4.3. 1 Vegetable Pests / Serpentine leafminer fly, Liriomyza huidobrensis (Blanchard 1926)![]()
Synonyms: Agromyza huidobrensis (Blanchard 1926)
Liriomyza cucumifoliae (Blanchard 1938)
Liriomyza decora (Blanchard 1954)
Liriomyza langei (Frick 1958)
Liriomyza dianthi (Frick 1951)
Taxonomic position: Insecta, Diptera, Agromyzidae
Authors: N. Mujica, P. Carhuapoma, & J. Kroschel
Common names
Serpentine leafminer, pea leafminer, South American leafminer, South American miner fly (English), Südamerikanische Minierfliege (German), mosca minadora, minadora sudamericana (Spanish), mouche mineuse sud-américaine (French), mosca minatrice sudamericana (Italian), larva mineira sul-americana (Portuguese)
Hosts
Liriomyza huidobrensis is highly polyphagous and has been recorded from 15 plant families. Host preferences vary according to location, production practices, and host availability. The major ornamental plants and crops affected are the following: Apiaceae: celery (Apium graveolens L.); Asteraceae: Aster spp., Chrysanthemum spp., Gerbera spp., Dahlia spp., Ixeris stolonifera, lettuce (Lactuca sativa L.), Lactuca spp., Zinnia spp.; Brassicaceae: cabbages (Brassica spp.); Caryophyllaceae: Gypsophila spp.; Chenopodiaceae: spinach (Spinacia oleracea L.), beetroot (Beta vulgaris L.); Cucurbitaceae: cucumbers (Cucumis spp.), pumpkins (Cucurbita spp.); Fabaceae: soybean [Glycine max (L.) Merr.], alfalfa (Medicago sativa L.), bean (Phaseolus vulgaris L.), pea (Pisum sativum L., Pisum spp.), Trifolium spp., faba bean (Vicia faba L.); Liliaceae: onion (Allium cepa L.), garlic (Allium sativum L.); Solanaceae: peppers (Capsicum annuum L., Capsicum frutescens L.), Petunia spp.; potato (Solanum tuberosum L.); tomato (Lycopersicon esculentum L.); tobacco (Nicotiana tabacum L.); eggplant (Solanum melongena L.); and wild species of the Solanaceae family, including important weeds (e.g., black night shade, Solanum nigrum L.).
Detection and identification
Adults and larvae of L. huidobrensis damage the plant foliage. The larva is primarily a leafminer; however, on peas the larvae also feed on the outer surface of young pods (Photo 1A, B). Feeding punctures and leaf mines are usually the first and most obvious signs of the presence of leafminer flies. Adults cause damage by puncturing the leaf surface to feed on the leaf tissue and also to lay eggs. Newly hatched larvae mine into the leaf and feed on the chloroplast-rich mesophyll, making a serpentine mine whose diameter increases as the larva grows. A large proportion of grown larvae remain close to the midrib. Leaf tissue affected by larval mining becomes necrotic and brownish. Time taken for damaged area to become necrotic depends on the variety, physiological conditions, and age of the plant. As necrotic areas coalesce in highly infested leaves, the whole leaf dries out and dies. Highly infested crop fields appear burned. The form and shape of a mine differ between host plants and depend on the physical and physiological condition of each leaf and by the number of larvae mining on same leaf.
|
|
|
Morphology
Egg
Eggs are round, translucent, and about 0.3 x 0.1 mm in size (Photo 2A).
Larva
First-instar larvae are colorless after hatching, turning to pale yellow-orange. Later instars are yellow-orange and the third-instar larvae measure 3.2 x 1 mm (Photo 2B). Mouth hooks remaining in the mines after each successive molt can be used to identify stages.
Pupa
The puparium is oval, slightly flattened ventrally, and varies in size (1.6–3.25 mm long, 0.7–1.1 mm wide) and color (light brown to almost black) (Photo 2C).

Adult
Adults are small (wing length: 1.7–2.3 mm), black flies with bright-yellow spots on the thorax (Photo 3). L. huidobrensis adults may be distinguished from other Liriomyza species because the yellow parts of the head and legs are darker orange-yellow, the third antennal segments are very dark, sometimes almost black on top, and the mesoplura is largely black.
|
|
Biology
Adult females feed on leaf tissue by puncturing the leaf surface with their ovipositors and ingesting the liquid cell contents. Females also puncture leaf tissue to lay their eggs. Both feeding and oviposition punctures turn white and damaged foliage looks stippled or speckled. After hatching eggs, each larva feeds on leaf tissue making a winding tunnel (serpentine leaf mine) or blotch on the leaf. The leaf mines become increasingly larger in size as the larvae feed and grow through three immature stages (instars). Once feeding is completed, mature larvae cut a slit in the leaf surface, exit the leaf, and drop to the ground where they pupate.
Temperature-dependent development
L. huidobrensis completed its development from egg to adult at temperatures of 10°–30°C but not at 32°C (see Annex 7.3.9). Leafminer total development was almost five times longer at 10ºC (65.5 days) than at 30°C (14.9 days). Estimated lower threshold for development of immature stages was 6.4°C (egg), 3.4°C (larva), and 4.8°C (pupa). For all three immature stages, lowest mortality occurred at 20°C (32.4%) and highest at 30°C (97.3%). At 32°C all pupae died before adults emerged. Pupae were more vulnerable to unfavorable temperatures compared with other stages. Optimal temperature for survival ranged 20°–25°C (32–38% survival of the entire immature stage). Significant differences were observed in the longevity between male and female adults, with female longevity twice as long as males at temperatures >10°C. The lowest senescence rate was observed within the temperature range of 10°–20°C. Highest fecundity is found at around 22°C.
The established functions (see Annex 7.3.9) were used to estimate the life-table parameters of L. huidobrensis and to build an overall stochastic phenology model. Population parameters indicate that the intrinsic rate of natural increase (rm) augments almost linearly with increasing temperature to reach a maximum at 22°C (0.037) and then sharply decreased at 25°C (0.022). The negative r values below 14°C and above 25°C indicate that population size is decreasing due to high mortality and no or very low reproduction. Similarly, the finite rate of increase for leafminer fly peaked at 22°C (λ=1.038) and was smallest when exposed to 12°C (λ=0.991) and 26°C (λ=0.993); λ values of <1 indicate that the population is decreasing. The highest values for both reproductive parameters (gross reproductive rate and net reproductive rate, Ro) were found at 20°–22°C. The shortest mean generation time (T) and doubling time (Dt) were observed at 25°C (19.04 days) and 22°C (18.6 days), respectively. The optimum temperature for overall population growth ranged from 20°C to 25°C.
Deterministic simulation of life-table parameters under prevailing temperature conditions of two contrasting agro-ecological Peruvian zones showed that lowland agro-ecologies (La Molina: 12º 05′ S, 76º 57′ W, 250 masl, with a mean annual precipitation of 6.4 mm and mean temperature of 19.7ºC) present more suitable temperature conditions than do the highlands (Huancayo: 12º 00′ S, 75º 22′ W, 3,250 masl, with a mean annual precipitation of 646 mm and temperature of 12.2ºC) for leafminer fly population increase in all evaluated seasons. For the lowland (La Molina) and highland (Huancayo) agro-ecologies, a T of 12.2 and 7.2 generations per year were determined.
Means of movement and dispersal
Little information exists on the natural dispersal of L. huidobrensis. Adults are capable of short-distance flights only, but winds can support long-distance flights into crops from surrounding vegetation. Trade is a key factor in the rapid spread and global distribution of Liriomyza species, particularly via infested host-planting material and cut flowers, which are the main means of long-distance dispersal.
Economic impact
L. huidobrensis is a serious pest of arable crops, vegetables, and ornamental plants under field and glasshouse conditions in many parts of the world. Plant injuries caused by adult and larval activities reduce photosynthesis activity and cause leaf wilting. Both crop yield and marketability yield are reduced, resulting in high economic losses to vegetable producers around the world. Losses in production of spinach, lettuce, and celery greater than 50% have been reported. For potato, yield losses of almost 100% were reported in Argentina, Chile, and Indonesia. In Peru, yield losses varied between potato varieties and early-maturing varieties (yield losses of 50% to >60%) are more affected than late-maturing ones (yield losses of 20–30%). The polyphagous nature of L. huidobrensis, combined with high reproductive rates and rapid development of insecticide resistance, contributed to the success of L. huidobrensis as an invasive species.
Geographical distribution
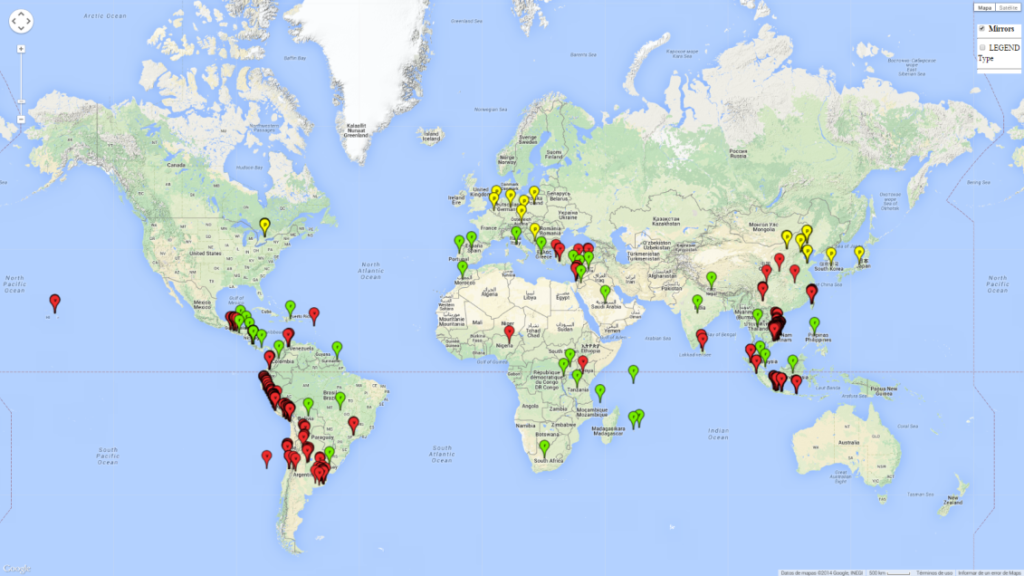
The pea leafminer is an agricultural pest endemic to South America. Since the early 1980s, L. huidobrensis has been recorded in numerous countries around the world, presumably associated with the global trade of ornamental plants (Fig. 1).
| Africa | Union of the Comoros, Kenya, Mauritius, Morocco (restricted distribution), Reunion, Seychelles, South Africa (restricted distribution), Spain (Canary Islands), Tanzania, Uganda |
| Asia | China (in the provinces of Chongqing, Fujian, Gansu, Guangdong, Guizhou, Hebei, Hubei, Nei Menggu, Shaanxi, Shandong, Sichuan, Xinjiang, Yunnan), India (Uttar Pradesh), Indonesia (Java, Sulawesi, Sumatra), Israel (widespread), Japan (Hokkaido, Honshu), Jordan, Korea Dem People’s Republic, Lebanon, Peninsular Malaysia, Philippines (widespread), Saudi Arabia, Singapore, Sri Lanka, Syria Arab Republic, Taiwan (widespread), Thailand, Vietnam |
| Europe | Austria (protected crops), Belgium (transient, protected crops), Bulgaria (under eradication), Croatia, Cyprus (widespread), Czech Republic (protected crops), Denmark (eradicated), Finland (transient, under eradication), France (restricted distribution), Germany (protected crop), Greece (widespread), Hungary (eradicated), Ireland (eradicated), Italy (restricted distribution), Lithuania (eradicated), Malta (protected crops), Montenegro, the Netherlands (protected crops), Norway (eradicated), Poland (protected crops), Portugal (widespread), Serbia, Slovenia (eradicated), Spain (restricted distribution), Switzerland (protected crops), Turkey (restricted distribution), UK (eradicated) |
| North America | Canada (Ontario) |
| Central America and the Caribbean | Belize, Costa Rica, Dominican Republic, El Salvador, Guadeloupe, Guatemala, Honduras, Mexico, Nicaragua, Panama |
| South America | Argentina, Brazil (Goias, Minas Gerais, Sao Paulo), Chile, Colombia, Ecuador, French Guyana, Peru, Uruguay, Venezuela |
| Oceania | Australia (absent, intercepted only) |
Phytosanitary risks
L. huidobrensis is primarily a tropical and warm temperate species that has been found up to 3,400 masl in tropical regions. In Europe it has become a major pest of a wide variety of ornamental or vegetable crops grown under glass. It is a major quarantine pest and is officially listed as an A2 pest since its introduction into several northern European countries (EPPO 2013).
Risks mapping under current and future climates
Global Risks
Changes in establishment and future distribution
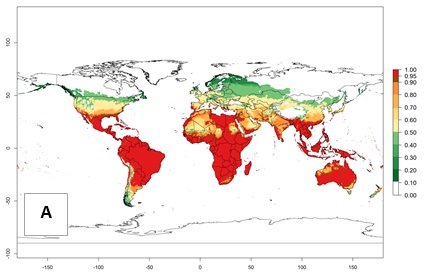
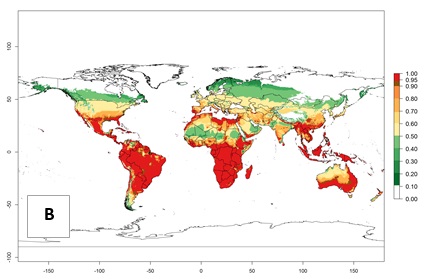
An establishment risk index (ERI)>0.95 reflects well the current global distribution of L. huidobrensis under the current climate of the year 2000 (Fig. 2). Those regions include Central and South America; Africa (eastern countries of Kenya, Uganda, Rwanda, and Tanzania, as well as some areas of Morocco); Southeast Asia (Indonesia, Taiwan, Vietnam, Philippines); South Asia (Nepal and India); Middle East (Israel and Jordan); southern region of China (subtropical provinces of Yunnan and Fuijang); and southern European countries such as Portugal (Algarve), Spain (Almeria), and Italy (Sicilia) (compare with Fig. 1).
 |
 |
 |
|
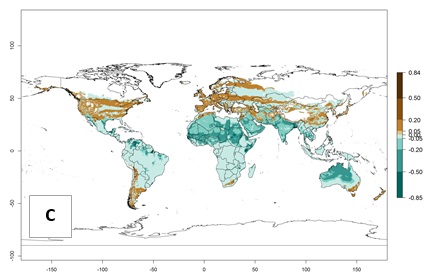
Figure 2. Changes in establishment and potential distribution of the serpentine leafminer fly, Liriomyza huidobrensis, worldwide according to model predictions, using the ERI for the years 2000 (A) and 2050 (B), and changes of the ERI between 2000 and 2050 (C). An ERI>0.7 is associated with potential permanent establishment.
In zones where the ERI drops below the maximum number of 1, the likelihood of long-term establishment is reduced, but L. huidobrensis also occurs in regions with an ERI of 0.7–0.9 (light- and dark-orange zones) as in central region of China (Sichuan and Hubei), southern region of Africa (South Africa), and some areas of southern European countries (Greece, Turkey, France). L. huidobrensis has also been reported in regions with a low likelihood of establishment (ERI<0.6) as in temperate regions of China (Beijing and Inner Mongolia); some areas of Korea Democratic People’s Republic or Japan (Hokkaido and Honshu); northern and central European countries; and southern Canada (Ontario). Under these conditions, L. huidobrensis survives unfavorable conditions under natural conditions or continues its development in protected production systems. Reported occurrences are due to survival and infestations in greenhouses, not to survival under open-field conditions.
Global predictions for 2050 indicate that L. huidobrensis will continue to be a high-risk pest in most of the tropical and subtropical areas (Fig. 2B). However, a decrease in the high risk of establishment (ERI>0.8) is predicted in South Asia (India); regions in North and West Africa; Middle East (Saudi Arabia and Yemen); and Australia. A slight range expansion is observed in more temperate areas such as western, central, and eastern Europe, North America (northern U.S. and southern Canada), and western Pacific (Japan and Korea) (Fig. 2C), but still with a low establishment potential of the pest (ERI<0.6). Further, it is predicted that L. huidobrensis will potentially shift further into southern South America (Chile and Argentina).
Changes in abundance
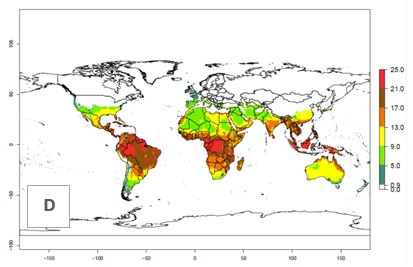
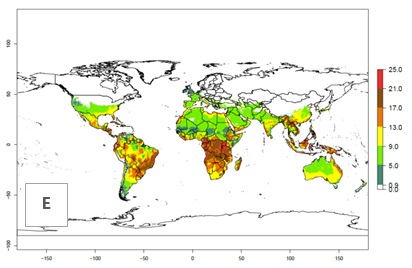
Figures 3A and B illustrate the mean numbers of generations that might theoretically develop worldwide within a year in the years 2000 and 2050 scenarios.
| GI | AI | |
| 2000 | 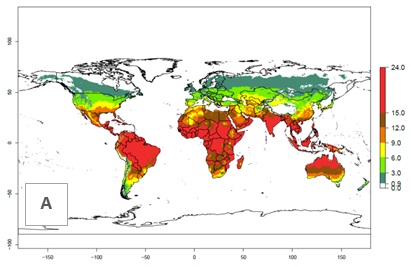 |
 |
| 2050 | 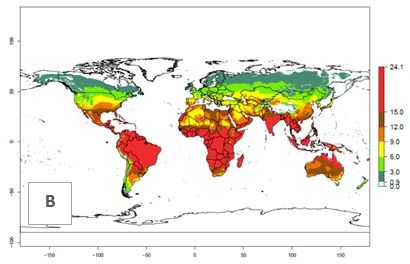 |
 |
| Index change (2000 – 2050) | 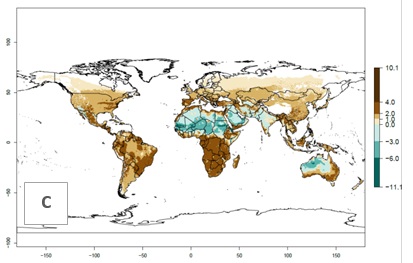 |
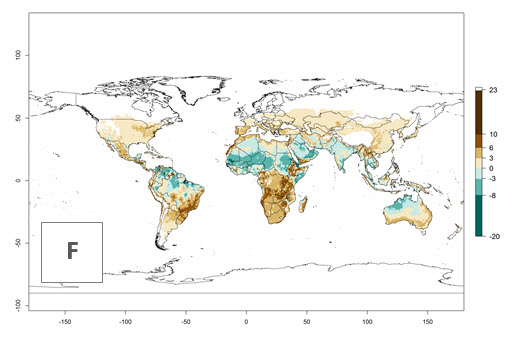 |
Figure 3. Changes in abundance (generation index [GI], damage potential) and activity (activity index [AI], potential population growth) of the serpentine leafminer fly, Liriomyza huidobrensis, worldwide according to model predictions, using the GI (A, B) and the AI (D, E) for the years 2000 and 2050, and the absolute index change (C, F).
The generation index (GI) change indicates that in most subtropical and tropical regions, an increase of 2–4 generations per year can be potentially expected, as in Central and South America; regions in East and Southern Africa; the Middle East (Yemen); and southern Europe (Portugal and Spain) (Fig. 3C). In temperate regions such as North America, southern South America, central and east Asia, and Europe (except southern countries), only a slight increase of 1–2 generations per year is expected. By contrast, a decrease of 1–3 generations per year is estimated for some areas in the Middle East (Arabia Saudi, Iraq, Iran) and in South (India and Pakistan) and Central Asia (Turkmenistan); and among 3–6 generations per year in some areas of West and North Africa.
Globally, simulated GIs gave reasonable predictions when compared with generation numbers reported in the literature. For example, in the Peruvian localities of La Molina (central lowland, 500 masl, mean annual temperature of 20ºC) and Huancayo (central highland 3,300 masl, mean annual temperature of 12ºC), mean numbers of 12 and 7 generations per year, respectively, have been reported. Estimations made by the GI are consistent with these data. Change of L. huidobrensis GI between 2000 and 2050 scenarios shows that the number of leafminer fly generations per year will increase more rapidly in La Molina (2–4 more generations per year) in comparison with the cold climates of Huancayo, with a predicted increase of less than 1 generation per year.
Global maps of the activity index (AI) in year 2000 estimates a high activity of L. huidobrensis by a factor of 13–25 in most tropical regions, and by a factor of 5–13 for most subtropical regions of South America, East and Central Africa, and Southeast Asia (Fig. 3D). Predictions of potential changes for the 2050 temperature scenario indicate a decrease in the potential growth of L. huidobrensis in tropical regions of Central and South America, Africa, and Asia (Fig. 3D, F). In most of the subtropical and temperate regions, the AI increases from the years 2000–2050 by values up to 3.
Regional Risks for Africa
Changes in establishment and future distribution
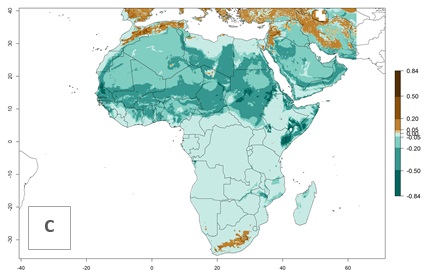
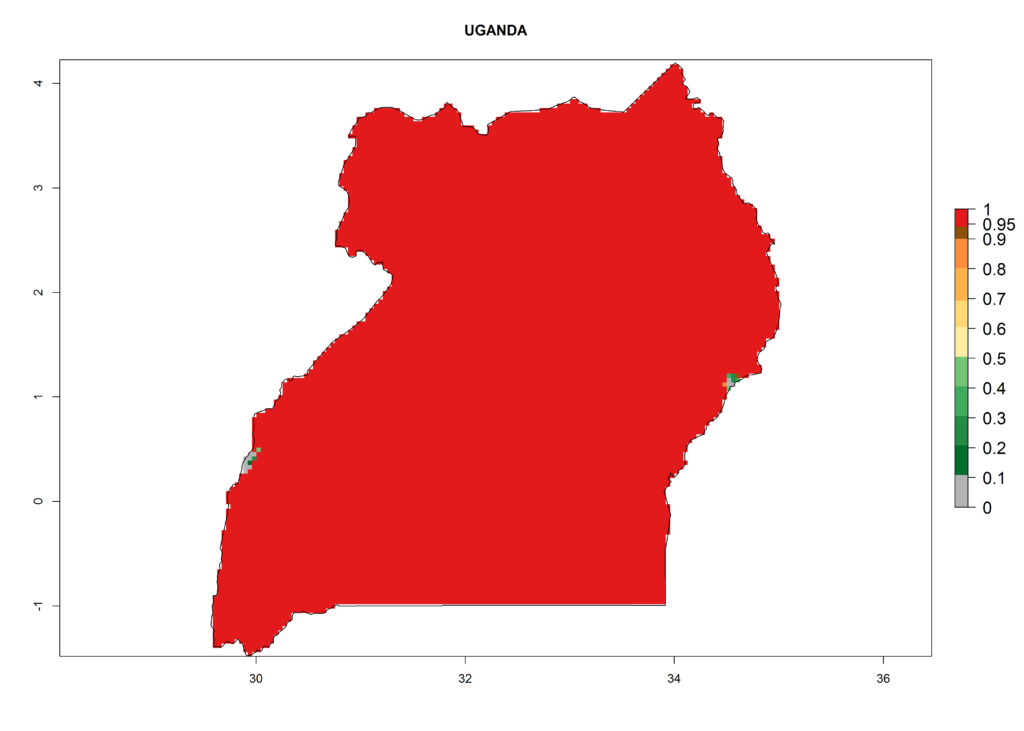
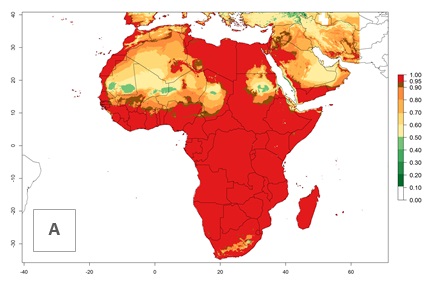
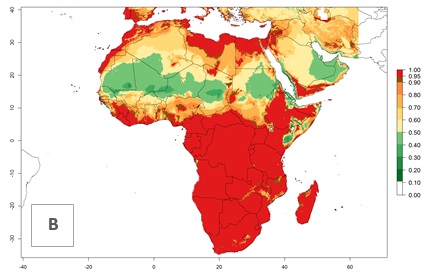
In Africa, L. huidobrensis is already present in Kenya, Rwanda, Uganda, and Tanzania, with an ERI>0.95 (Fig. 4A). The pest also represents a high risk (ERI>0.95) for production areas of other countries of East, Central, and Southern Africa. However, L. huidobrensis is already distributed under open-field conditions in regions with an ERI>0.7–0.9 (light- and dark-orange zones) as in some areas of South Africa and Morocco. Under the year 2050 temperature scenario, a slight decrease (<-0.1) in most of the eastern, central, and southern regions is observed, but L. huidobrensis will remain a potential risk and threat for food production. A high decrease (-0.2 to -0.5) in the risk of establishment is predicted in most of the countries in West, North (Sudan, Egypt, Libya), and East (Somalia) Africa. By contrast, an increase in the spread of L. huidobrensis (0.1–0.3) with a high establishment potential (ERI>0.7) is predicted for some areas of South Africa and Mediterranean countries (Morocco and northern Algeria) (Fig. 4B, C).
|
|
Figure 4. Changes in establishment and potential distribution of the serpentine leafminer fly, Liriomyza huidobrensis, in Africa according to model predictions, using the ERI for the years 2000 (A) and 2050 (B), and changes of the ERI between 2000 and 2050 (C). An ERI>0.7 is associated with potential permanent establishment.
Changes in abundance
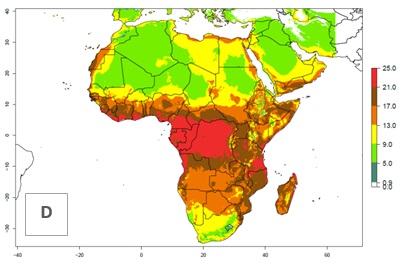
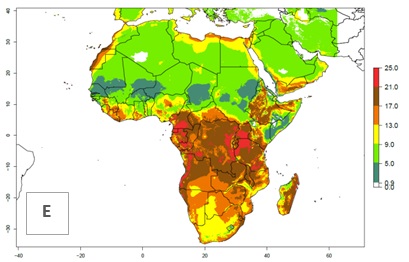
Under the present climate scenario, the GI indicates 15–24 generations of L. huidobrensis per year in most countries of East, West, and Central Africa (Fig. 5A). In northern and southern countries, the numbers of generations predicted for the leafminer fly range mainly from 6 to 12. In Africa the highest increase in the number of L. huidobrensis generations (1–5 per year) is predicted for most sub-Saharan countries and for the Mediterranean region (Fig. 5C). It is estimated that in Africa the change of activity between 2000 and 2050 will be concentrated in the eastern and southern regions (Fig. 5E, F). A population growth potential by a factor of 1–5 is expected in Ethiopia, Uganda, Kenya, Rwanda, Tanzania, Zambia, Angola, Namibia, Zimbabwe, South Africa, Lesotho, and Madagascar.
| GI | AI | |
| 2000 | 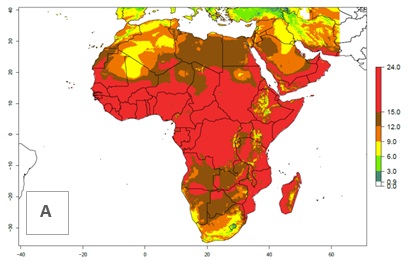 |
 |
| 2050 | 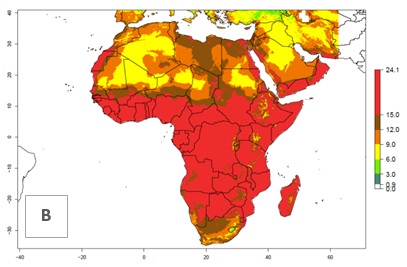 |
 |
| Index change (2000 – 2050) | 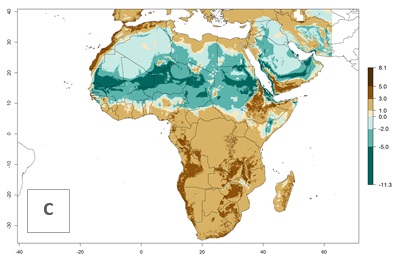 |
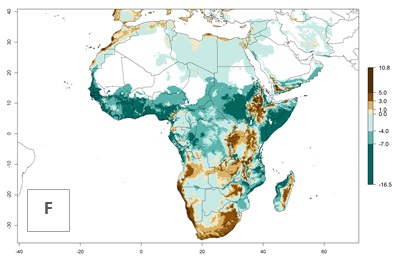 |
Figure 5. Changes in abundance (GI, damage potential) and activity (AI, potential population growth) of the serpentine leafminer fly, Liriomyza huidobrensis, in Africa according to model predictions, using the GI (A, B) and the AI (D, E) for the years 2000 and 2050, and the absolute index change (C, F).
Country Risk Maps
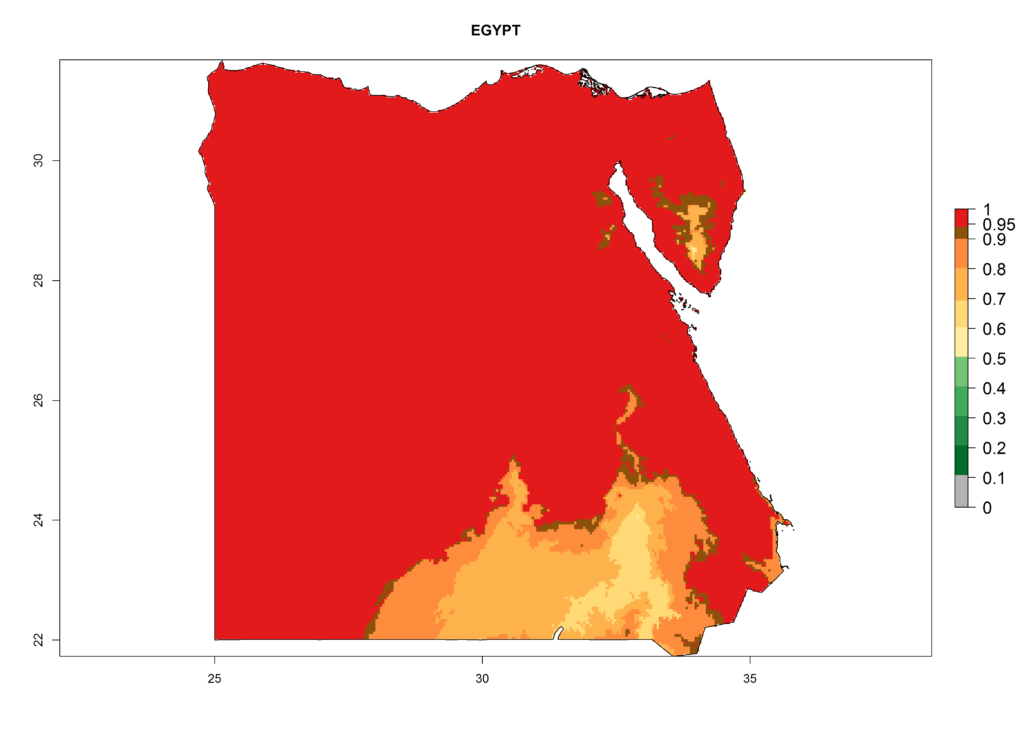
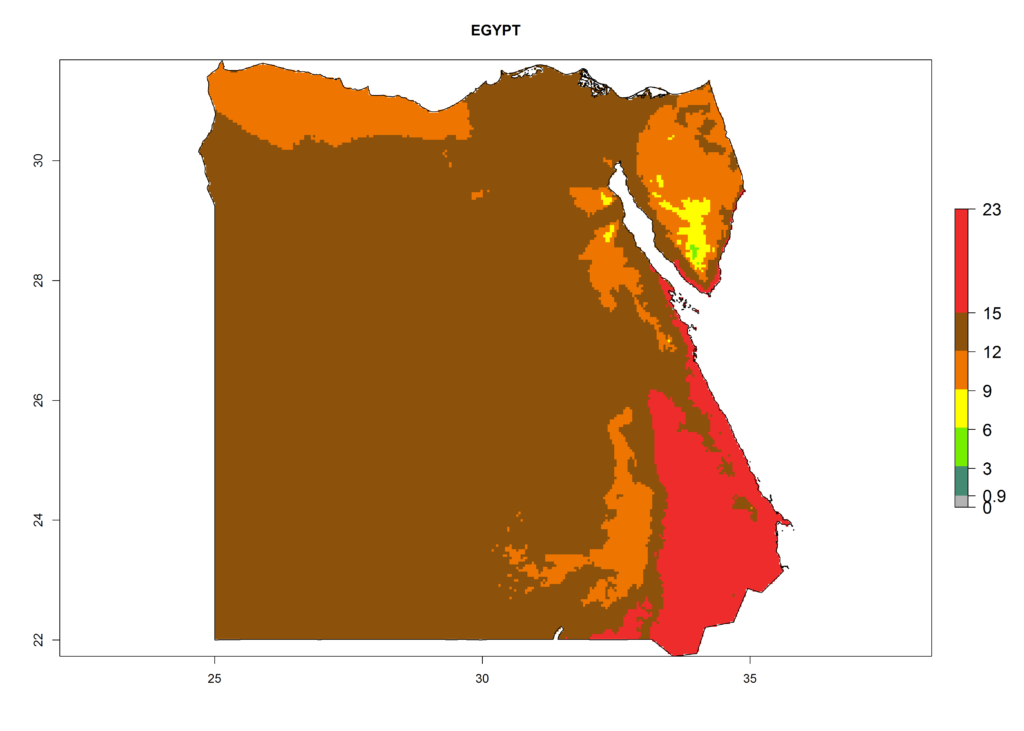
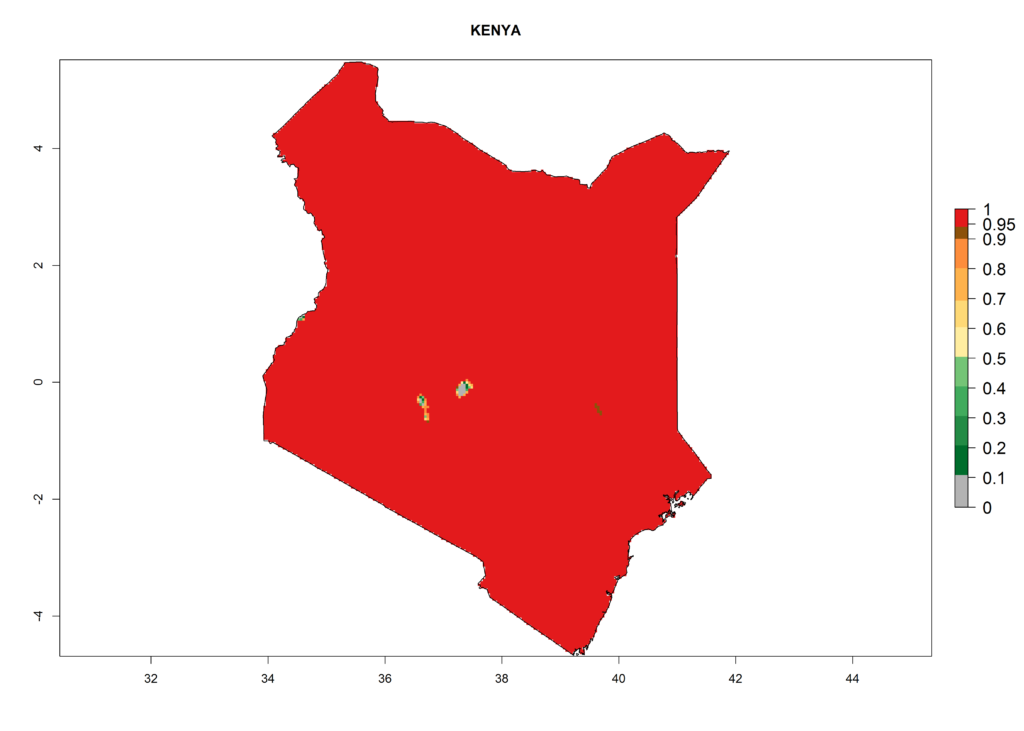
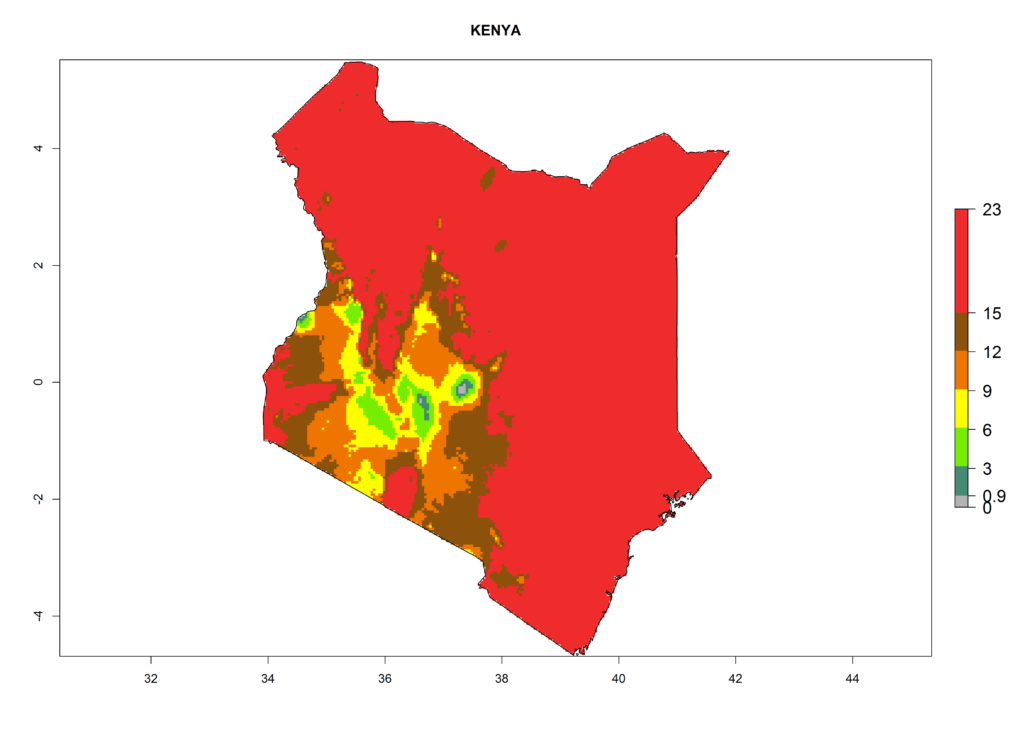
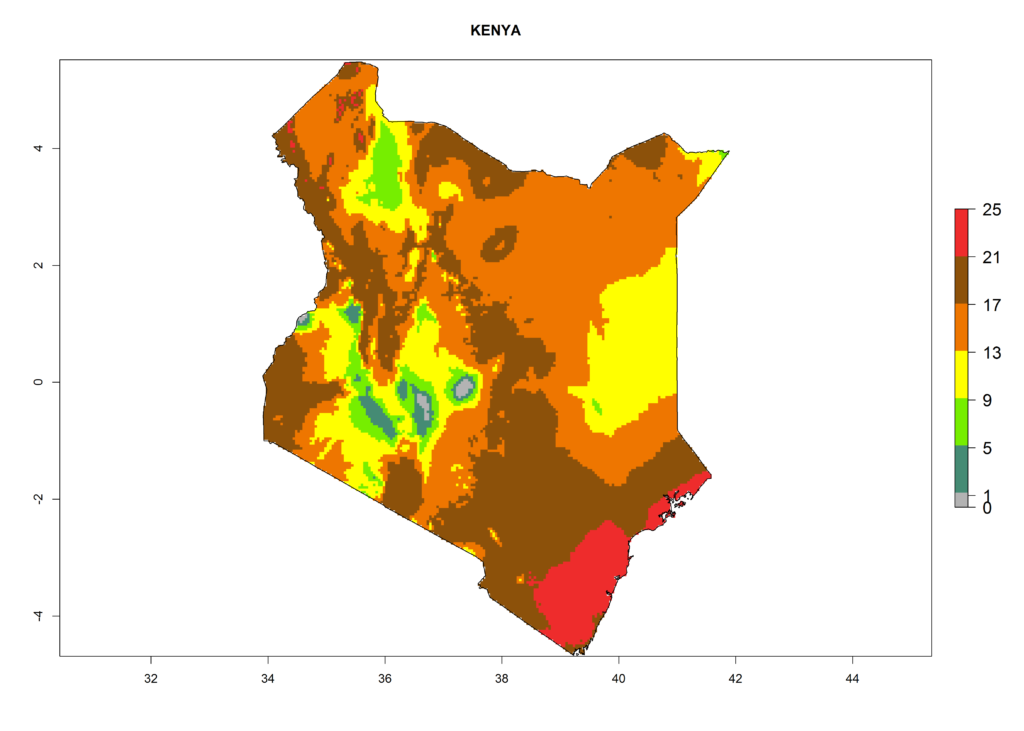
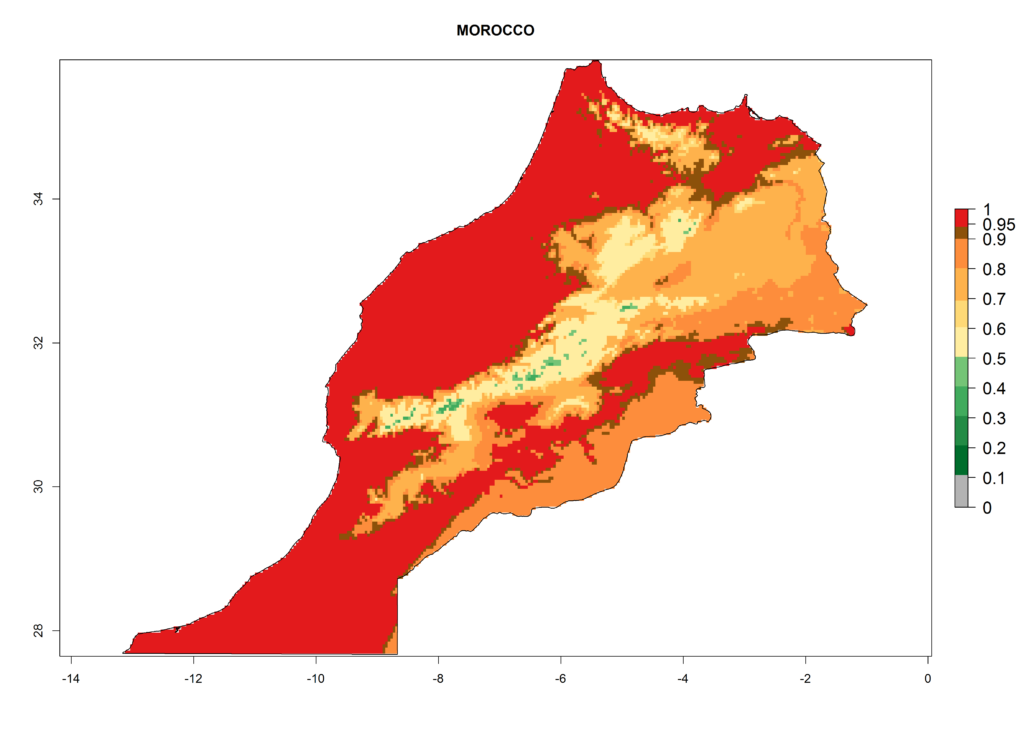
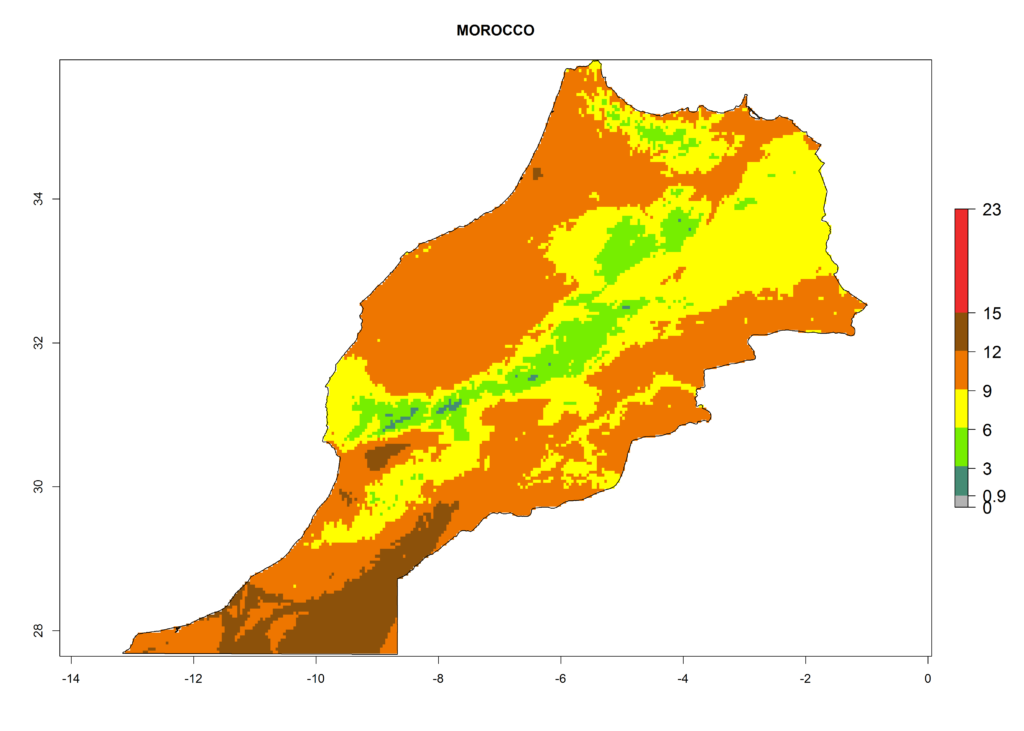
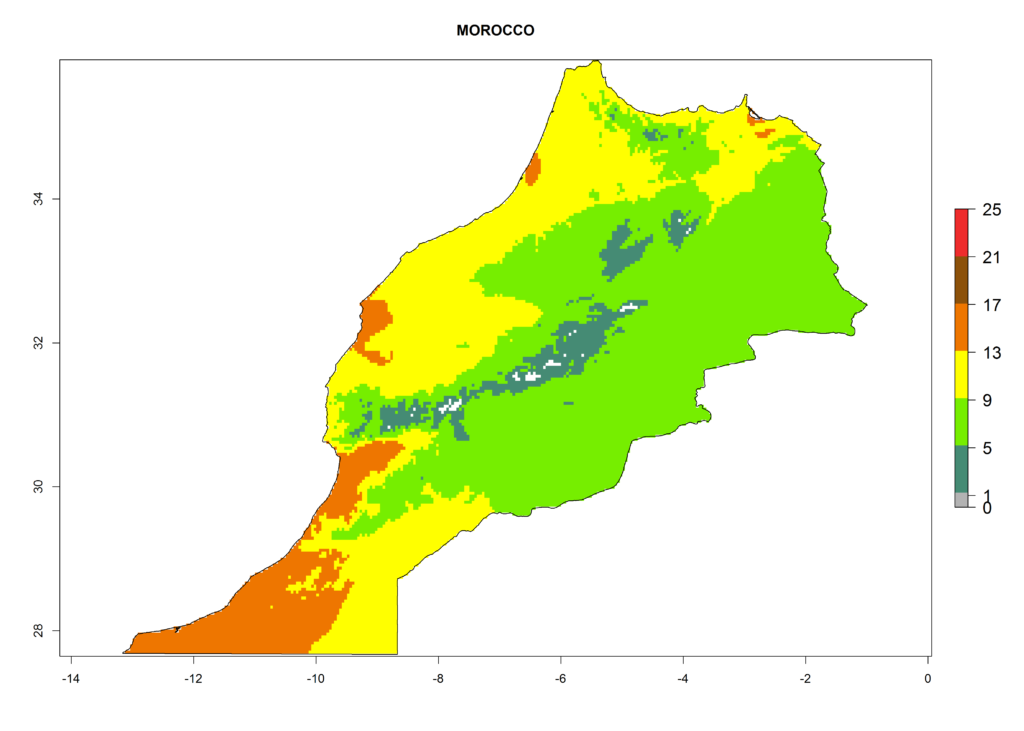
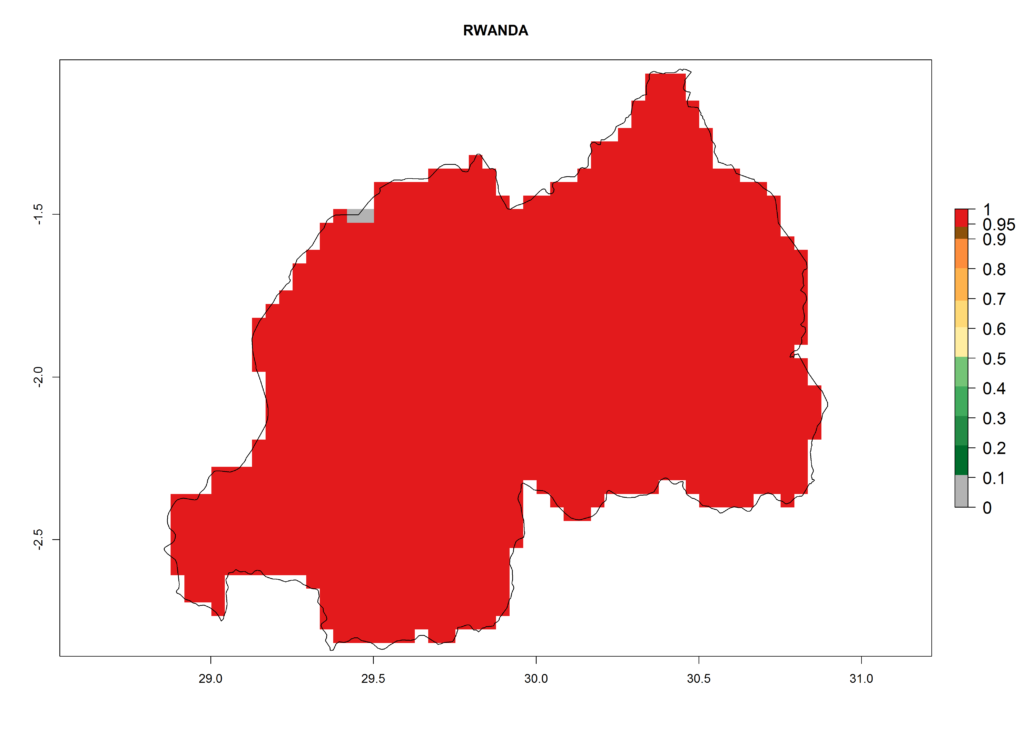
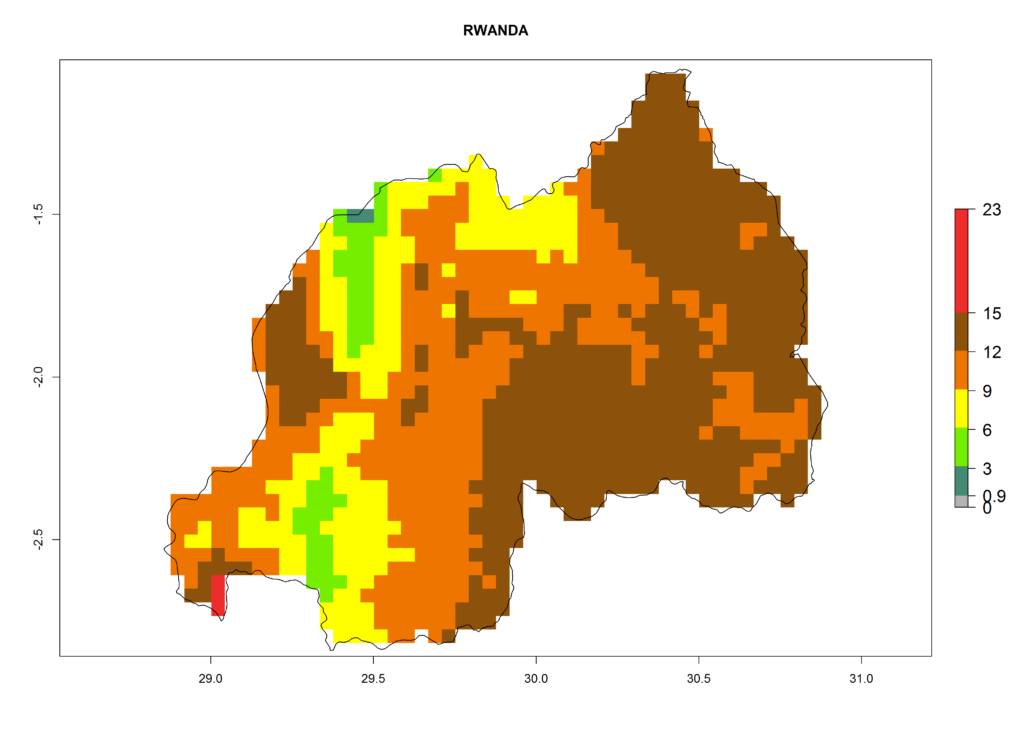
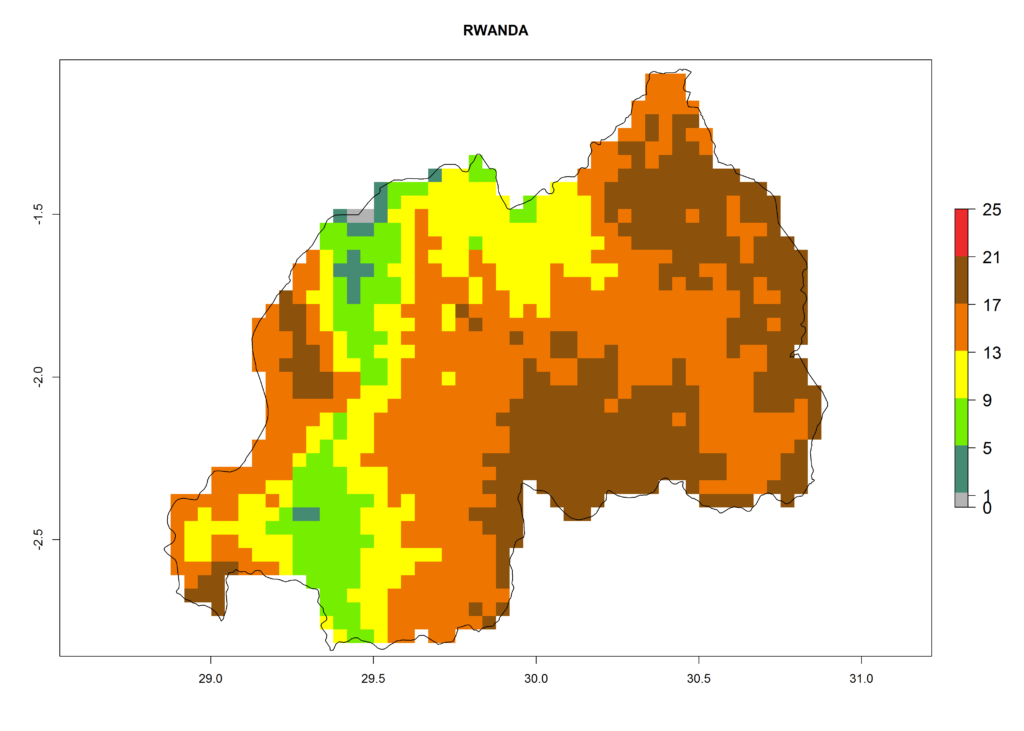
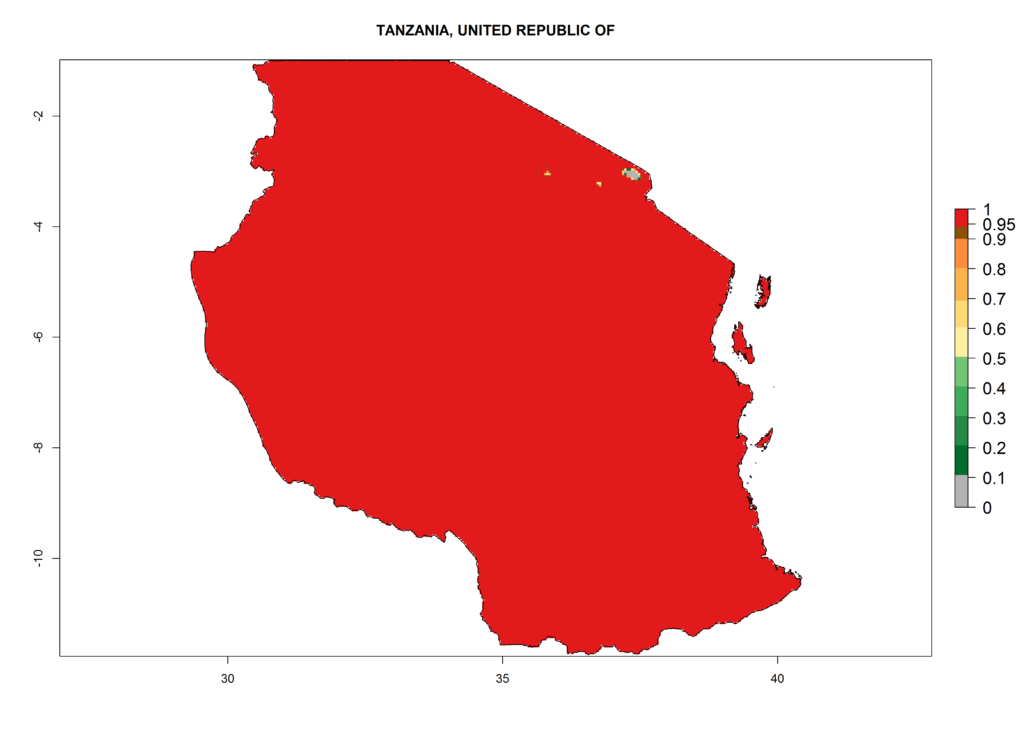
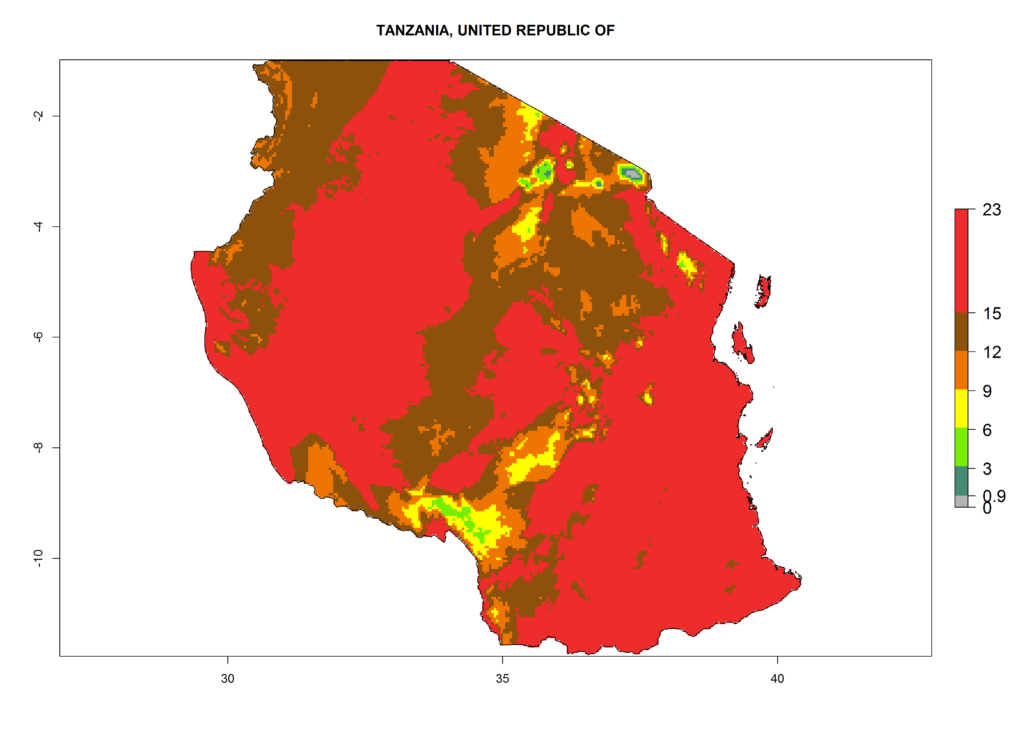
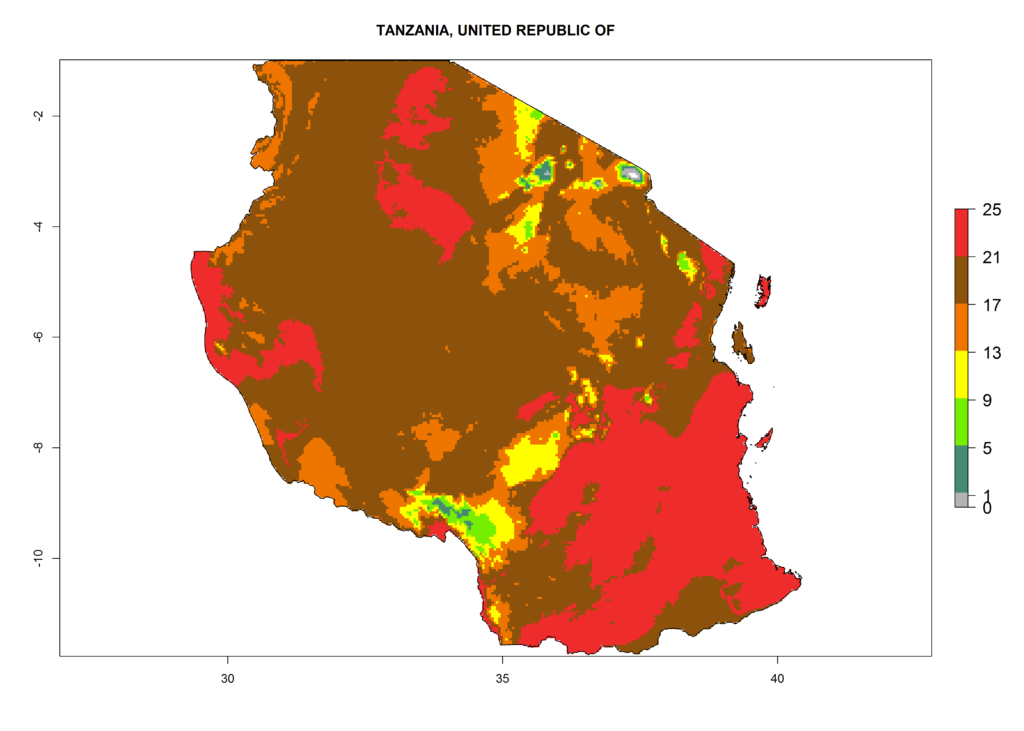
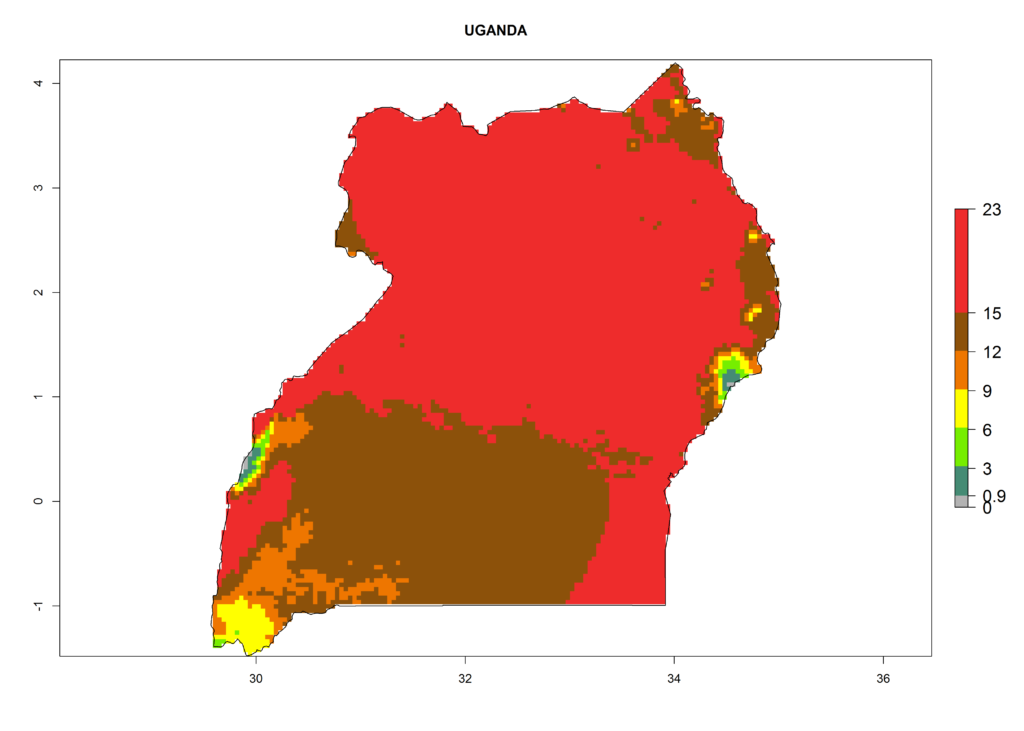
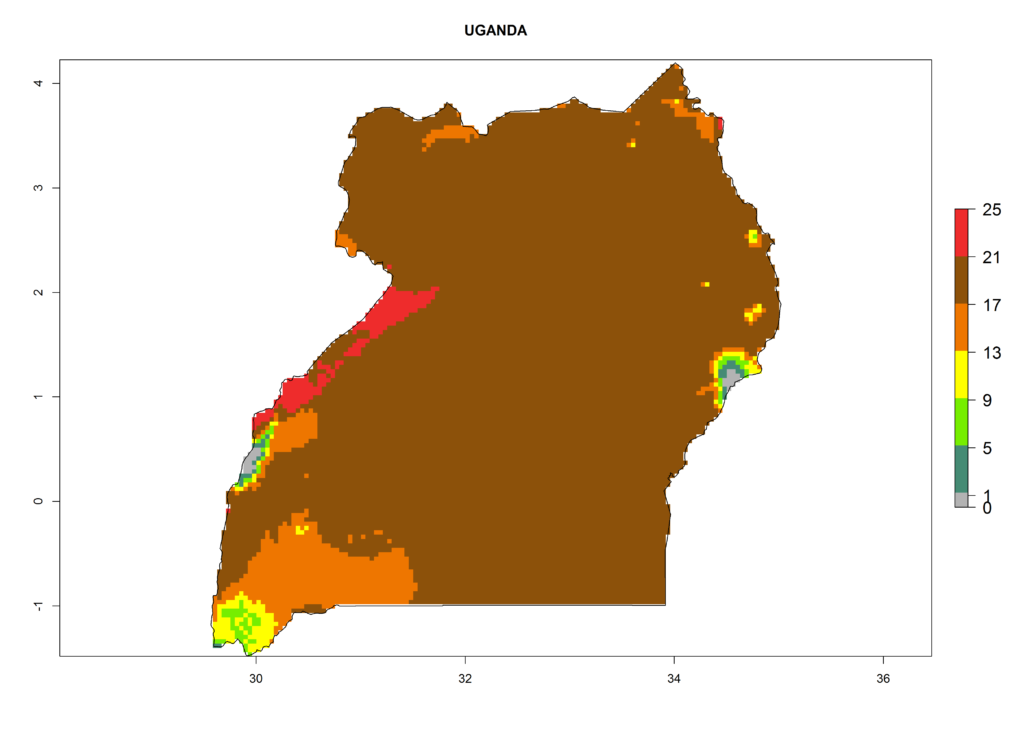
Under the present climate, L. huidobrensis represents a severe risk (ERI>0.95) in East Africa, which is associated with a GI>12–24 generations per year and a high AI (AI>15) (Fig. 6). The highest risks of establishment (ERI>0.95, red zones) are observed in production zones of each country. In the case of Rwanda (Fig. 6d), the entire country is situated at an altitude of >900 masl and characterized by a mostly temperate tropical highland climate. Rwanda presents the largest area at high risk of establishment, and L. huidobrensis is known to be highly damaging in potato and bean production. In Mediterranean countries (e.g., Egypt and Morocco), the risk of establishment is lower (ERI<0.9), except for those zones near the coast (ERI>0.95) (Fig. 6a, c). At coastal zones the GI gives values between 12 (Morocco) and 15 (Egypt) generations per year and with an AI<13 for Morocco and an AI<17 for Egypt.
| ERI | GI | AI |
| a) Egypt | 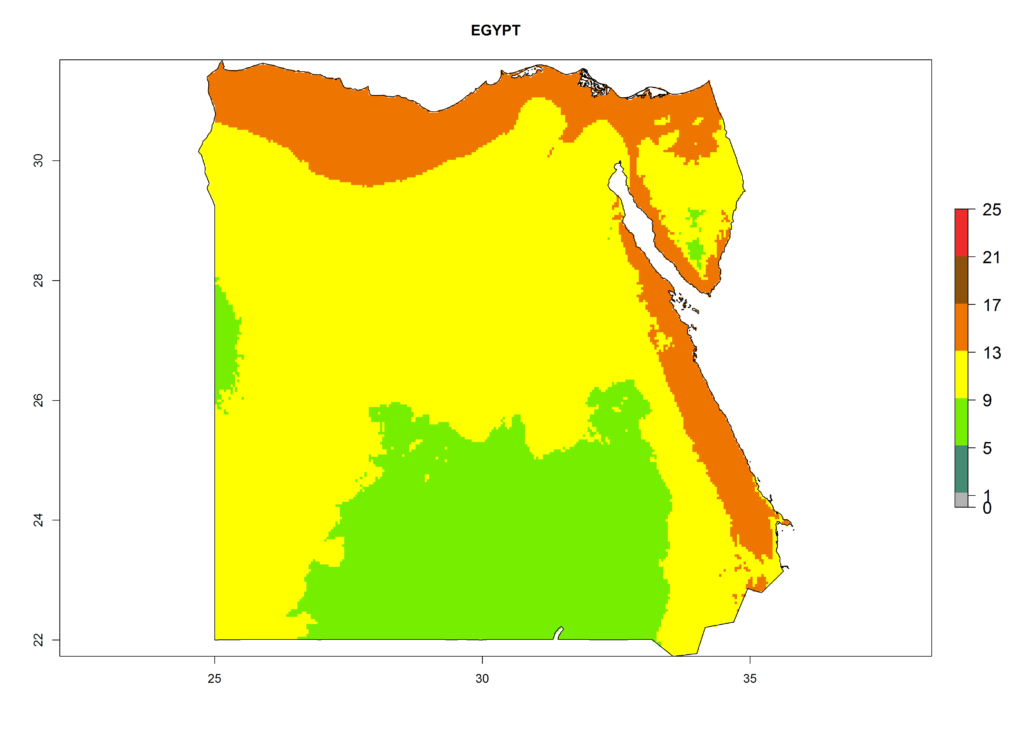 |
|
| b) Kenya | ||
| c) Morocco | ||
| d) Rwanda | ||
| e) Tanzania | ||
| f) Uganda |
Figure 6. ERI, abundance (GI, potential damage), and activity (AI, potential population growth) of the serpentine leafminer fly, Liriomyza huidobrensis, in selected African countries according to model predictions for the year 2000. An ERI>0.7 is associated with potential permanent establishment.
Phytosanitary measures
To avoid the introduction of L. huidobrensis (and other leafminer species) into European countries, the European Plant Protection Organisation recommended that propagating material (except seeds) of chili pepper, carnations, celery, chrysanthemums, melon, Gerbera, Gypsophila sp., lettuces, Senecio hybridus, and tomatoes from countries where the pests occur must have been inspected at least every month during the previous 3 months and found to be free of pests. A phytosanitary certificate should be required for cut flowers and for vegetables with leaves. Studies on Liriomyza have shown that newly laid eggs in chrysanthemums can survive cold storage at 0°C for up to 3 weeks, but that larvae were killed in 1–2 weeks under the same conditions. However, before cold storage, cuttings of infested plants should be maintained under normal glasshouse conditions for 3 or 4 days in order to allow eggs to hatch. This is required to ensure the elimination of the most resistant stage of the pest.
Adaptation to risk avoidance at farm level
Ecological and economical sound control of the leafminer fly is best realized when based on integrated pest management by promoting natural regulation and combining cultural practices with physical and chemical control.
Monitoring pest populations. Counting the number of flies captured in yellow sticky traps monitors adult leafminer fly activity. Counting the number of larvae or fresh tunnels per leaflet by sampling the bottom, middle, and top parts of the plant is used to monitor larvae infestation. Both methods can be adapted for decision-making to avoid unnecessary applications of insecticides.
Conserving beneficial insects. Leafminer flies are controlled by a large number of beneficial insects, which are either predators or parasitoids. The species involved are very diverse and composition strongly changes among agro-ecologies where the leafminer fly occurs. In regions rich in natural enemies, it has been shown that leafminer flies are less damaging. Strategies to conserve beneficial insects can be manifold and include diversified cropping systems, high structural floristic diversity in agricultural landscapes, special weed management practices, and reduced use of selective insecticides.
Crop management. Healthy, vigorously growing potato plants can counteract the damaging effect of leafminers, particularly during the vegetative phase. The fast-growing foliage enhances the egg extrusion reaction of the foliar tissue. Plants deficient in irrigation, water, and fertilizer—or evolved from low-quality seed (e.g., virus-infested seed)—show damage much earlier and mined leaves dry more rapidly. Under these circumstances, the negative effects of leafminer fly infestations further reduce yields due to inadequate agronomic practices. Balanced N-fertilization is important as high N-content in leaves promotes leafminer fly development. Continuous food availability by re-planting hosts crop will favor the abundance of the leafminer fly. Rotation with non-hosts is therefore recommended.
Physical control. Yellow attracts leafminer fly adults. The use of mobile and stationary yellow sticky traps has effectively reduced leafminer fly adult populations, thereby decreasing insecticide application costs by more than 50%.
Use of control thresholds and crop variety selection. Potato varieties can tolerate considerable levels of foliar damage by the leafminer fly before control measures are needed. Pest infestation-crop loss relationships indicated that the accumulated foliar damage up to the growth stages of flowering and berry formation produced highest yield losses in the different potato varieties. Level of foliar injury at which control measures should start to prevent the pest population from reaching an economic injury level has been studied in different potato varieties: ‘Desiree’ (17–22%), ‘Revolucion’ (27–38%), ‘Canchan’ (25–33%), ‘Maria Tambeña’ (32–42%), ‘Tomasa’ (44–60%), and ‘Yungay’ (32–43%). Preemptive insecticidal control is economically not justified until foliar injury exceeds these values. Late-maturing varieties (‘Tomasa’, ‘Yungay’) generally compensate higher injury levels better and hence economic injury levels were higher than for early-maturing varieties (‘Desiree’, ‘Revolucion’).
Chemical control. Insecticides must be used only according to the monitoring results and when the leafminer population is expected to cause economic damage. Studies have shown that with frequent sprayings, leafminer flies have grown rapidly resistant to insecticides. Systemic insecticides with translaminar properties are most effective in controlling leafminer fly larvae. The active ingredients ciromazine, abamectin, and spinosad can be recommended. Ciromazine has a residual effect of more than 20 days, followed by abamectin and spinosad. Abamectin is most specific with fewer negative effects on beneficial insects.
Further reading
Andersen, A., and T. Hofsvang. 2010. Pest risk assessment of the South American Leafminer Liriomyza huidobrensis in Norway. Opinion of the Panel on Plant Health of the Norwegian Scientific Committee for Food Safety, 09/904-3 final, 46 pp. SBN 978-82-8082-399-1 (electronic edition). pp. 46, VKM, Oslo, Norway. Available from http://www.vkm.no/dav/ 89df414b97.pdf
CABI. 2012. Liriomyza huidobrensis (Blanchard). Invasive Species Compendium: Datasheets, maps, images, abstracts and full text on invasive species of the world. Available from http://www.cabi.org/isc/datasheet/30956
Chen, B., and L. Kang. 2004. Variation in cold hardiness of Liriomyza huidobrensis (Diptera: Agromyzidae) along latitudinal gradients. Environmental Entomology 33: 155–164.
Cisneros, F., and N. Mujica. 1999. The leafminer fly in potato: plant reaction and natural enemies as natural mortality factors. Lima, Peru, International Potato Center, 129–140.
European Food Safety Authority (EFSA). 2012. Panel on Plant Health (PLH); Scientific Opinion on the risks to plant health posed by Liriomyza huidobrensis (Blanchard) and Liriomyza trifolii (Burgess) to the EU territory with the identification and evaluation of risk reduction options. EFSA Journal 10(12): 3028. [190 pp.] doi:10.2903/j.efsa.2012.3028. Available online: www.efsa.europa.eu/efsajournal
European Plant Protection Organization (EPPO). 2013a. Data Sheets on Quarantine Pests: Liriomyza huidobrensis, 6 pp. Prepared by CABI and EPPO for the EU under Contract 90/399003. Available from https://www.eppo.int/QUARANTINE/insects/Liriomyza_huidobrensis/ LIRIHU_ds.pdf
Food and Environment Research Agency (Fera). 2012. Consultation on the plant health status of Liriomyza species. 6 April 2012, 25 pp. Available from http://www. fera.defra.gov.uk/plants/plantHealth/pestsDiseases/ documents/lirioConsultPaper.pdf
Gitonga, Z.M., A. Chabi-Olaye, D. Mithofer, J.J. Okello, and C.N. Ritho. 2010. Control of invasive Liriomyza leafminer species and compliance with food safety standards by small scale snow pea farmers in Kenya. Crop Protection 29: 1472–1477.
Kang, L., B. Chen, J-N Wei, and T-X Liu. 2009. Roles of thermal adaptation and chemical ecology in Liriomyza distribution and control. Annual Review of Entomology 54: 127–145.
Lanzoni, A., G.G. Bazzocchi, G. Burgio, and M.R. Fiacconi. 2002. Comparative life history of Liriomyza trifolii and Liriomyza huidobrensis (Diptera: Agromyzidae) on beans: effect of temperature on development. Environmental Entomology 31: 797–803.
Martin, A.D., R.H. Hallett, M.K. Sears, and M.R. McDonald. 2005. Overwintering Ability of Liriomyza huidobrensis (Blanchard) (Diptera: Agromyzidae) in Southern Ontario, Canada. Environmental Entomology 34: 743–747.
Milla, K., and S. Reitz. 2005. Spatial/temporal model for survivability of pea leafminer (Liriomyza huidobrensis) in warm climates: a case study in south Florida, USA. European Journal of Scientific Research 7: 65–73.
Plant Health Australia. 2009. Threat Specific Contingency Plan: Serpentine leaf miner Liriomyza huidobrensis. Industry Biosecurity Plan for the Nursery & Garden Industry: 42 pp.
Scheffer, S.J. 2000. Molecular evidence of cryptic species within the Liriomyza huidobrensis (Diptera: Agromyzidae). Journal of Economic Entomology 93: 1146–1151.
Spencer, K.A. 1990. Host specialization in the world Agromyzidae (Diptera). Dordrecht, Netherlands: Kluwer Academic Publishers, 444 pp.
Weintraub, P.G. 2001. Effects of cyromazine and abamectin on the pea leafminer Liriomyza huidobrensis (Diptera: Agromyzidae) and its parasitoid Diglyphus isaea (Hymenoptera: Eulophidae) in potatoes. Crop Protection 20: 207–213.