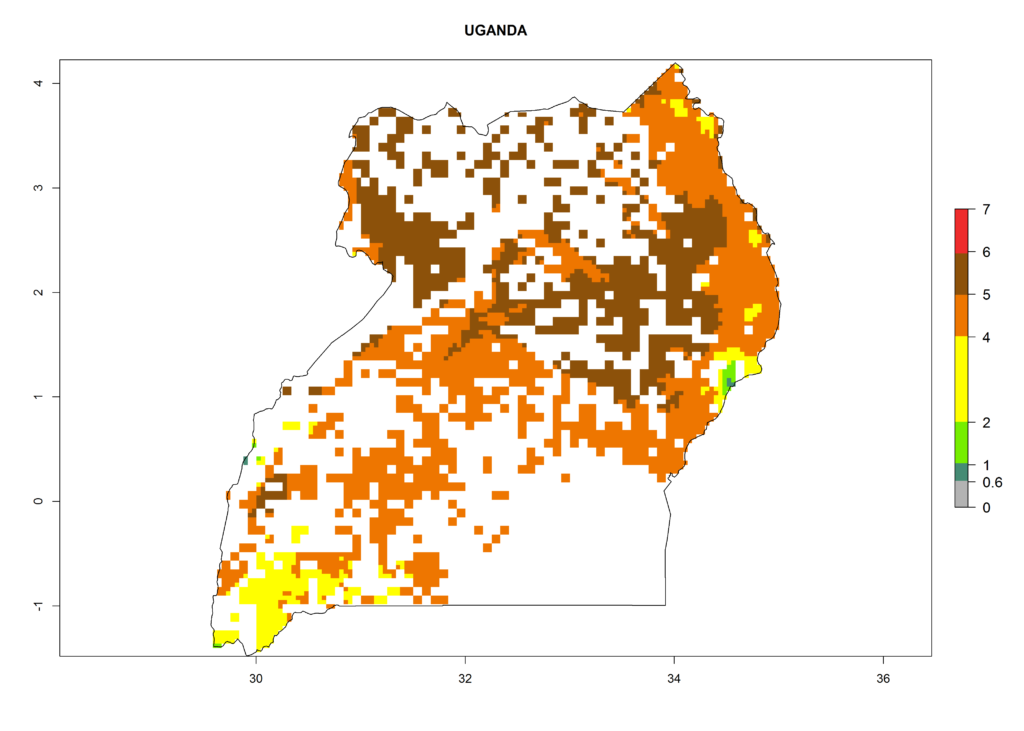
As examples, in Kenya S. calamistis potentially occurs in the main maize-growing zones with an ERI>0.95 and about 2–5 generations per year. Lower numbers of generations (GI>2) are predicted for the highlands. The AI (AI>0.9–9) widely varies in the different maize-growing regions (Fig. 6f). In Uganda, an ERI>0.95 is indicated for most maize cultivation areas. Four to five generations are predicted to develop per year. An AI>4–6 is projected (Fig. 6o). For Tanzania, an ERI>0.95 is indicated for all maize cultivation areas except for the central part of the country and around Lake Victoria. S. calamistis potentially develops 2–7 generations per year and has an AI>2–8 in the different maize growing regions (Fig. 6n).
| ERI | GI | AI |
a) Angola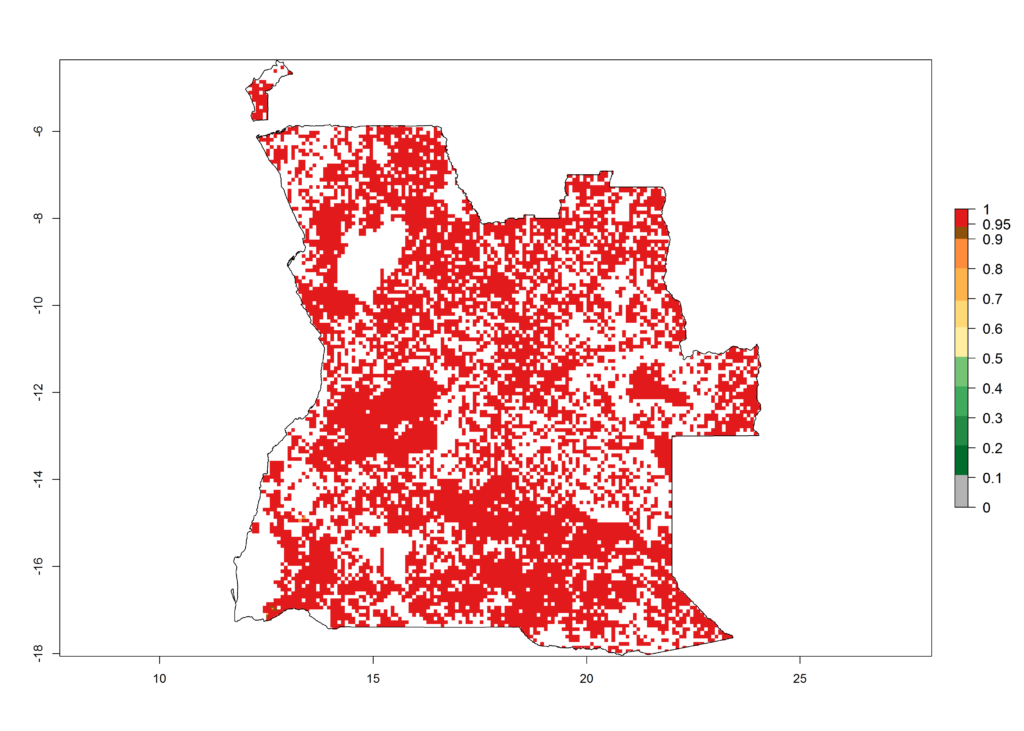 |
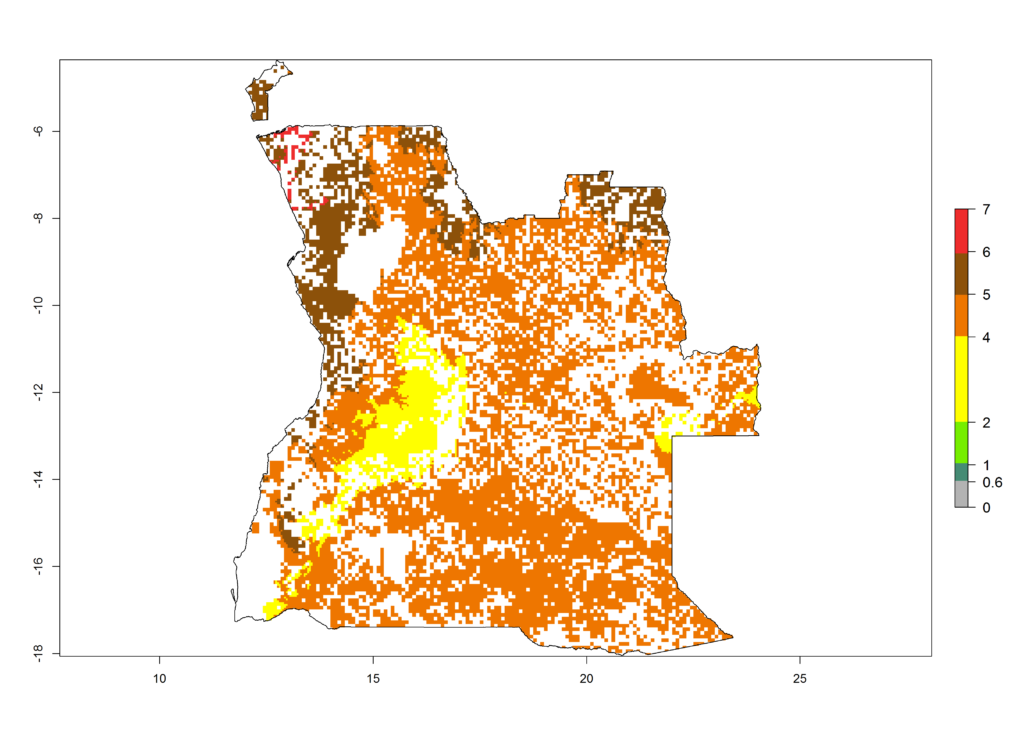 |
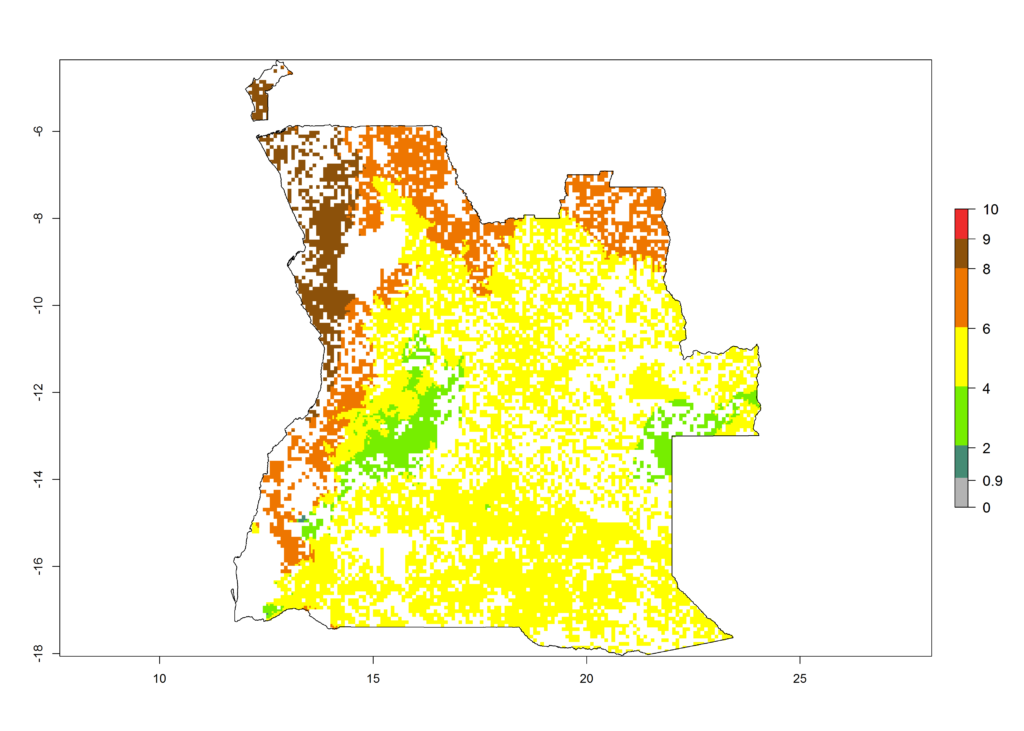 |
b) Burkina Faso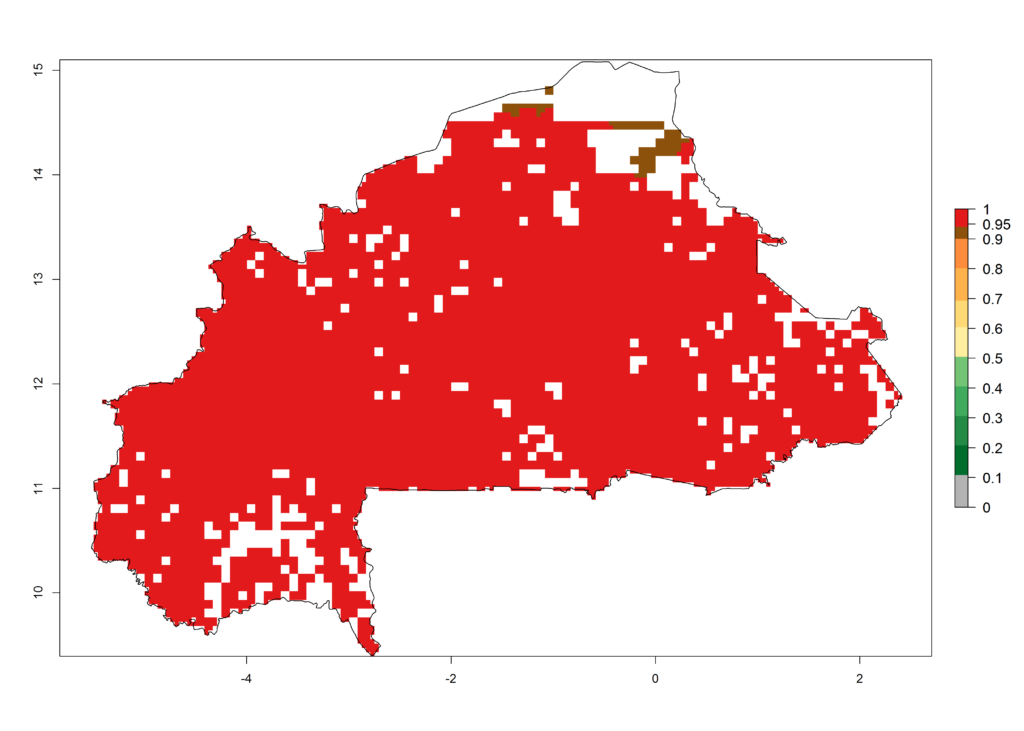 |
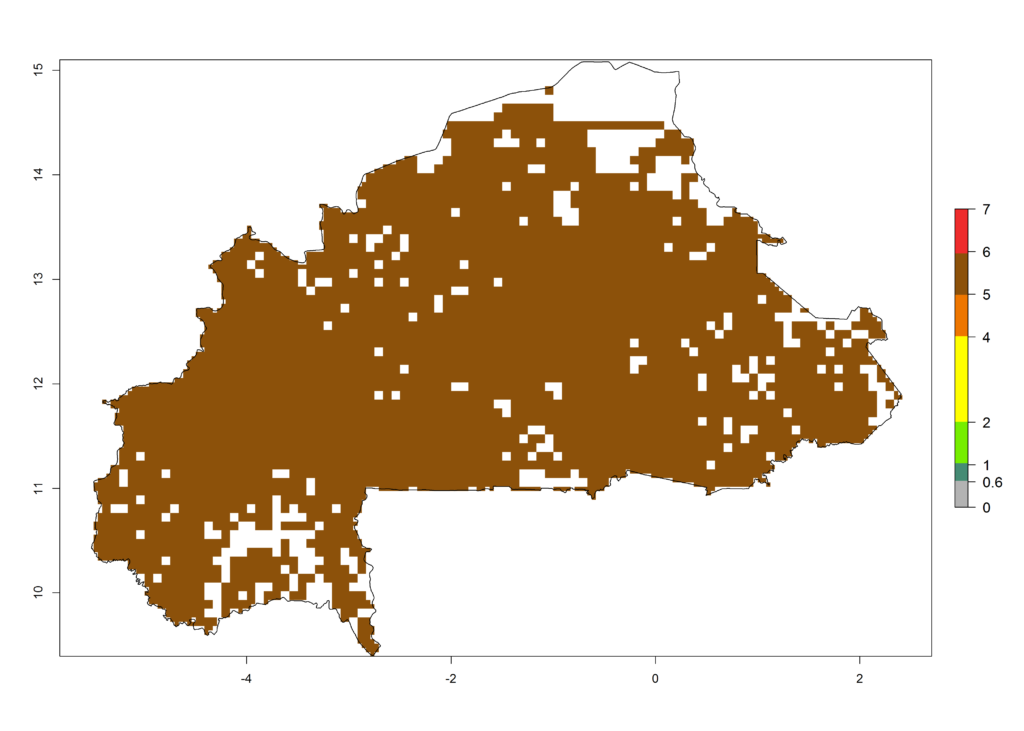 |
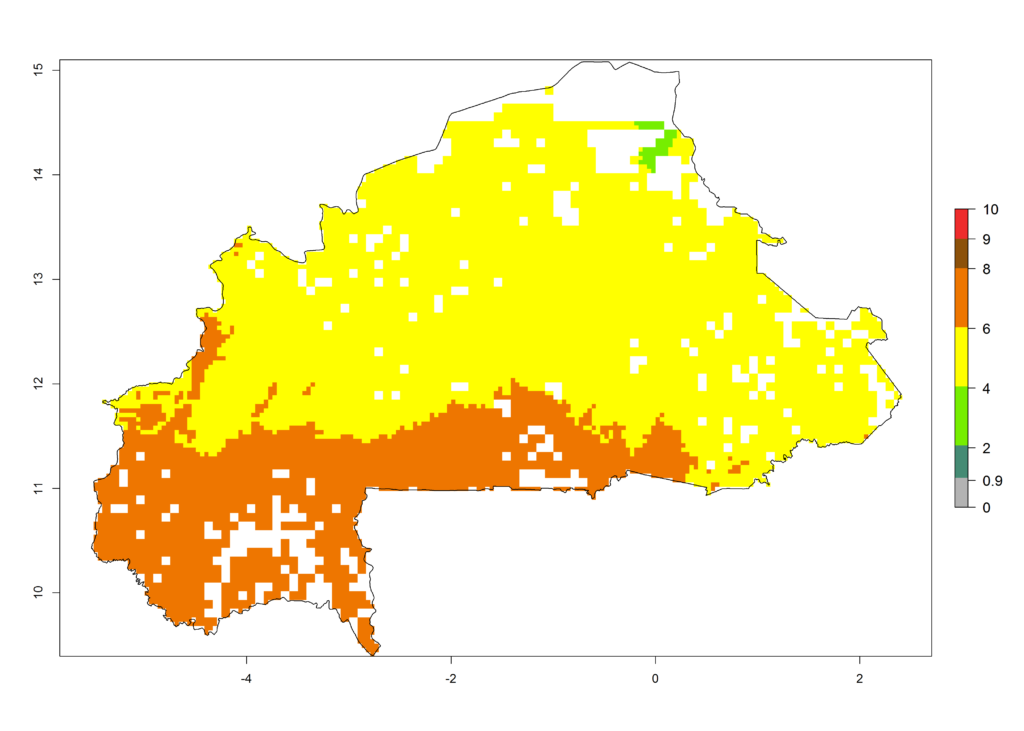 |
c) Burundi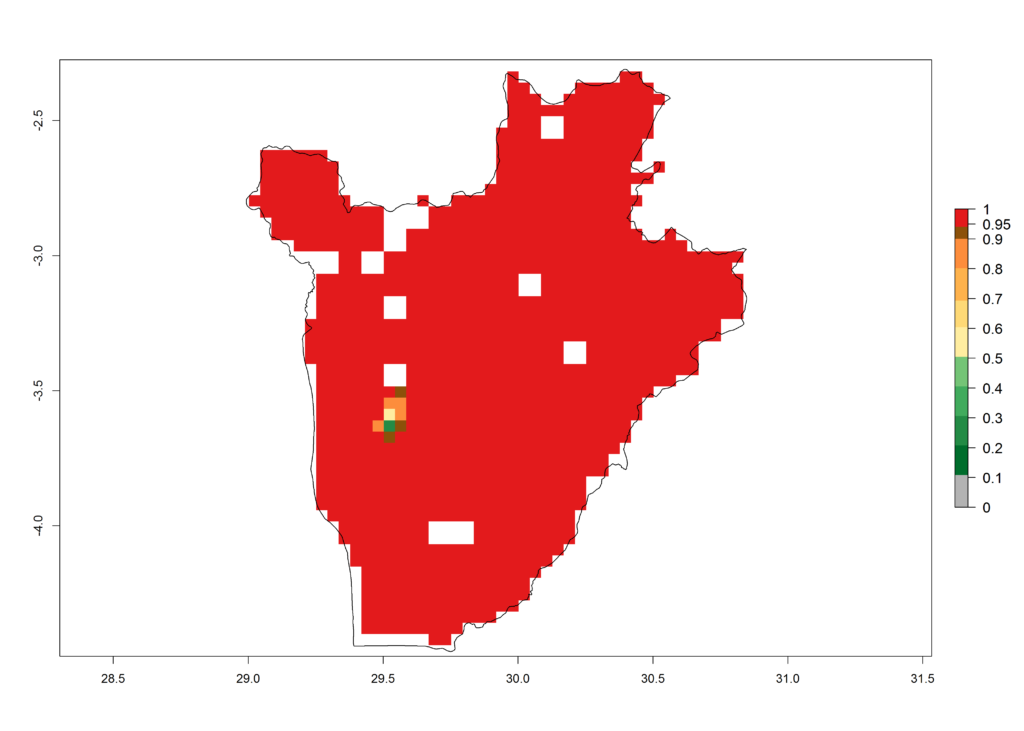 |
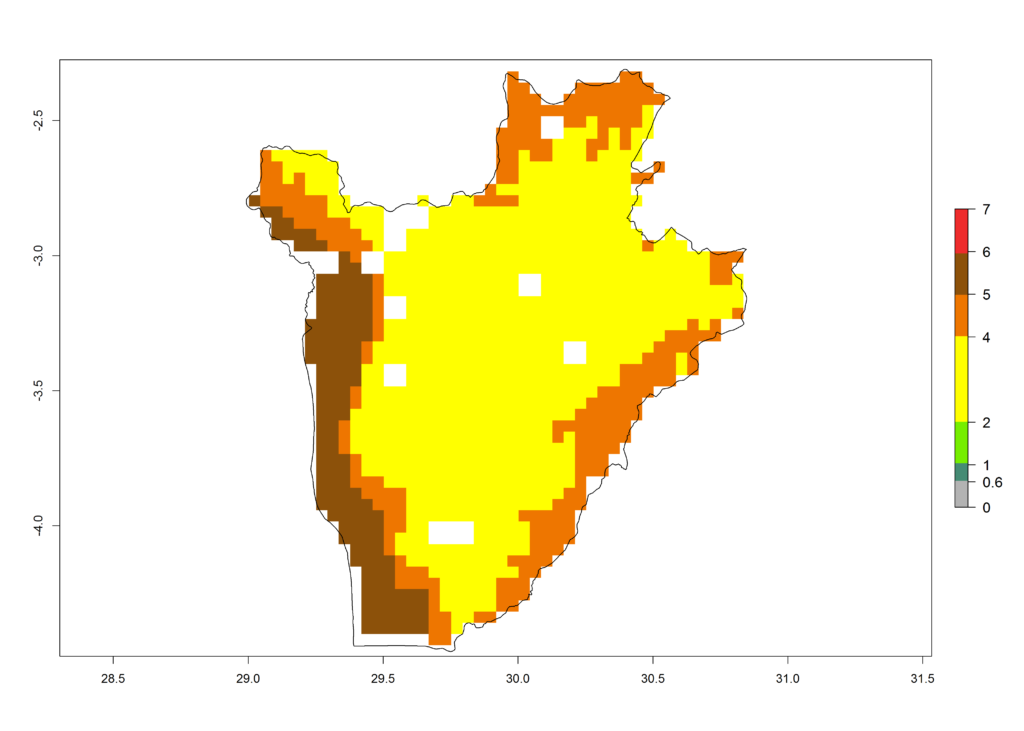 |
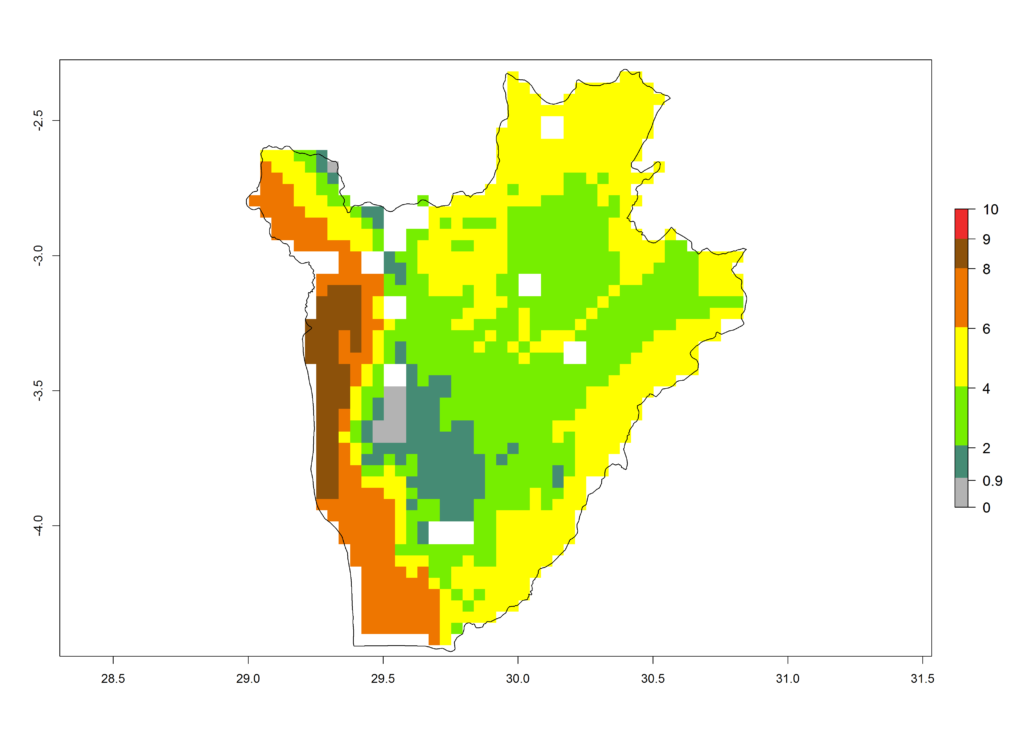 |
d) DR Congo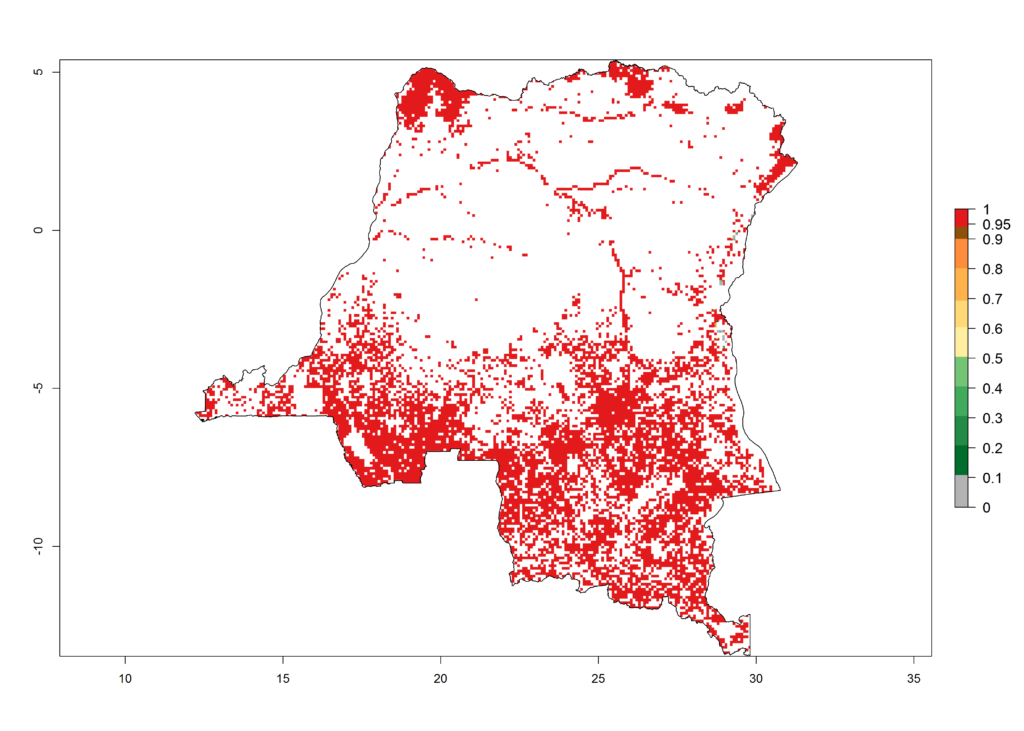 |
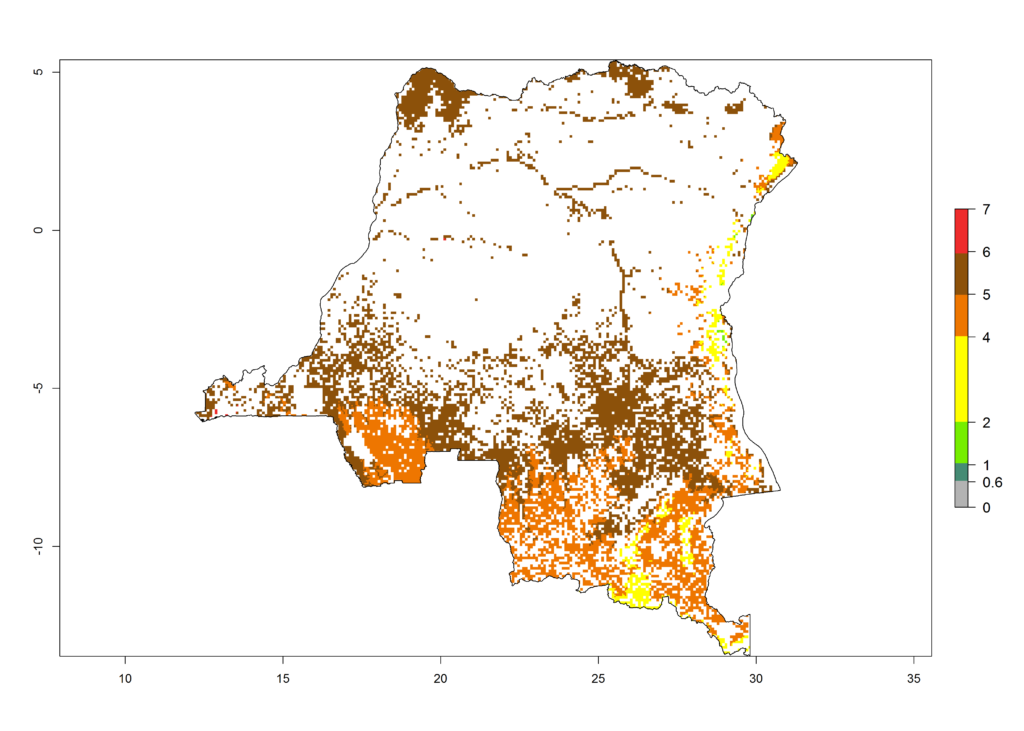 |
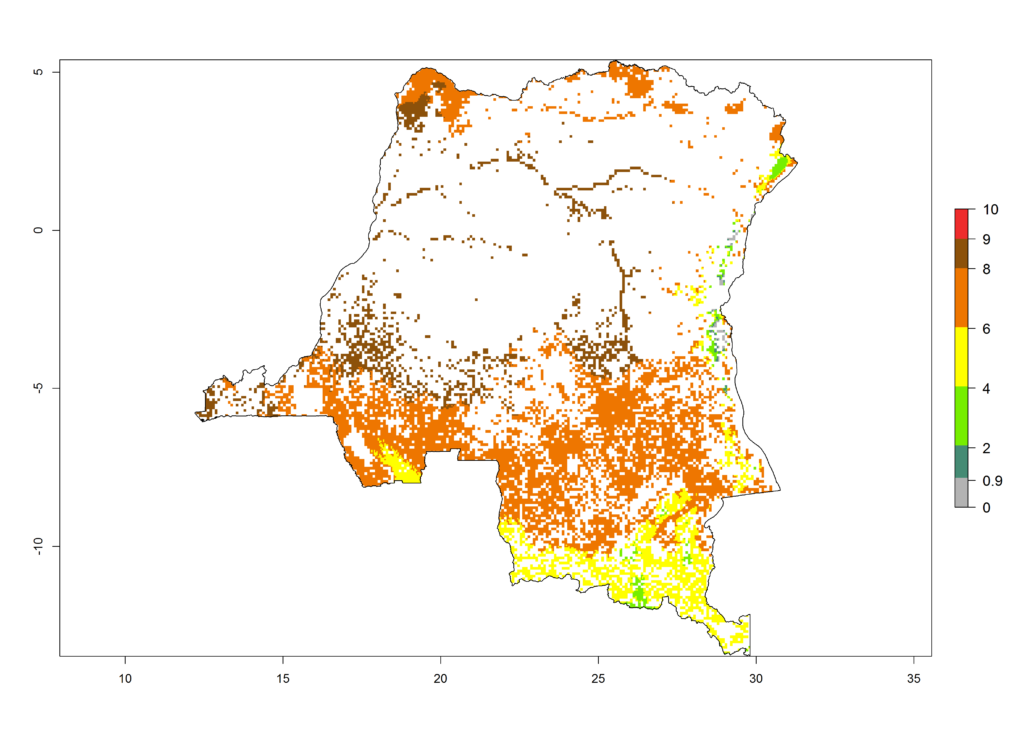 |
e) Ivory Coast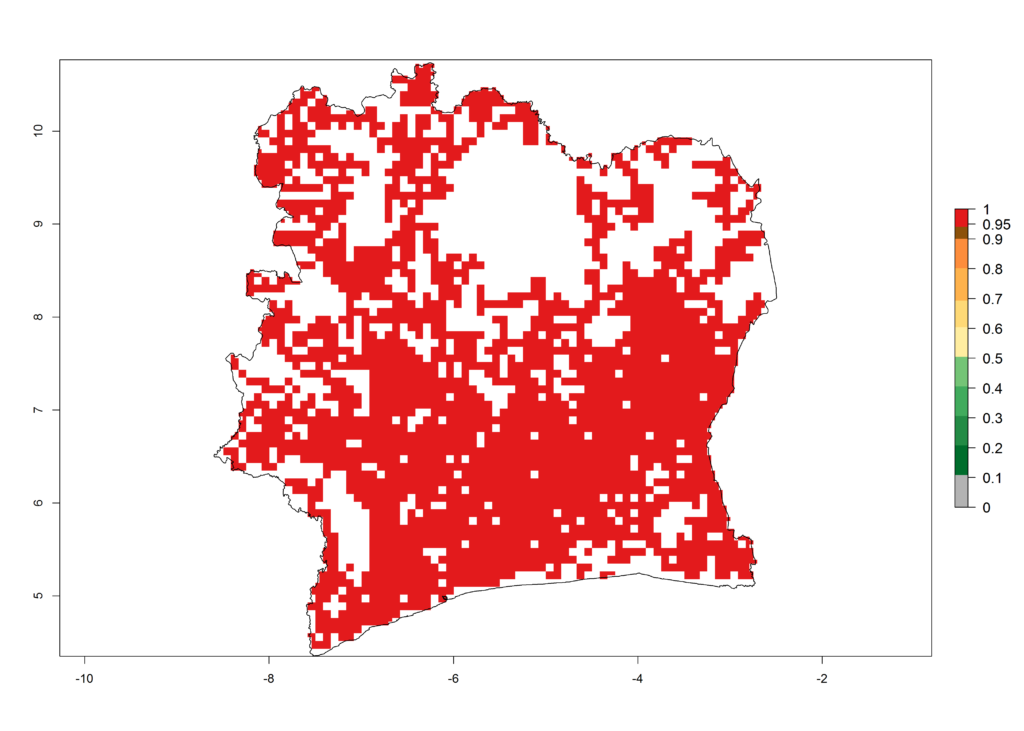 |
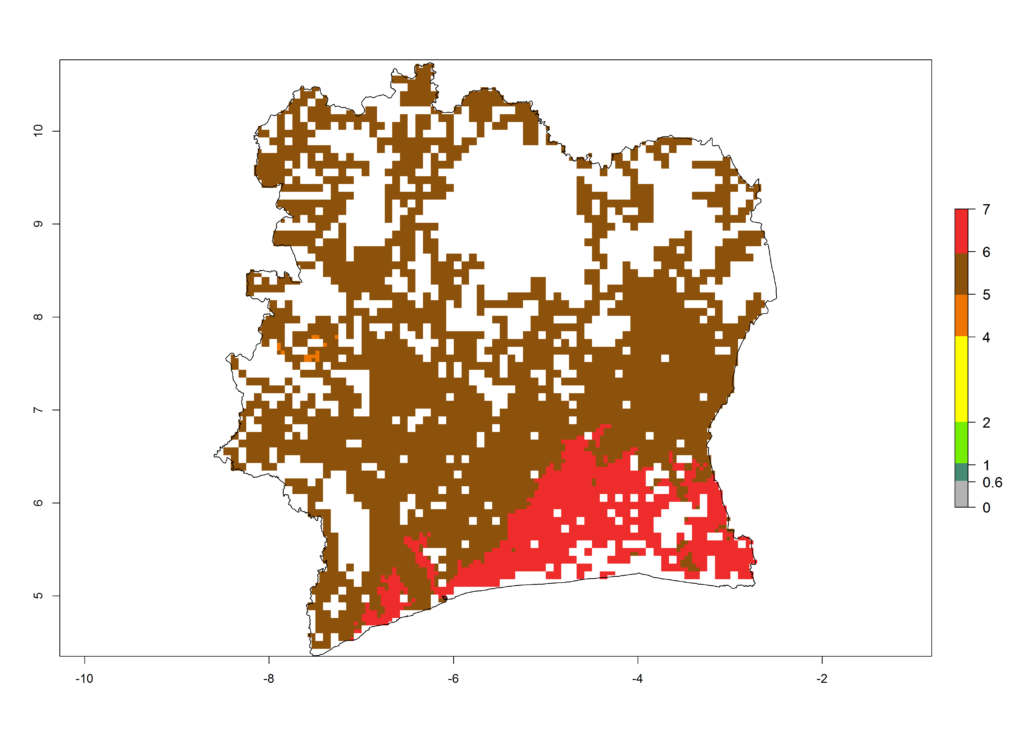 |
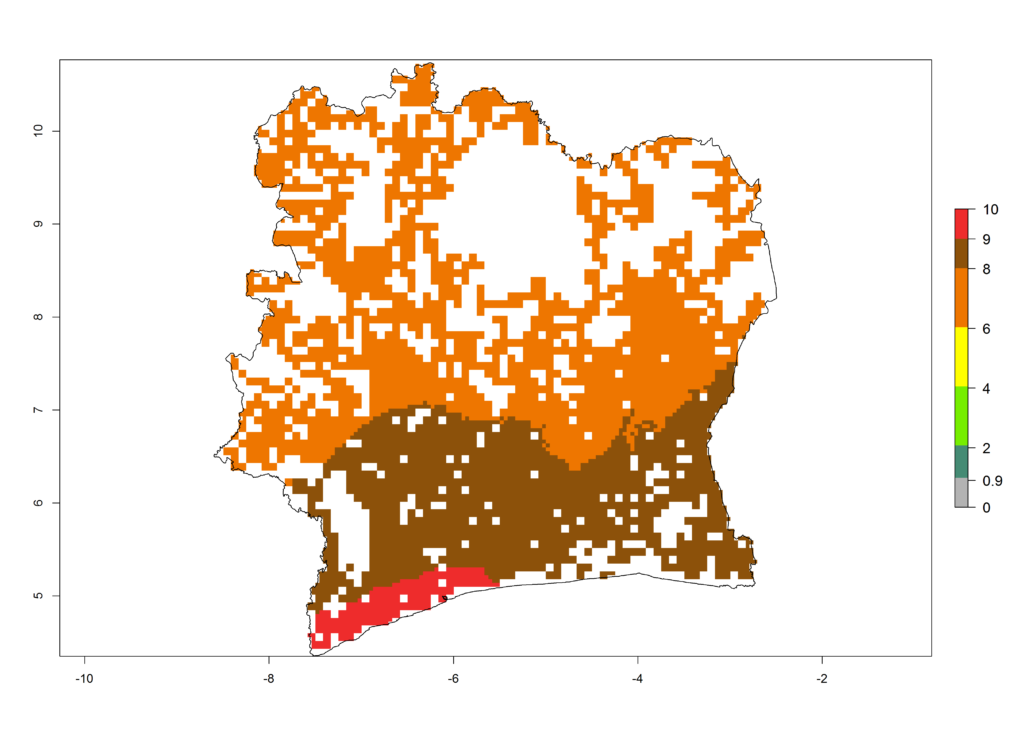 |
f) Kenya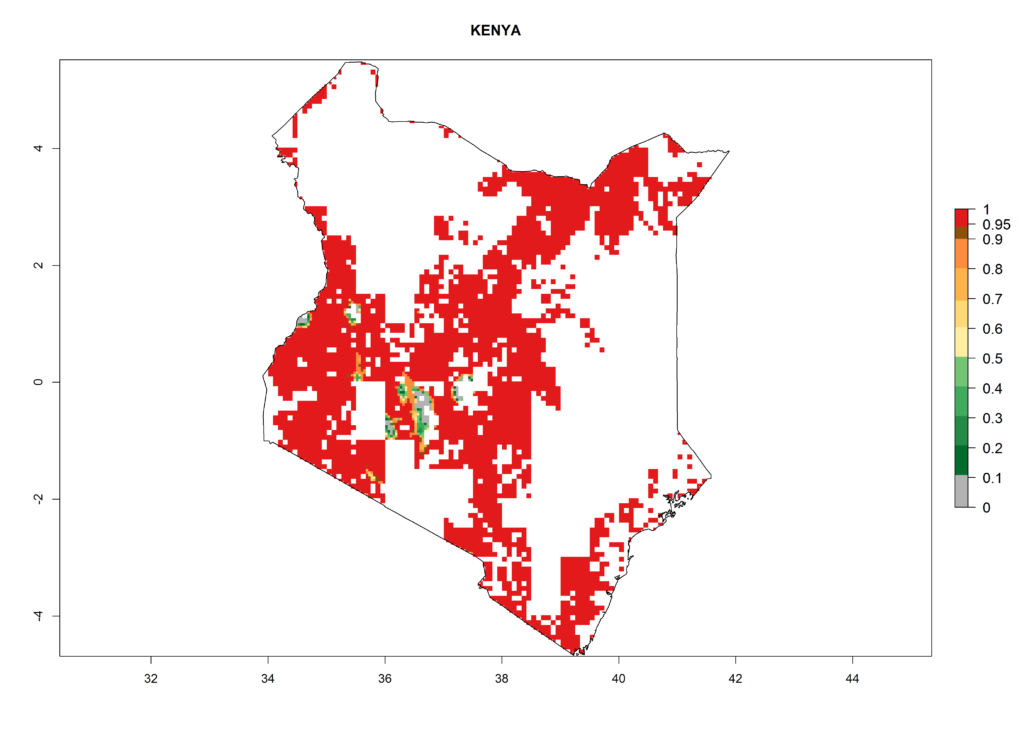 |
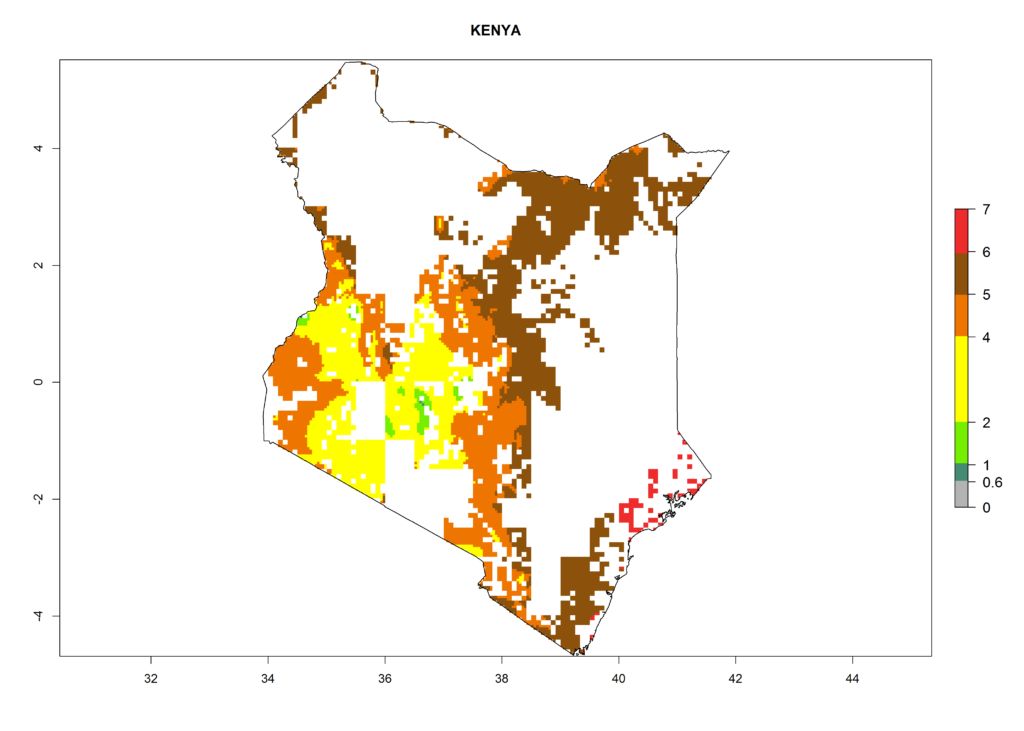 |
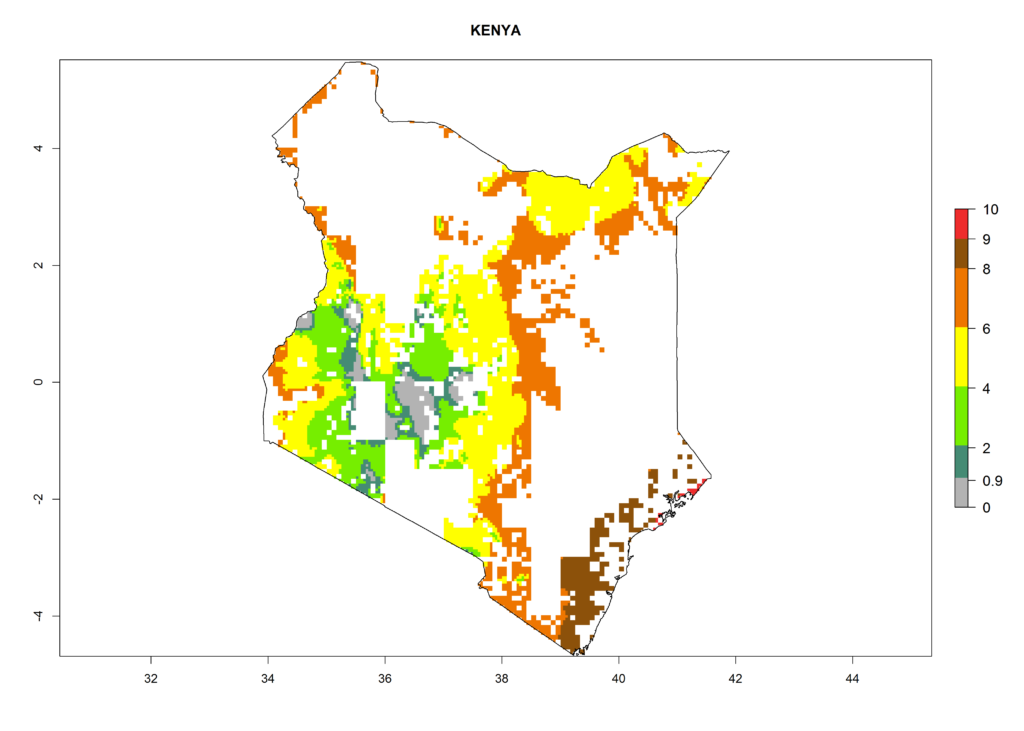 |
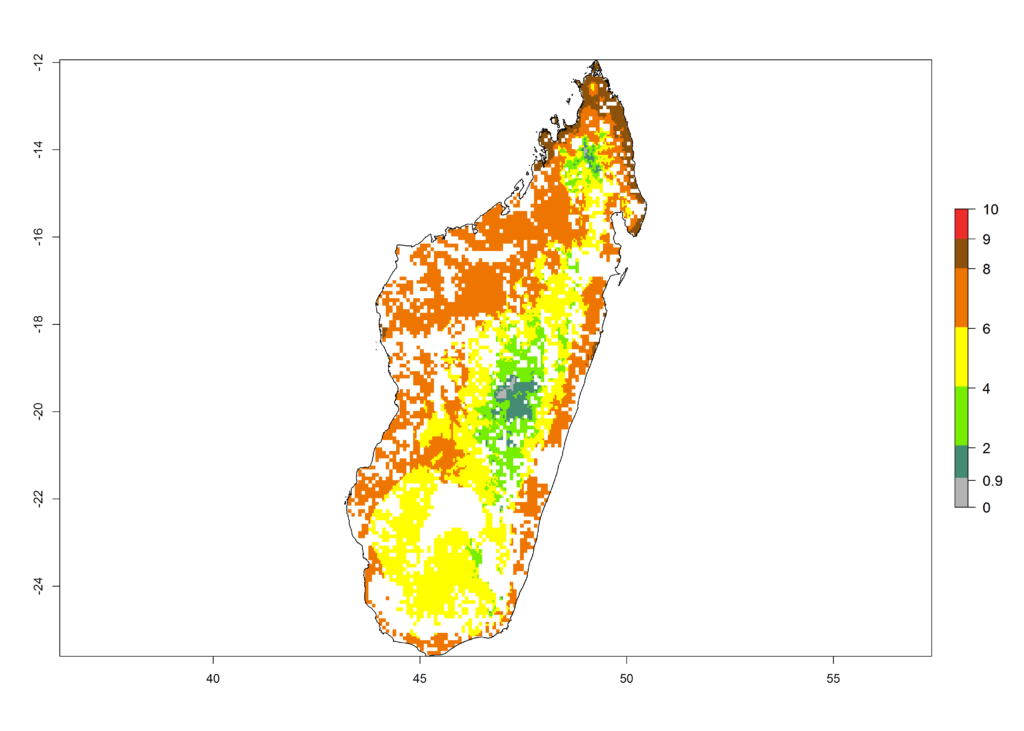
g) Madagascar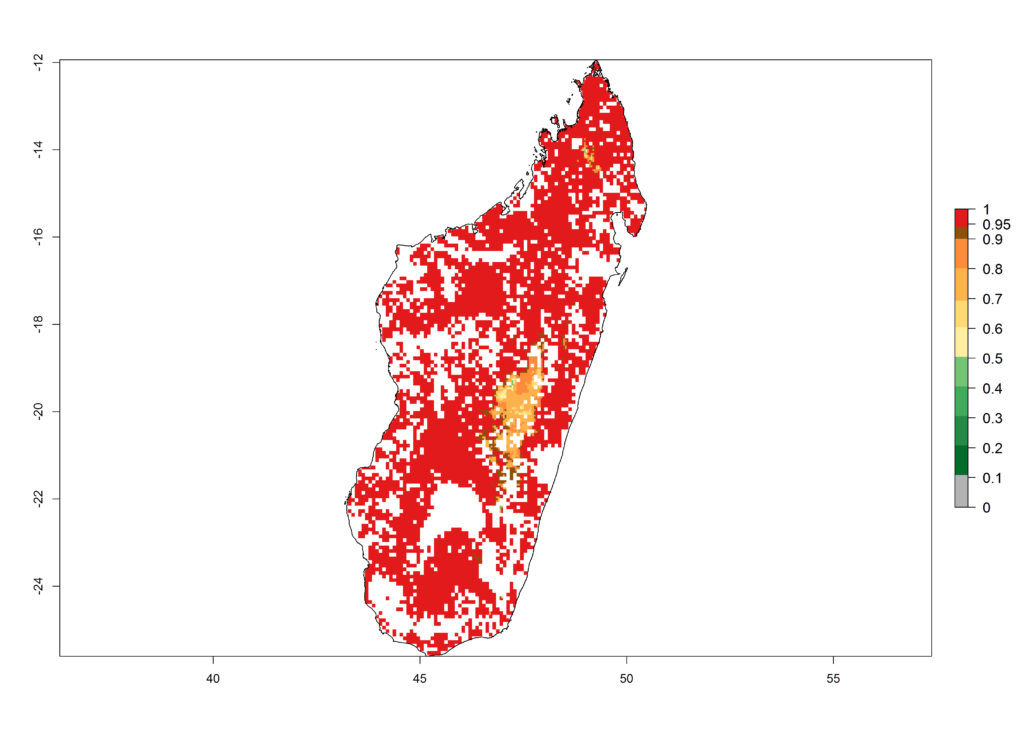 |
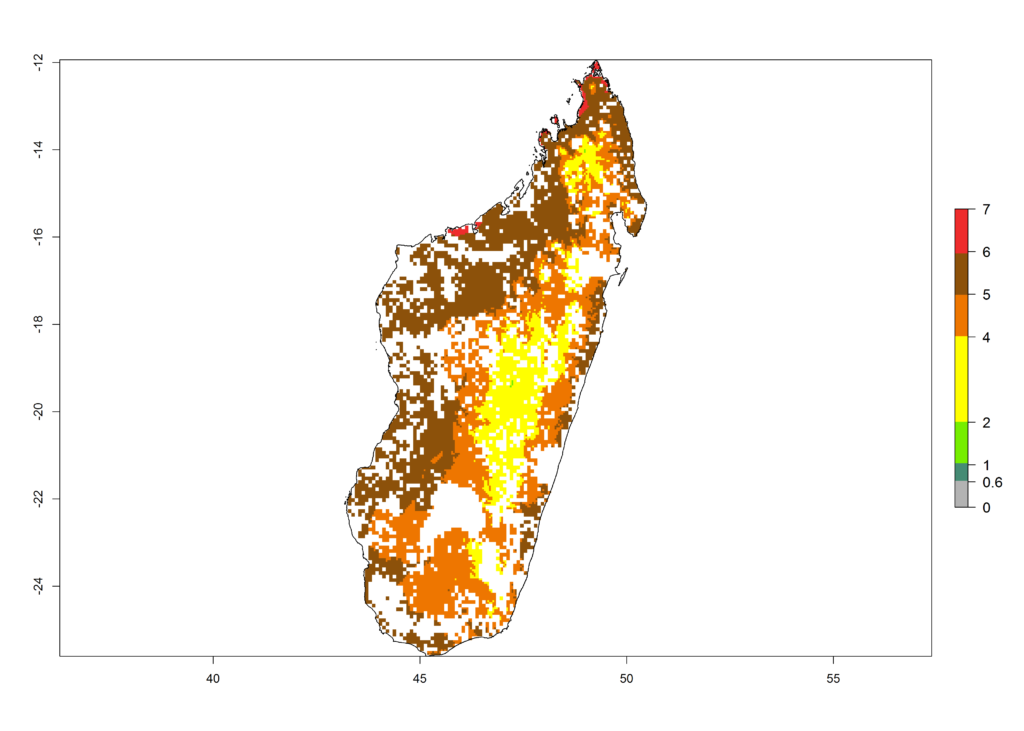 |
 |
h) Mozambique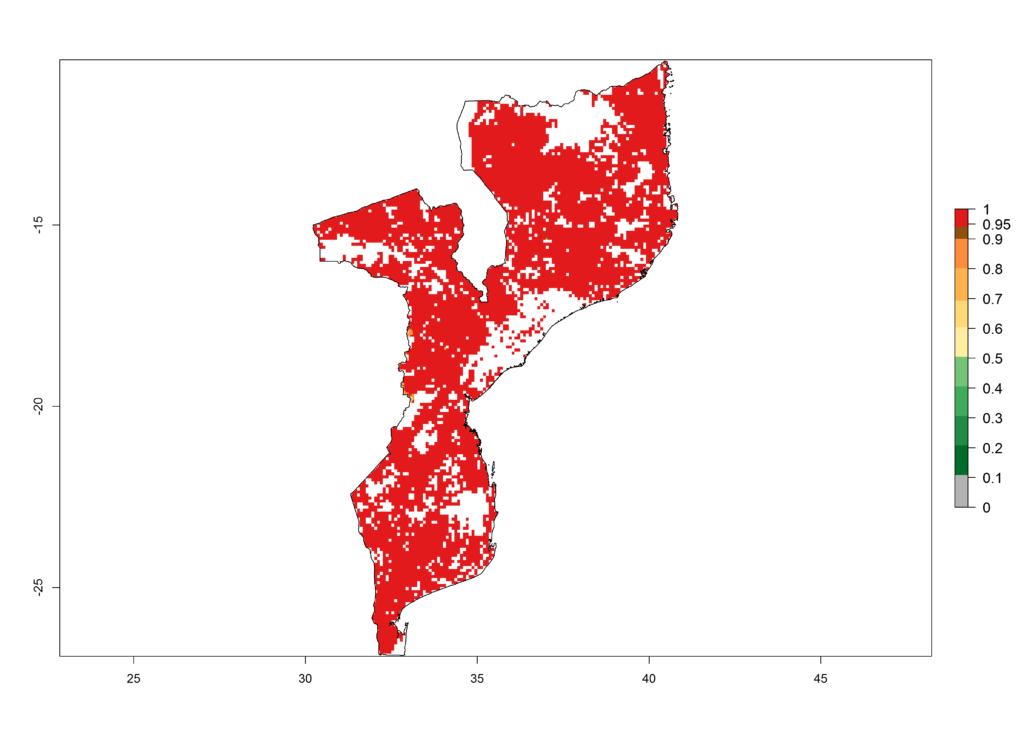 |
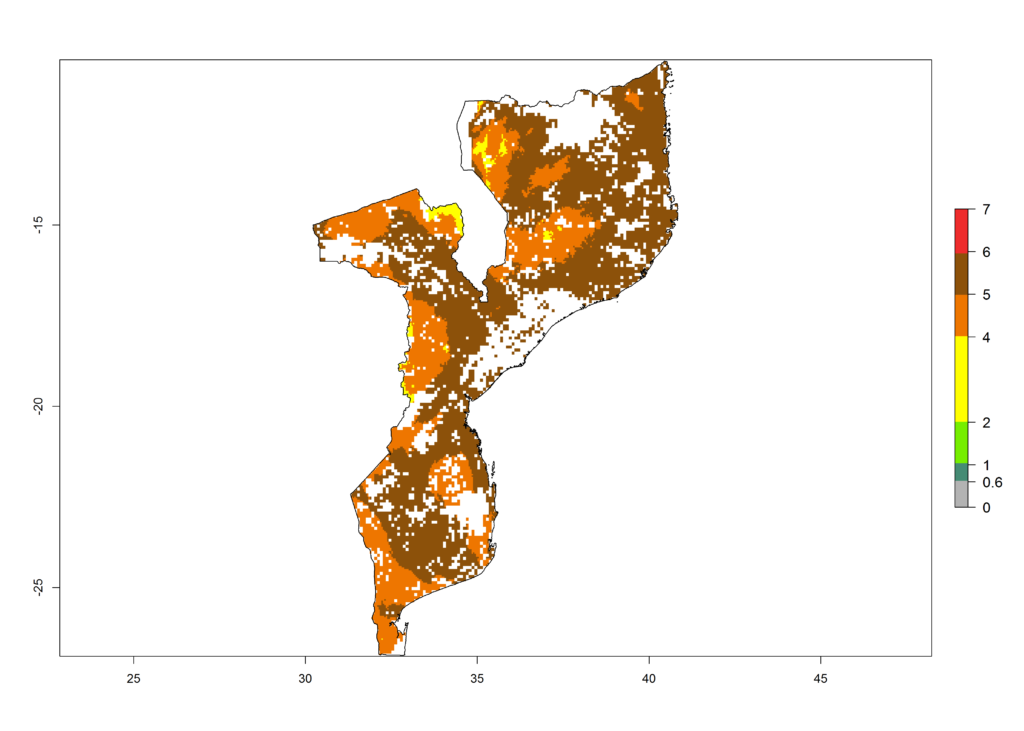 |
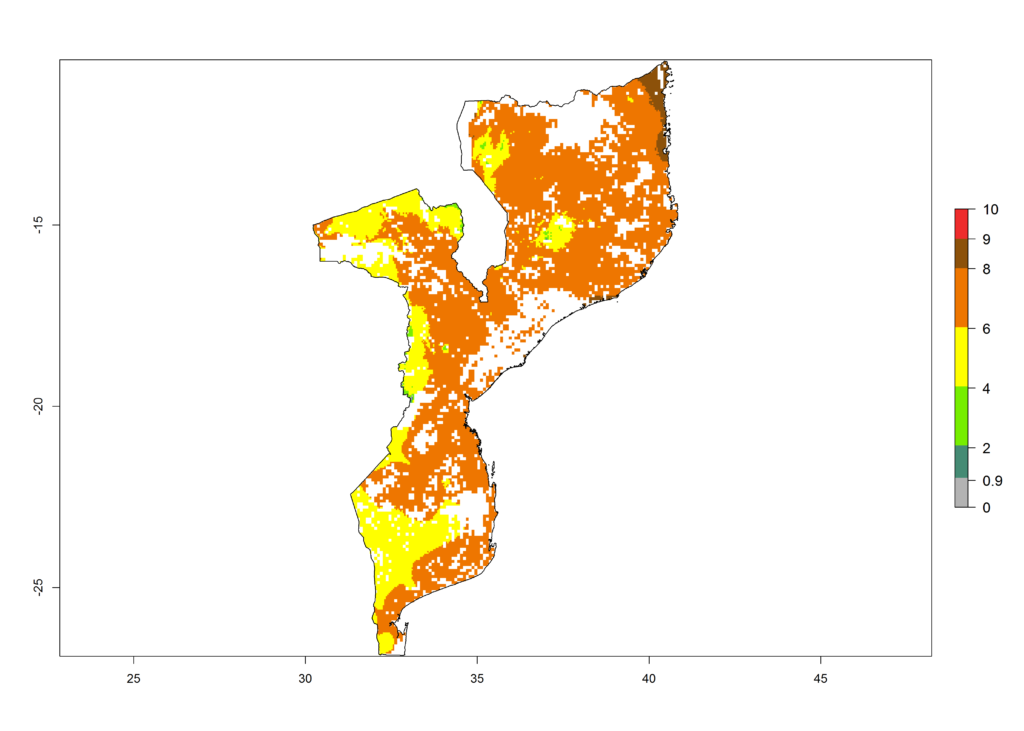 |
i) Nigeria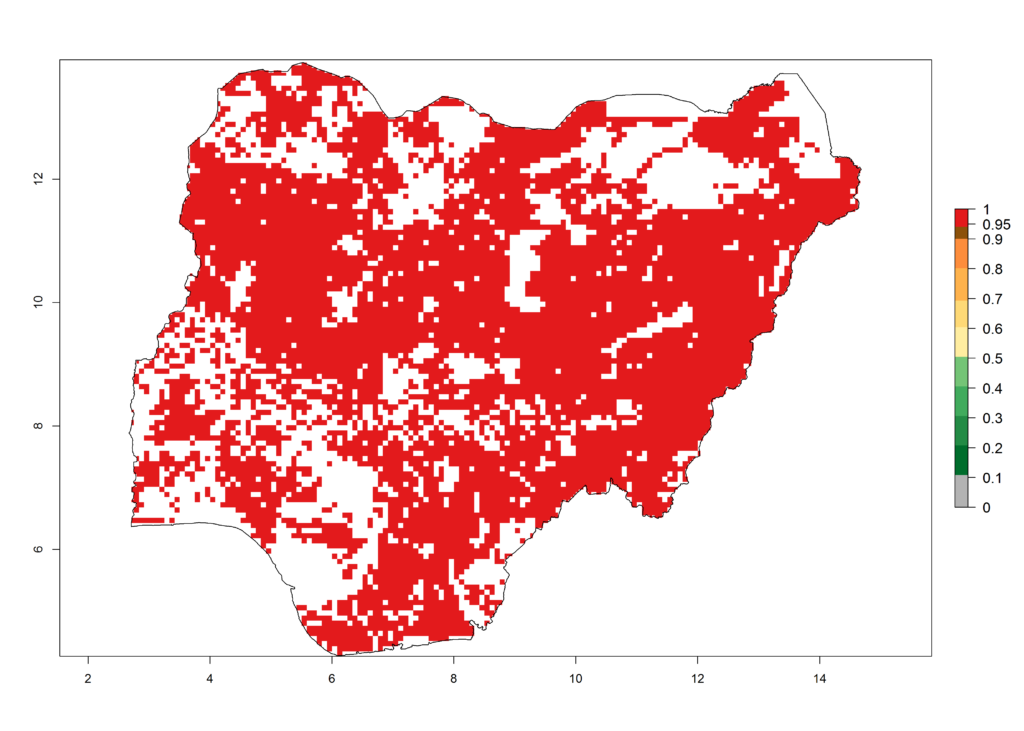 |
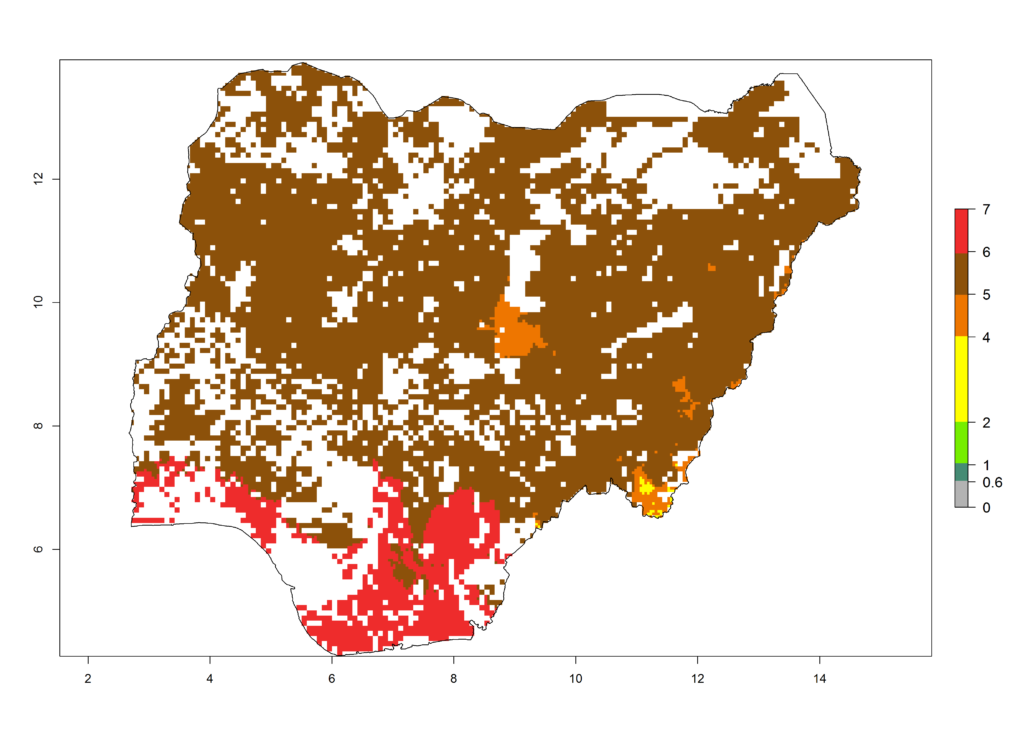 |
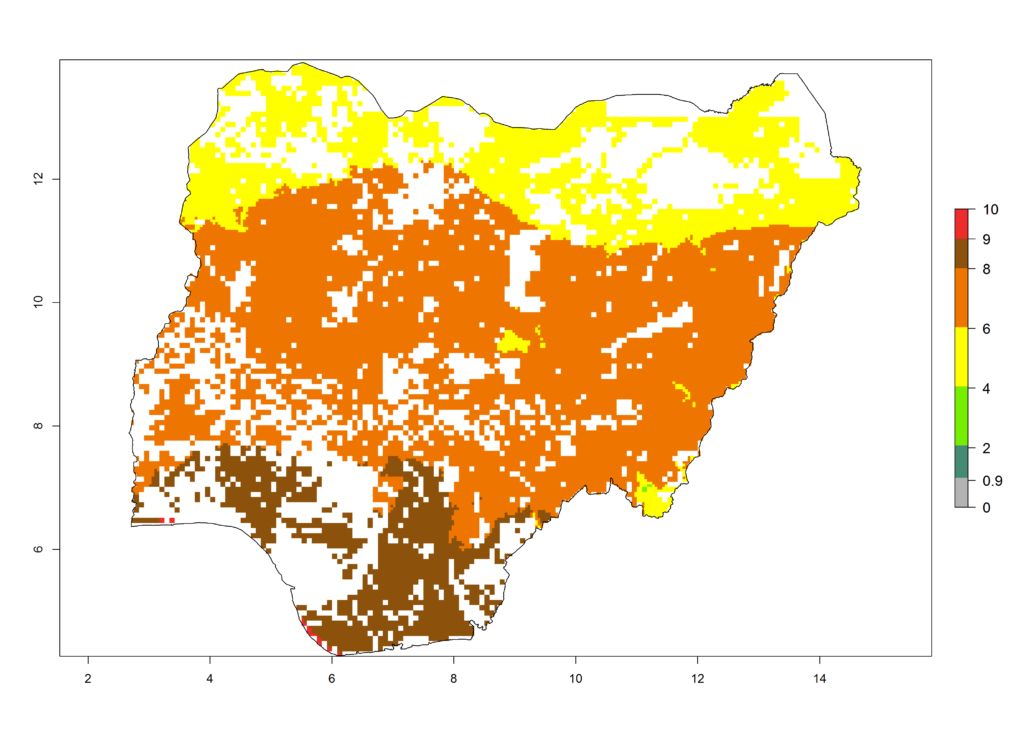 |
j) Rwanda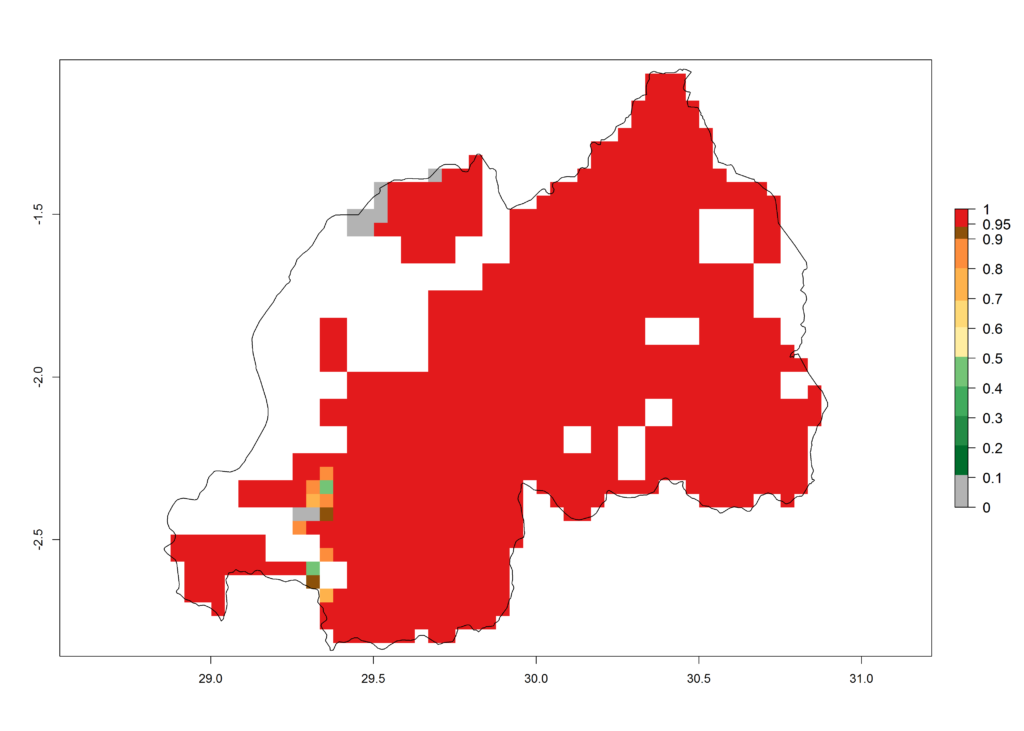 |
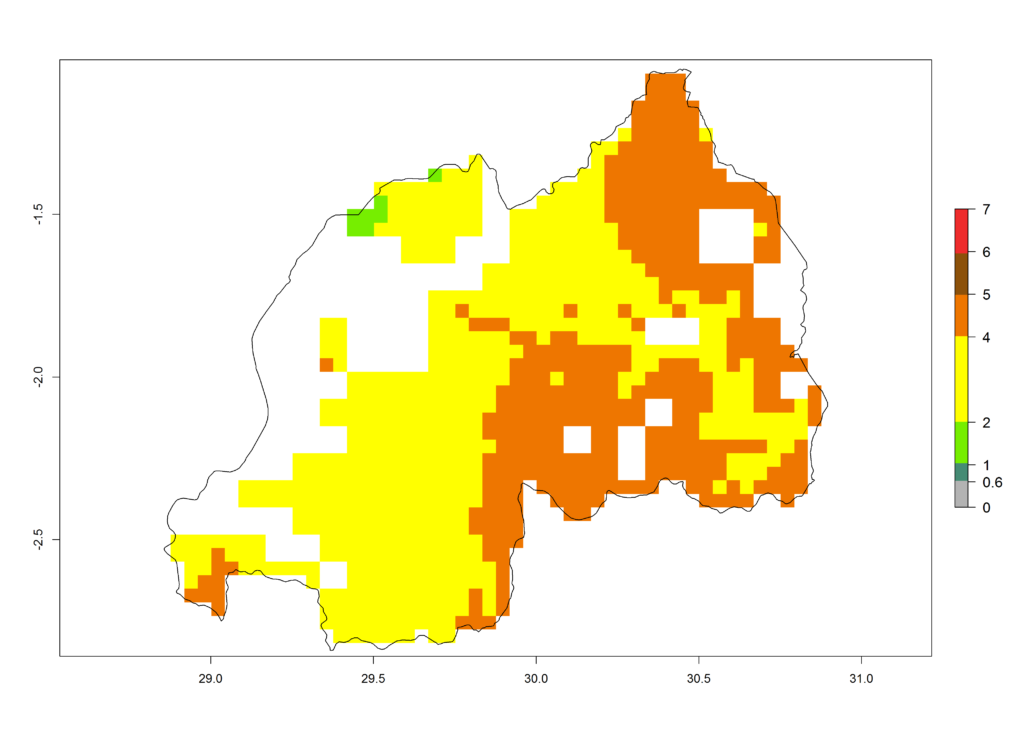 |
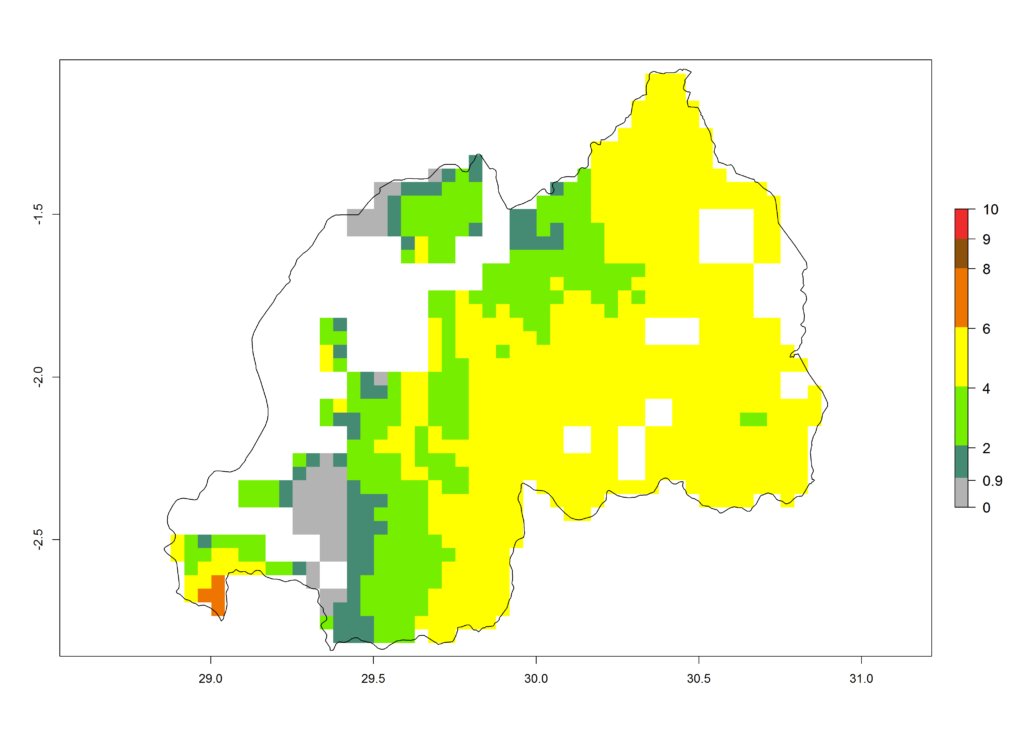 |
k) Senegal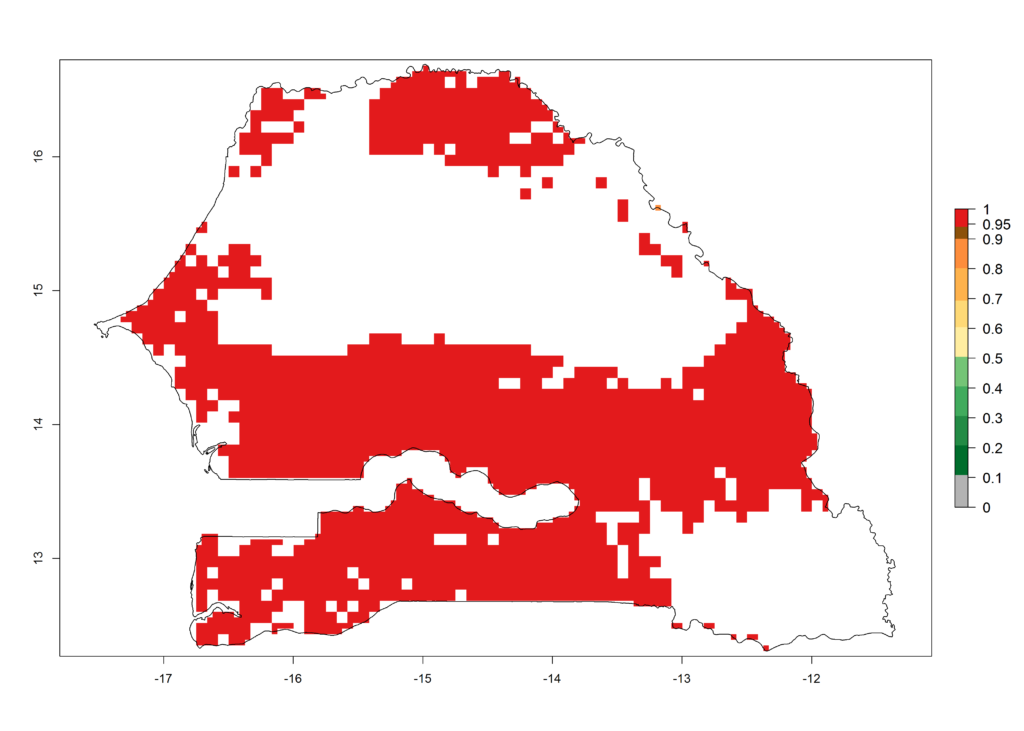 |
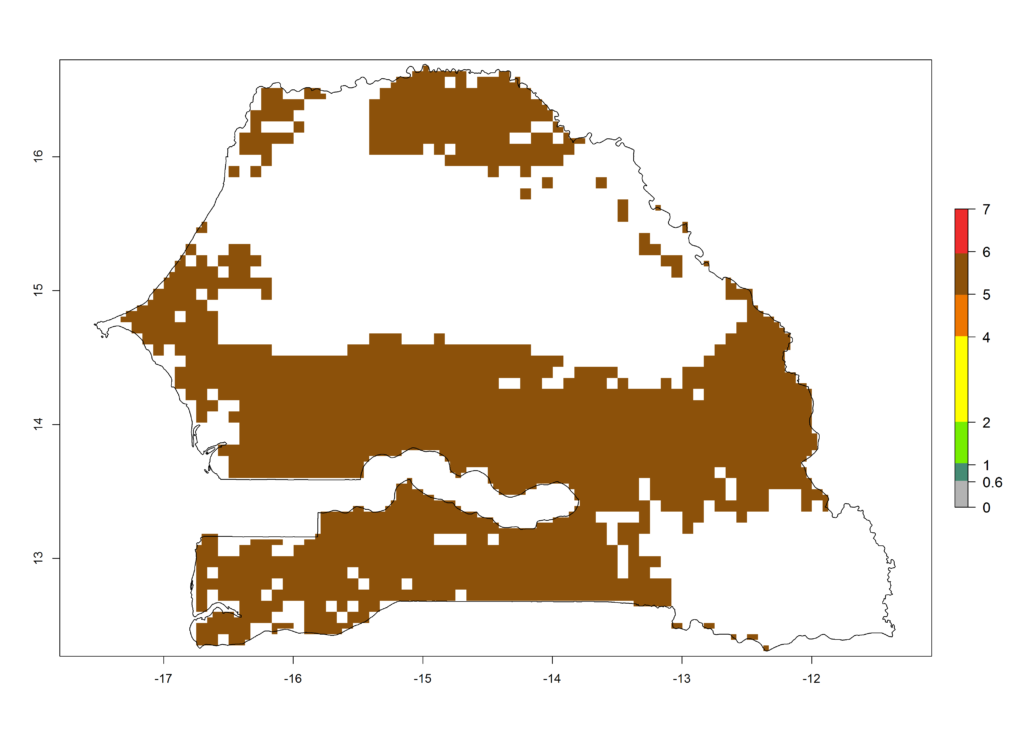 |
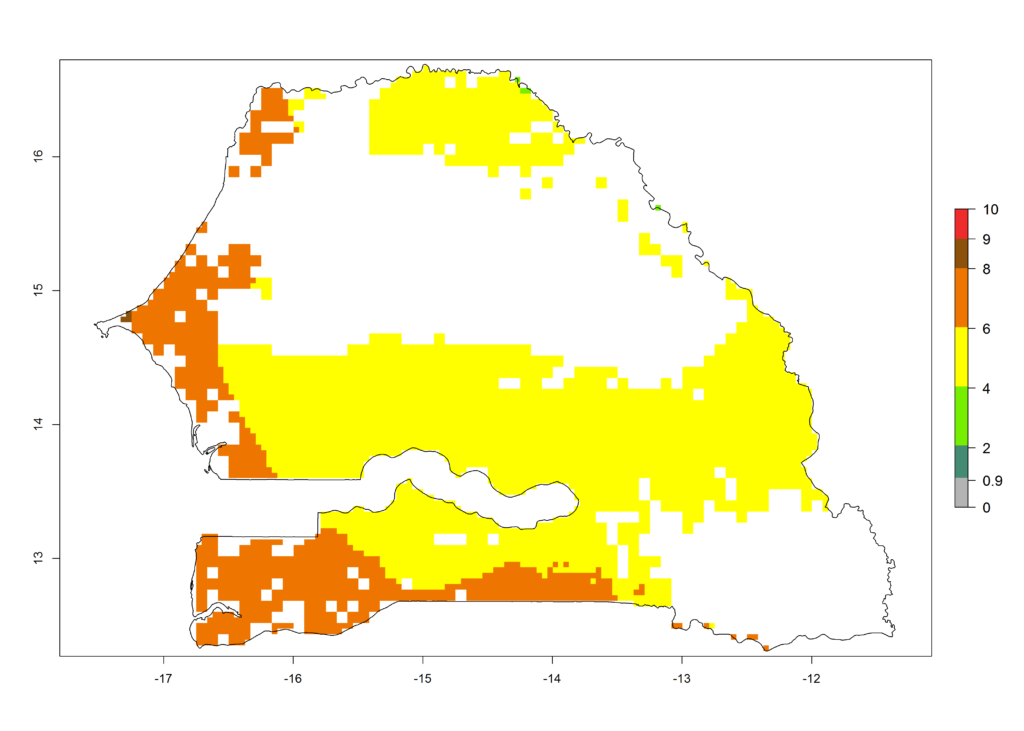 |
l) South Africa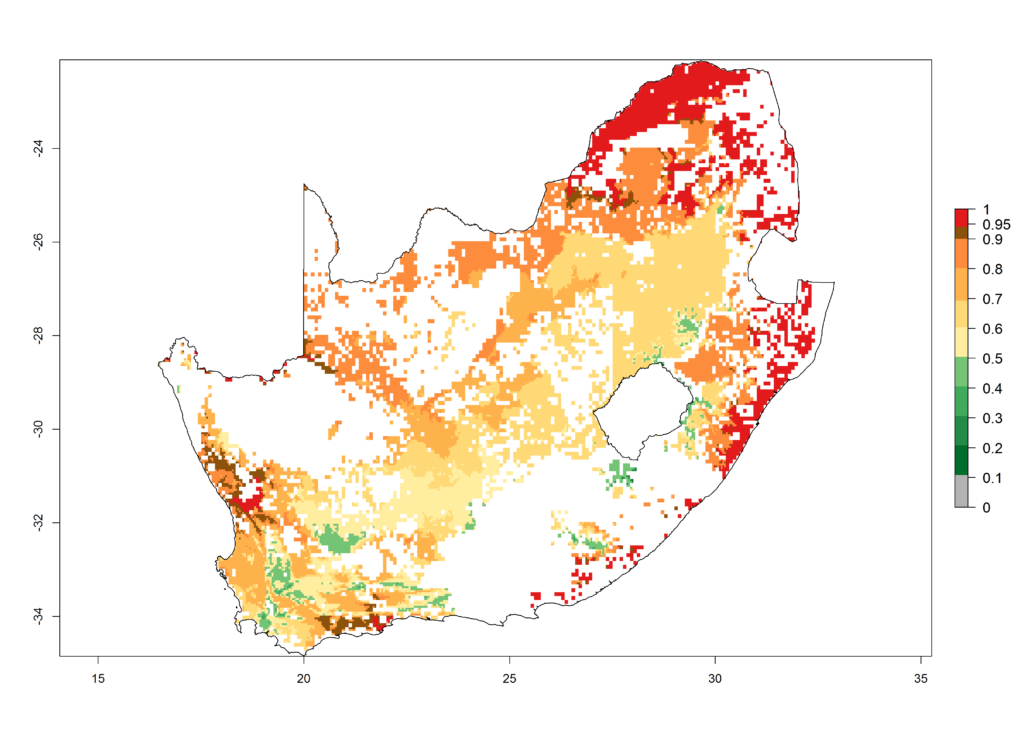 |
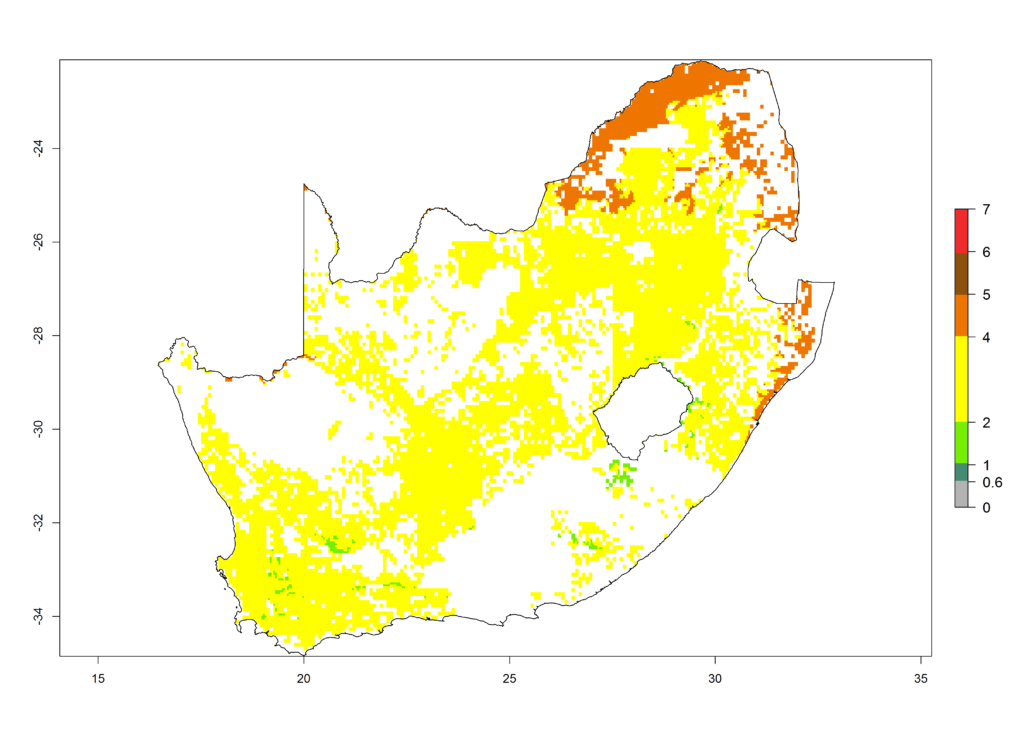 |
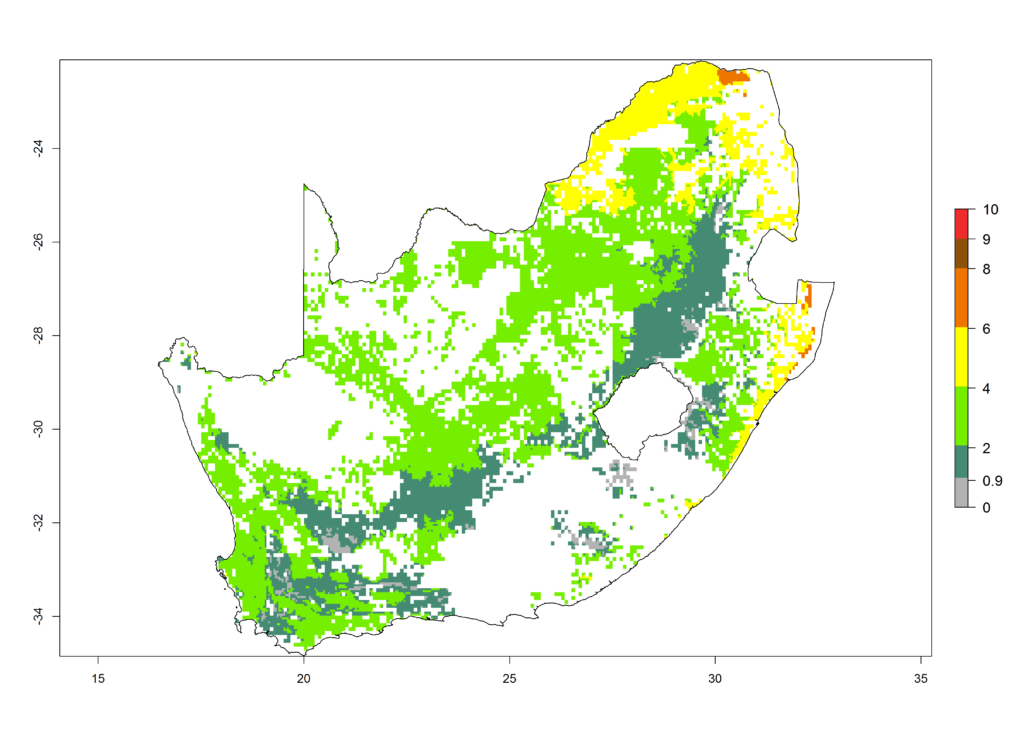 |
m) Sudan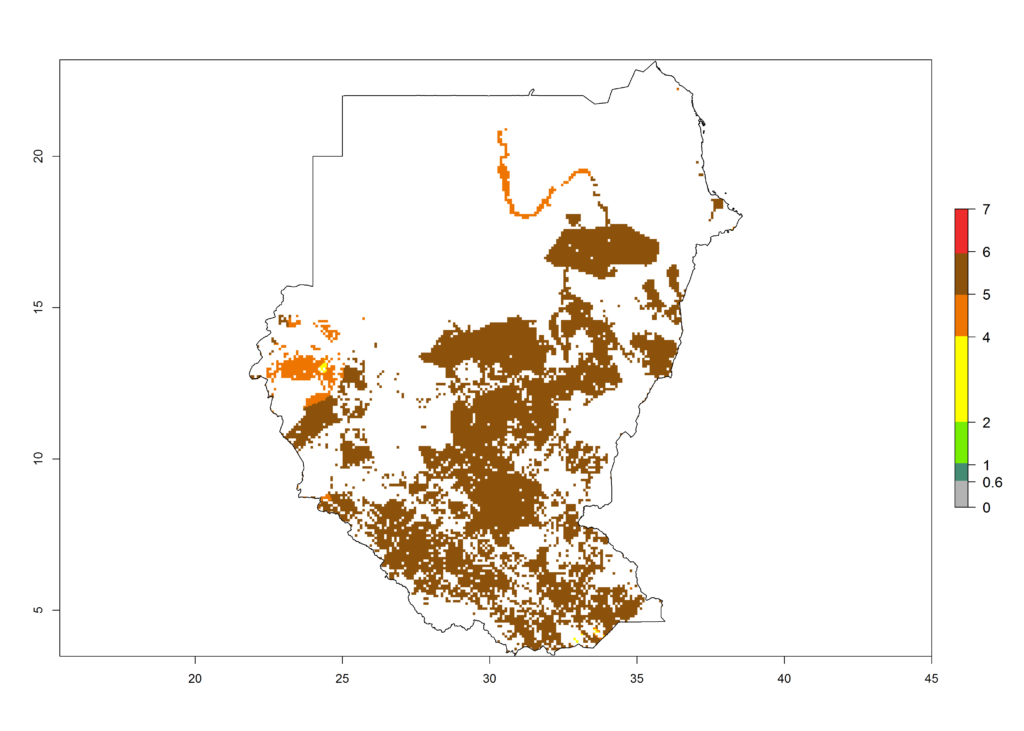 |
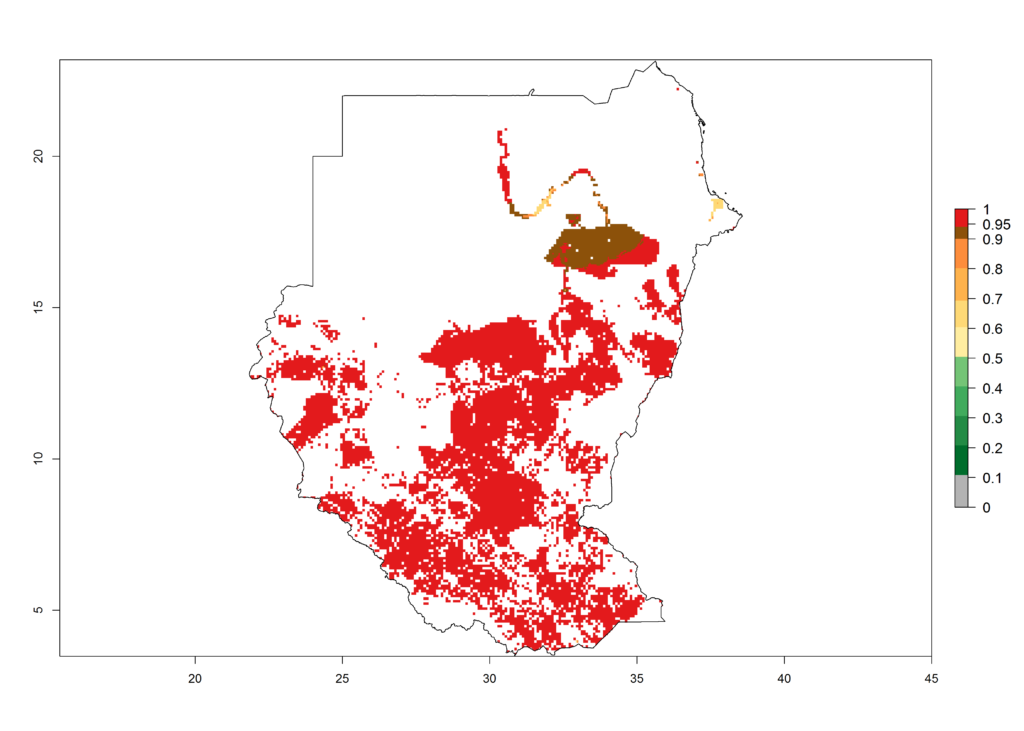 |
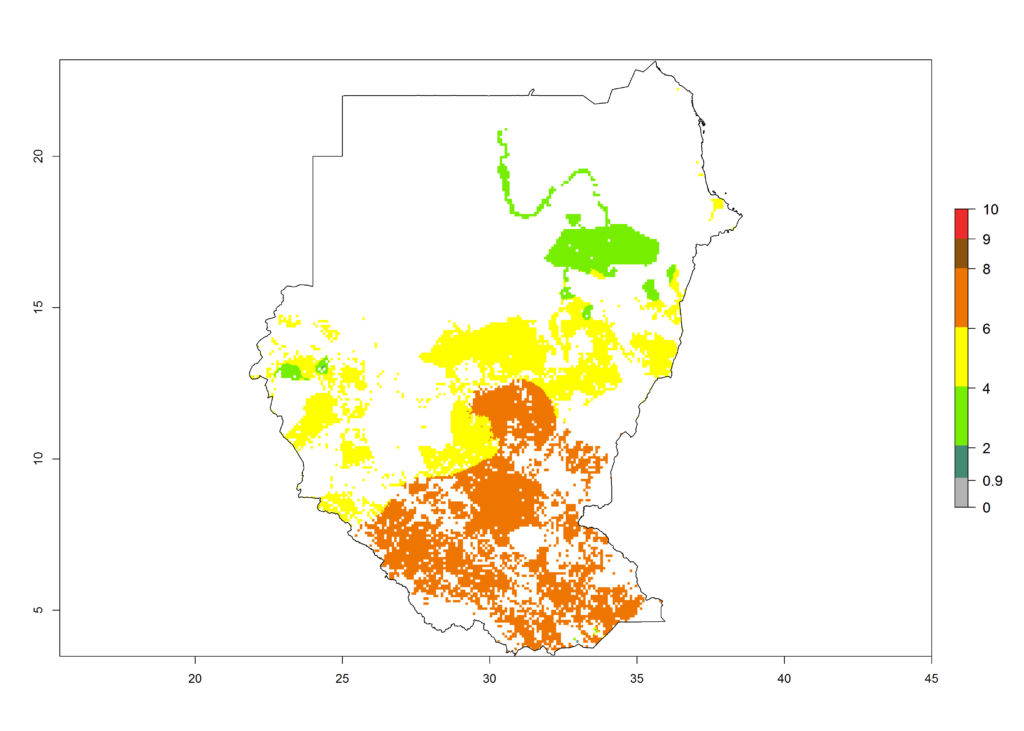 |
n) Tanzania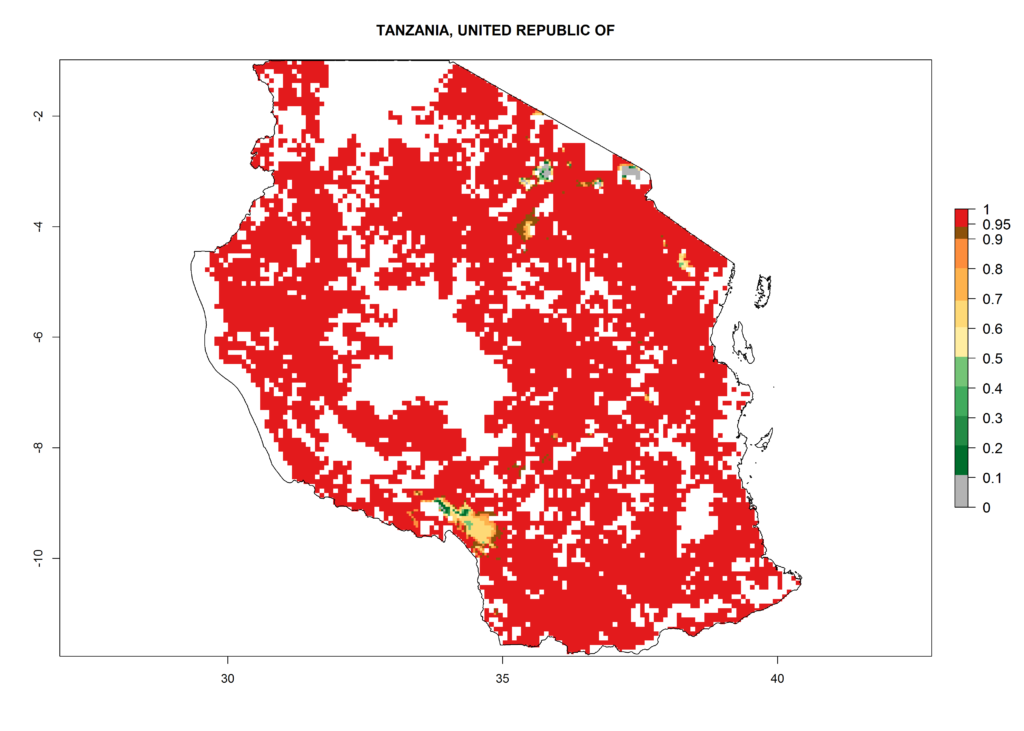 |
 |
 |
o) Uganda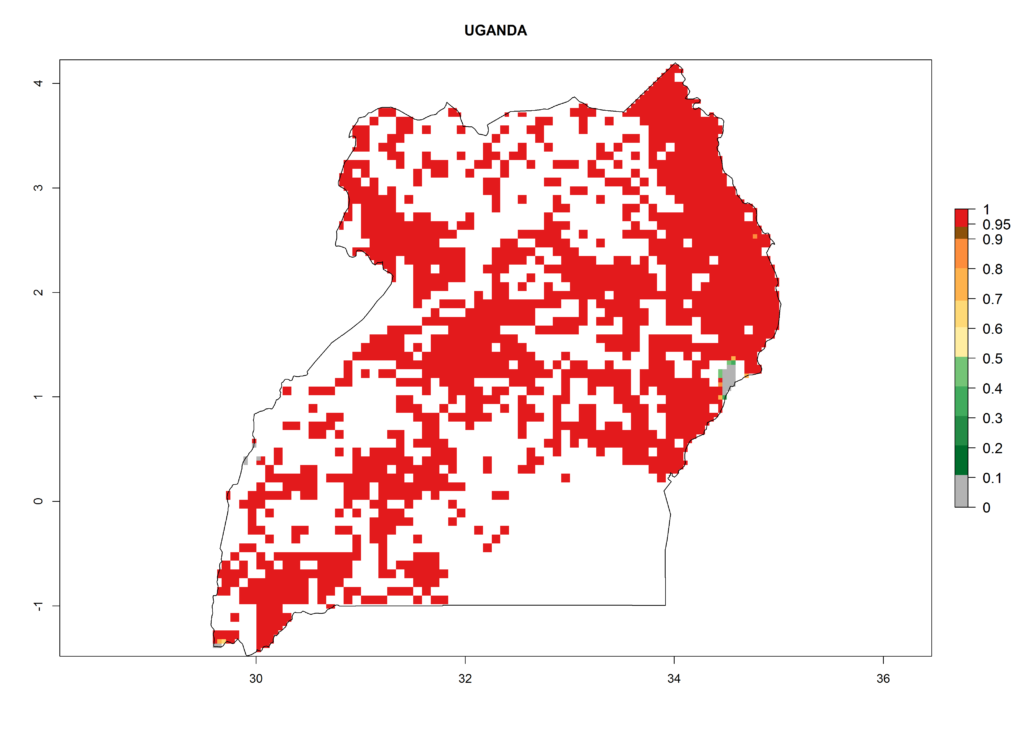 |
 |
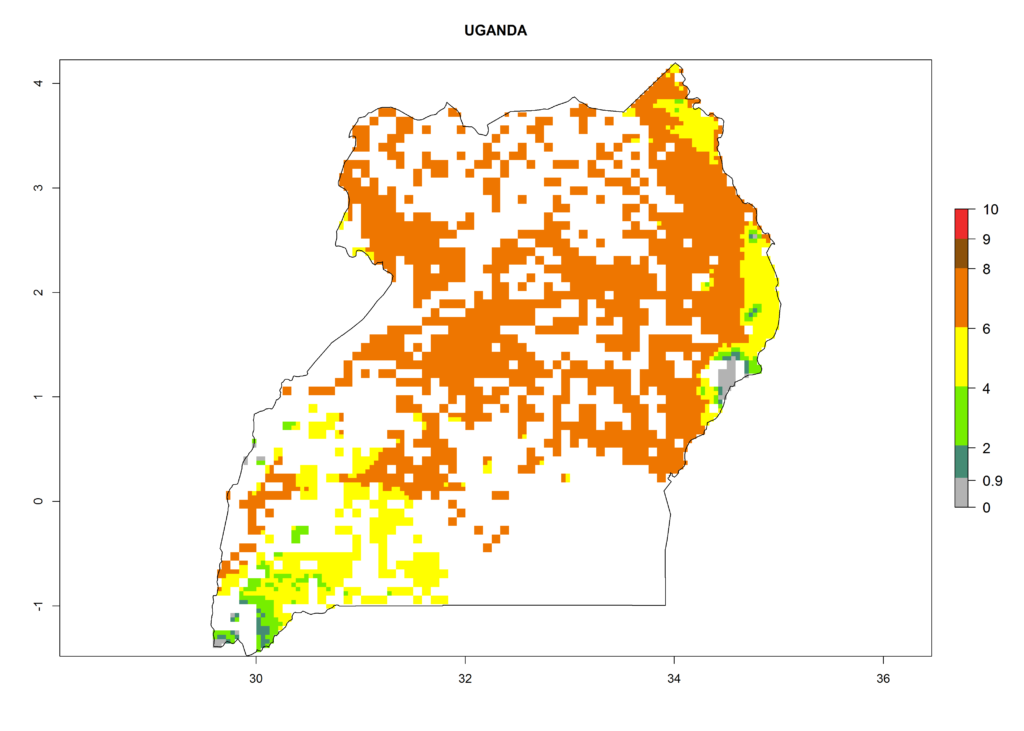 |
Figure 6. ERI, abundance (GI, potential damage), and activity (AI, potential population growth) of the African pink stemborer, Sesamia calamistis, in maize production systems of selected African countries according to model predictions in the year 2000. An ERI>0.6 is associated with potential permanent establishment.
Phytosanitary measures
Crop residue management. Placing stems horizontally on the ground after harvest to expose them to the sun’s heat kills larvae and pupae; spreading out the stalks thinly to dry can kill larvae. Complete burning of the stalks kills all stages of the pest.
Adaptation to risk avoidance at farm level
Monitoring pest populations. There is need to monitor agro-ecosystems to detect action threshold.
Biological control. For biological control, several species of parasitic wasps attack S. calamistis, but significant control has been observed by Cotesia sesamiae (on larvae), Descampsina sesamiae (on larvae and pupae), Pediobius furvus (on pupae), Sturmiopsis parasitica (on larvae and pupae), and egg parasitoids, Telenomus busseolae and T. isis. Habitat management practices that conserve these parasitoids should be practiced.
Cultural control. Cultural practices include intercropping maize with non-host crops like cassava and cowpea. Good crop hygiene (e.g., destruction of maize residues by burning to destroy larvae and pupae within the stems; removal of volunteer crop plants and/or alternative hosts) prevents carry-over populations. This helps to limit the initial establishment of stemborers that would infest the next crop.
Physical control. Physical measures include early slashing and drying of maize stubble, crop rotation, and burning.
Chemical control. Chemical control can be achieved by application of granules or dust to the leaf whorl early in crop growth. Low toxic insecticides should be used, considering all safety measures.
Further reading
Akol, A.M., M.Y. Chidege, H.A.L. Talwana, and J.R. Mauremootoo. 2011. Invertebrate pests of maize in East Africa (Kenya, Uganda and Tanzania), Lucid v. 3.5 key and fact sheets. Makerere University, TPRI, BioNET-EAFRINET, CABI & The University of Queensland. Available from keys.lucidcentral.org/keys/v3/EAFRINET.
Bosque-Pérez, N.A., and J.H. Mareck. 1990. Distribution and species composition of lepidopterous maize borers in Southern Nigeria. Bulletin of Entomological Research 80: 363–368.
CABI. 2014. Sesamia calamistis. Crop Protection Compendium. Available from http://www.cabi.org/cpc/.
ECOPORT. 2014. Sesamia calamistis. Available from http:/www.ecoport.org.
European Plant Protection Organisation (EPPO). 2014b. Data sheets on Sesamia calamistis. Available from http://gd.eppo.int/taxon/SESACA.
Khadioli, N., Z.E.H. Tonnang, G. Ong’amo, T. Achia, I. Kipchirchir, J. Kroschel, and B. Le Ru. 2014. Effect of temperature on the life history parameters of noctuid lepidopteran stem borers, Busseola fusca (Fuller) and Sesamia calamistis Hampson. Annals of Applied Biology 165(3): 373–386.
Kfir, R., W.A. Overholt, Z.R. Khan, and A. Polaszek. 2002. Biology and management of economically important lepidopteran cereal stem borers in Africa. Annual Review of Entomology 47: 701–731.
Ong’amo, G.O., B.P. Le Ru, S. Dupas, P. Moyal, P.A. Calatayud, and J.F. Silvain. 2006. Distribution, pest status and agro-climatic preferences of lepidopteran stem borers of maize in Kenya. Annales de la Société Entomologique de France 42: 171–177. doi:10.1080/00379271.2006.10700620.
Overholt, W.A., S.K. Kimani, J. Mbapila, P. Lammers, and E. Kioko. 1994. Ecological considerations of the introduction of Cotesia flavipes Cameron (Hymenoptera : Braconidae) for biological control of Chilo partellus (Swinhoe) (Lepidoptera : Pyralidae) in Africa. Biocontrol News and Information 15: 19–24.
Polaszek, A. ed. 1998. African cereal stemborers; economic importance, taxonomy, natural enemies and control. Wallingford, UK: CAB International, 530pp.
Warui, C.M., and J.N. Kuria. 1983. Population incidence and the control of maize stalk borers Chilo partellus (Swinh.) and Chilo orichalcociliellus Strand and Sesamia calamistis Hampson in Coast province, Kenya. Insect Science and its Application 4: 11–18.