Cryopreservation
Cryopreservation is an effective and efficient strategy for the long-term conservation of clonally propagated crops. Plant material is stored at ultra-low temperatures in the vapor or liquid phase of liquid nitrogen (-196°C) and at this temperature physiological, chemical, and metabolic activities slow to an extremely low rate. Germplasm can theoretically remain viable for centuries.
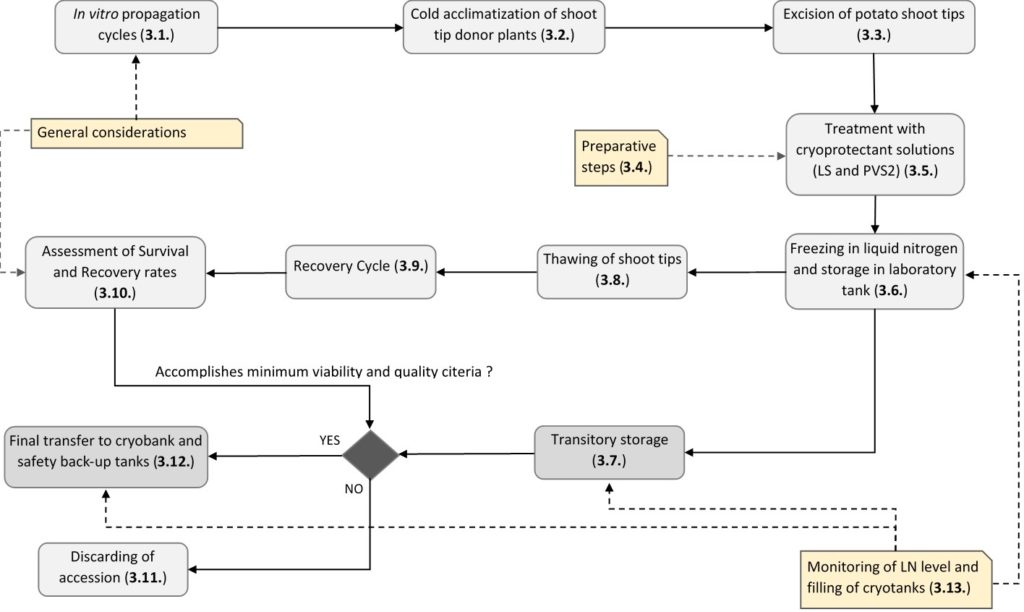
CIP began cryopreserving potatoes in 1996 and after experimenting with a wide range of protocols and variables, the CIP genebank adopted a PVS2-droplet method, originally developed for the Musa (banana) collection. The CIP genebank has improved the protocol where to date the genebank successfully cryopreserves >450 potato accessions per year.
In 2013, strict standards for viability assessment, decision-making (thresholds on material to keep or discard), operation, health and safety, and quality control were implemented to ensure long-term fidelity of the cryopreserved germplasm. A crucial part of our standards are that cryopreserved shoot tips maintain the capacity to develop into morphologically normal in vitro plants with a functional apex, stem, leaves, and roots, without any intermediate callus formation after removing them from liquid nitrogen (LN) and passing them through the post-thawing recovery steps.
Additionally, we implemented a long-term (50+ year) viability-monitoring program for potato. Each year we are cryopreserving 240 shoot tips of 10 accessions. Under this program, thirty shoot tips are removed from LN for initial viability testing and then another 30 shoot tips after 2, 4, 8, 16 and 32 years with 60 samples per accession still in the bank for future genebank managers to decide when to test the last two intervals (64 and 128 years). This experiment will allow future generations to have tools to monitor viability of cryopreserved germplasm over long term storage.











![Effect of preculture and PVS2 exposure on the cryopreservation of sweet potato shoot tips [Ipomoea batatas (L.) Lam.] using the PVS2 droplet vitrification.](https://cipotato.org/genebankcip/wp-content/uploads/sites/3/2023/08/78382-2.jpg)